ZANTAC
ranitidine hydrochloride
CAS: 71130-06-8;66357-59-3
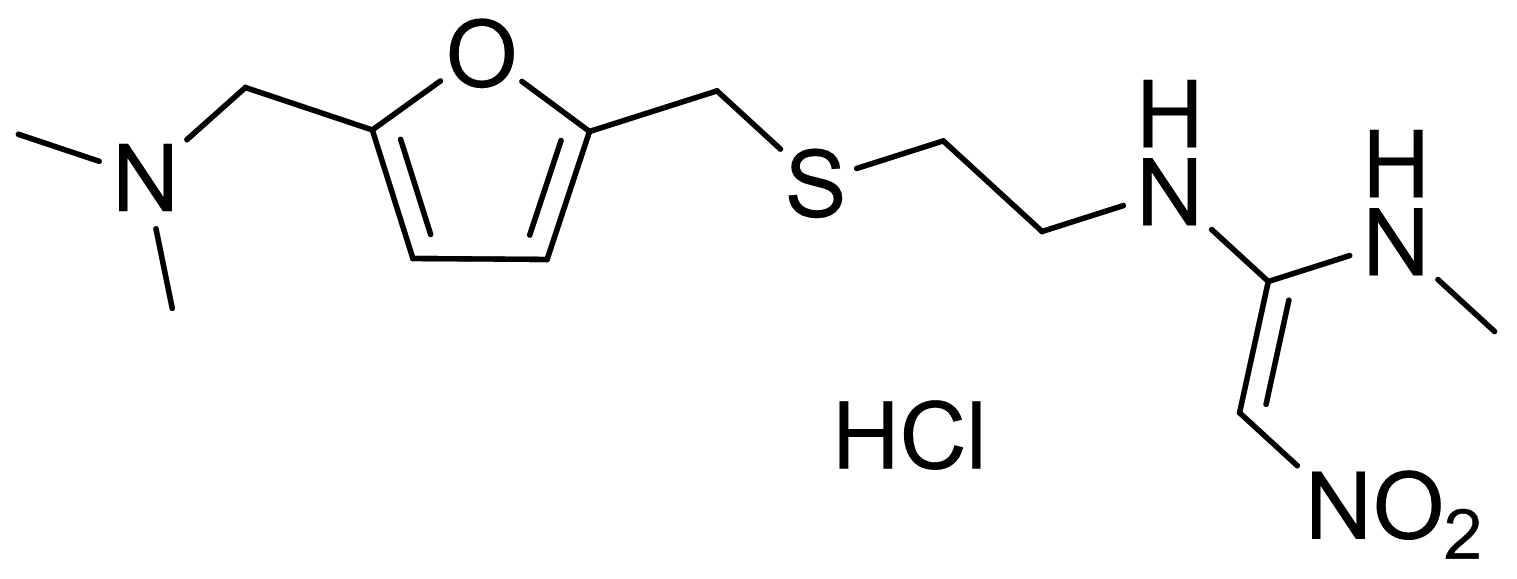
Molecular Formula: C13H23ClN4O3S
ZANTAC - Names and Identifiers
| Name | ranitidine hydrochloride |
| Synonyms | ZANTAC Noctone Ranitidine hcl RANITIDINE HCL RANITIDINE (ranitidine) RanitidineHydrochloride ranitidine hydrochloride Ranitidine Hydrochlooride -N-(2-(((5-((Dimethylamino) -N-methyl-2-nitroethene-1,1-diamine [(Dimethylamino)methyl]furfuryl]thio]ethyl]-N'-methyl-2-nitrovinylidenediamine monohydrochloride N-[2-[[5-[(dimethylamino)methyl]furfuryl]thio]ethyl]-N'-methyl-2-nitrovinylidenediamine monohydrochloride N-[2-[[[5-[(DIMETHYLAMINO)METHYL]-2-FURANYL]METHYL]THIO]ETHYL]-N'-METHYL-2-NITRO-1,1-ETHANEDIAMINE HYDROCHLORIDE N-[2-[[[-5-[(DIMETHYLAMINO)METHYL]-2-FURANYL]METHYL]THIO]ETHYL]-N'-METHYL-2-NITRO-1,1 ETHENEDIAMINE, HYDROCHLORIDE (E)-N-{2-[({5-[(dimethylamino)methyl]furan-2-yl}methyl)sulfanyl]ethyl}-N'-methyl-2-nitroethene-1,1-diamine hydrochloride |
| CAS | 71130-06-8 66357-59-3 |
| EINECS | 275-207-4 |
| InChI | InChI=1/C12H20N4O3S.ClH/c1-13-11(8-16(17)18)14-6-7-20-9-10-4-5-12(19-10)15(2)3;/h4-5,8,13-14H,6-7,9H2,1-3H3;1H/b11-8+ |
ZANTAC - Physico-chemical Properties
| Molecular Formula | C13H23ClN4O3S |
| Molar Mass | 350.86 |
| Melting Point | 134°C |
| Boling Point | 437.1 °C at 760 mmHg |
| Solubility | H2O: 1.8 mg/mL |
| Appearance | solid |
| Color | tan |
| Storage Condition | 2-8℃ |
| Sensitive | Sensitive to air and heat |
| MDL | MFCD00069339 |
| Physical and Chemical Properties | This product is a white or light yellow crystalline powder, there are different odor, taste slightly bitter with astringent, easy to deliquescence, after absorbing moisture color darkened. This product is soluble in water or methanol, slightly soluble in ethanol, almost insoluble in acetone. |
| Use | Can reduce gastric acid secretion, promote ulcer healing. |
ZANTAC - Risk and Safety
| Safety Description | S22 - Do not breathe dust. S24/25 - Avoid contact with skin and eyes. |
| WGK Germany | 2 |
| RTECS | KM6557000 |
ZANTAC - Standard
Authoritative Data Verified Data
This product is N-methyl-N-[2-[[5-[(dimethylamino)] methyl]-2-furyl] methyl] Thio] ethyl] -2-nitro-1, 1-ethylenediamine hydrochloride. The content of C13H22N403S • HC1 shall be between 97.5% and 102.0% calculated as dry.
ZANTAC - Trait
Authoritative Data Verified Data
- This product is a white to light yellow crystalline powder; There is a bad odor; Easy to deliquescence, after moisture absorption, the color darkened.
- This product is soluble in water or methanol, slightly soluble in ethanol, almost insoluble in acetone.
ZANTAC - Differential diagnosis
Authoritative Data Verified Data
- take about 0.2g of this product, put it in a test tube, and slowly heat it with small fire. The gas generated can make the wet lead acetate test paper black.
- in the spectrum recorded under the content determination item, the retention time of the main peak of the test solution should be consistent with the retention time of the main peak of the reference solution.
- take this product, add water to make a solution containing 10ug per 1 ml, according to ultraviolet-visible spectrophotometry (General 0401), there is a maximum absorption at the wavelength of 228nm and 314nm.
- The infrared absorption spectrum of this product should be consistent with that of the control (Spectrum set 401).
- the aqueous solution of this product was chloride identification (1) of the reaction (General 0301).
- items (2), (4) and (5) or (1), (2), (3) and (5) are optional.
ZANTAC - Exam
Authoritative Data Verified Data
acidity
take 0.20g of this product, Add 10ml of water to dissolve, and measure according to law (General rule 0631). The pH value should be 4.5~6.5.
clarity and color of solution
take this product l. Add water to dissolve 100ml, the solution should be clear and colorless; If the color is colored, it should not be deeper compared with the yellow No. 3 Standard Colorimetric solution (General rule 0901 first method).
Related substances
take this product, add water to dissolve and dilute to make a solution containing about 1 mg per 1ml as a test solution; Take 1ml for precision measurement, put it in a 100ml measuring flask, dilute it with water to the scale, as a control solution. According to the chromatographic conditions under the content determination item, 10ul of each of the test solution and the control solution are accurately measured and injected into the liquid chromatograph respectively, and the chromatograms are recorded. If there are impurity peaks in the chromatogram of the test solution, the area of the impurity peak with a relative retention time of about 1.75 shall not be greater than 0.5 times (0.5%) of the area of the main peak of the control solution, other single impurity peak area shall not be greater than 0.2 times (0.2%) of the main peak area of the control solution, and the sum of other impurity peak areas shall not be greater than 0.5 times (0.5%) of the main peak area of the control solution. The chromatogram of the test solution is 0.05 times smaller than the main peak area of the control solution.
loss on drying
taking this product, using phosphorus pentoxide as desiccant, drying under reduced pressure at 60°C for 4 hours, the weight loss shall not exceed 0.75% (General rule 0831).
ignition residue
take l.Og of this product and check it according to law (General rule 0841). The residue left shall not exceed 0.1%.
Heavy metals
The residue left under the item of taking the ignition residue shall not contain more than 20 parts per million of heavy metal when examined by law (General rule 0821, Law II).
ZANTAC - Content determination
Authoritative Data Verified Data
measured by high performance liquid chromatography (General 0512).
chromatographic conditions and system suitability test
using octa-alkyl silane-bonded silica gel as A filler (krolmasil C18,4.6mm X 150mm, 5um or equivalent chromatographic column); Mobile phase A was phosphate buffer (6.8 of phosphoric acid was taken into 1900ml of water, add 50% of 8.6 sodium hydroxide solution, add water to 50%, adjust pH to 7.1±0.05 with phosphoric acid or sodium hydroxide solution-acetonitrile (98:2 ), mobile Phase B was phosphate buffer-acetonitrile (78:22 ) ; Gradient elution was carried out as follows; Detection wavelength was 230nm; Flow rate was 1.5 ml per minute; Column temperature was 35°C. Take ranitidine hydrochloride about O.lg, put in a 100ml measuring flask, add 1ml of 50% sodium hydroxide solution, add about 60ml of water, shake to dissolve, dilute with water to the scale, shake well, and place at room temperature for 1 hour, 10UL was injected into the liquid chromatograph and the chromatogram was recorded. The flow rate or mobile phase ratio is adjusted so that the retention time of the main component chromatographic peak is about 12 minutes, the relative retention time of impurity I is about 0.85, and the separation degree of ranitidine peak and impurity I peak should be greater than 4.0.
assay
take about 22mg of this product, accurately weigh it, put it in a 200ml measuring flask, add water to dissolve and dilute it to the scale, shake it well, and use it as a test solution, A sample of 10ul was injected into the liquid chromatograph accurately, and the chromatogram was recorded. Another 22mg of ranitidine hydrochloride reference substance was taken, and the same method was used for determination. According to the external standard method to calculate the peak area, that is.
ZANTAC - Category
Authoritative Data Verified Data
H2 receptor antagonist.
ZANTAC - Storage
Authoritative Data Verified Data
It was shielded from light, sealed, and stored in a cool-dark dry place.
ZANTAC - Ranitidine hydrochloride tablets
Authoritative Data Verified Data
This product contains ranitidine hydrochloride according to ranitidine (C13H22N403S) calculation, should be the label amount of 93.0% ~ 107.0%.
trait
This product is sugar-coated tablet or film-coated tablet, which shows off-white to yellowish after removing the coating.
identification
- take the product, remove the coating, triturate, weigh an appropriate amount (about 0.2g equivalent to ranitidine), and show the same reaction according to the identification (1) Test under the item of ranitidine hydrochloride.
- in the chromatogram recorded under the content determination item, the retention time of the main peak of the test solution should be consistent with the retention time of the main peak of the reference solution.
- an appropriate amount of the fine powder under identification (1) was taken, and the filtrate was filtered by shaking with water. The filtrate showed chloride to identify the reaction of (1) (General 0301).
examination
- Related substances take an appropriate amount of fine powder of this product (about equivalent to ranitidine lOOmg ), put it in a 100ml measuring flask, add water to dissolve ranitidine hydrochloride and dilute to the scale, shake well, filter, the filtrate was taken as a test solution; 1 ml was accurately measured, placed in a 100ml measuring flask, diluted with water to scale, and shaken to serve as a control solution. According to the chromatographic conditions under the content determination item, respectively inject l0ul of the control solution and the test solution into the liquid chromatograph to record the chromatogram. If there are impurity peaks in the chromatogram of the test solution, the area of a single impurity peak shall not be greater than the area of the main peak of the control solution (1.0%), and the sum of each impurity peak area shall not be greater than 2 times the area of the main peak of the control solution (2.0%). The chromatogram of the test solution is 0.05 times smaller than the main peak area of the control solution.
- the dissolution of this product, according to the dissolution and release determination method (General rule 0931 second method), water 900ml as the dissolution medium, the speed of 50 rpm, according to the law, after 45 minutes, take 20ml of the solution, filter, Take 5ml of the filtrate accurately, put it in a 50ml measuring flask, dilute it with water to the scale, shake it well, the absorbance was measured at a wavelength of 0401 NM according to UV-Vis spectrophotometry (General rule 495), and the elution amount of each tablet was calculated as the absorption coefficient of C13H22N403S was. The limit is 80% of the labeled amount and shall be in accordance with the provisions.
- others shall be in accordance with the relevant provisions under the item of tablets (General rule 0101).
Content determination
- measured by high performance liquid chromatography (General 0512).
- chromatographic conditions and system suitability test using eighteen alkyl silane bonded silica gel as the filler (krolmasil C18,4.6mm X 150mm, 5um or performance equivalent column); the mobile phase A is phosphate buffer (6.8 of phosphoric acid is taken into 1900ml of water, 50% of 8.6 sodium hydroxide solution is added, and water is added to 50%, and the pH value is adjusted to 7.1±0.05 with phosphoric acid or sodium hydroxide solution)-Acetonitrile (98:2 ), mobile phase B was phosphate buffer-acetonitrile (78:22); Gradient elution was carried out according to the following table; Detection wavelength was 230nm. The flow rate was 1.5 per minute; The column temperature was 35°C. Take about 0.lg ranitidine hydrochloride, put it in a 100ml measuring flask, add 1ml of 50% sodium hydroxide solution, add about 60ml of water, shake to dissolve, dilute to the scale with water, shake well, and place it at room temperature for 1 hour, 10UL was injected into the liquid chromatograph and the chromatogram was recorded. The flow rate or mobile phase ratio is adjusted so that the retention time of the main component chromatographic peak is about 12 minutes, the relative retention time of impurity I is about 0.85, and the separation degree of ranitidine peak and impurity I peak should be greater than 4.0.
- determination Method: Take 20 tablets of this product, remove the coating of the sugar-coated tablets, precise weighing, fine grinding, take an appropriate amount of fine powder (about 20mg equivalent to ranitidine), precise weighing, and put it in a 200ml measuring flask, add water to dissolve ranitidine hydrochloride and dilute to the scale, shake, filter, take the filtrate as a test solution, take 10ul of precision, inject human liquid chromatography, record the chromatogram; another 22mg of ranitidine hydrochloride was added into a 200ml measuring flask, dissolved with water and diluted to the scale. According to the external standard method to calculate the peak area, and the result is multiplied by 0.8961, that is.
category
with ranitidine hydrochloride.
specification
Based on C13H22N403S (l )75mg (2)150mg
storage
light-shielded, sealed, and stored in a dry place.
ZANTAC - Ranitidine hydrochloride effervescent granules
Authoritative Data Verified Data
This product contains ranitidine hydrochloride according to ranitidine (C13H22N403S) calculation, should be the label amount of 90.0% ~ 110.0%.
trait
This product is light yellow granules, gas aroma, taste sweet; Water is effervescent.
identification
- take an appropriate amount of this product (about 0.2g equivalent to ranitidine), place it in a test tube, and slowly heat it with a small fire. The generated gas can make the wet lead acetate test paper black.
- in the chromatogram recorded under the content determination item, the retention time of the main peak of the test solution should be consistent with the retention time of the main peak of the reference solution.
- take an appropriate amount of this product, add water, shake, filter, filtrate shows chloride to identify (1) reaction (General rule 0301).
examination
- relevant substances take an appropriate amount of this product (about equivalent to ranitidine lOOmg ), put it in a 100ml measuring flask, shake with water to dissolve ranitidine hydrochloride, dilute it with water to the scale, and shake well, as a test solution; 1ml was accurately measured, placed in a 100ml measuring flask, diluted with water to scale, and shaken to serve as a control solution. According to the chromatographic conditions under the content determination item, 10ul of each of the control solution and the test solution are accurately measured and injected into the liquid chromatograph respectively, and the chromatograms are recorded. An appropriate amount of aspartame was taken, dissolved with water and diluted to make a solution containing about 0.37mg per 1 ml. 10u1 was injected into the liquid chromatograph and the chromatogram was recorded. If there are impurity peaks in the chromatogram of the test solution, the peaks of excipients and aspartame before the relative retention time of 0.15 shall be subtracted, and the area of a single impurity peak shall not be greater than the area of the main peak of the control solution (1.0%), the sum of each impurity peak area shall not be greater than 2 times (2.0%) of the main peak area of the control solution. The chromatogram of the test solution is 0.05 times smaller than the main peak area of the control solution.
- weight loss on drying this product shall be dried under reduced pressure at 80°C for 4 hours with phosphorus pentoxide as desiccant, and the weight loss shall not exceed 2.0% (General rule 0831).
- others should comply with the relevant provisions under The granule (General Principle 0104).
Content determination
- measured by high performance liquid chromatography (General 0512).
- chromatographic conditions and system suitability test using eighteen alkyl silane-bonded silica gel as a filler (krolmasil C18,4.6mm X 150mm, 5um or performance equivalent column) T the mobile phase A is phosphate buffer (Take 6.8ml of phosphoric acid into 50% ml of water, add 8.6ml of 50% sodium hydroxide solution, add water to 7.1 ml, and adjust the pH value to 0.05 ± with phosphoric acid or sodium hydroxide solution)-Acetonitrile (98:2 ), mobile phase B was phosphate buffer-acetonitrile (78:22); Gradient elution was carried out according to the following table; Detection wavelength was 230nm. The flow rate was 1.5ml per minute; The column temperature was 35°C. Take about 0.lg ranitidine hydrochloride, put it in a 100ml measuring flask, add 1ml of 50% sodium hydroxide solution, add about 60ml of water, shake to dissolve, dilute to the scale with water, shake well, filter, after standing at room temperature for 1 hour, 10u1 was injected into the liquid chromatograph, and the chromatogram was recorded. The flow rate or mobile phase ratio is adjusted so that the retention time of the main component chromatographic peak is about 12 minutes, the relative retention time of impurity I is about 0.85, and the separation degree of ranitidine peak and impurity I peak should be greater than 4.0.
- determination: take an appropriate amount of this product (about 20mg equivalent to ranitidine), weigh it accurately, put it in a 200ml measuring flask, shake it with water to dissolve ranitidine hydrochloride and dilute it to the scale, shake it well, filter it, the continuous filtrate was taken as the test solution, and 10u1 was injected into the liquid chromatograph for precise measurement. The chromatogram was recorded, ranitidine hydrochloride was dissolved and diluted to the scale by shaking with water, and then was determined by the same method. According to the external standard method to calculate the peak area, and the result is multiplied by 0.8961, that is.
category
with ranitidine hydrochloride.
specification
1.5g per bag (including 0.15g of C13H22N403S)
storage
shade, seal, and store in a cool and dry place.
ZANTAC - Ranitidine hydrochloride injection
Authoritative Data Verified Data
This product is a sterilized aqueous solution of ranitidine hydrochloride. The content of ranitidine hydrochloride shall be 93.0% to 107.0% of the labeled amount calculated as ranitidine.
trait
This product is colorless to light yellow clear liquid.
identification
- take 2ml of this product, put it in a test tube, evaporate it on a water bath, and slowly heat it with a small fire. The gas generated can make the wet lead acetate test paper black.
- take this product, according to the item of ranitidine hydrochloride identification (3), (5) test, showed the same results.
- in the spectrum recorded under the content determination item, the retention time of the main peak of the test solution should be consistent with the retention time of the main peak of the reference solution.
examination
- the pH value should be 6.5 to 7.5 (General 0631).
- the color of this product, compared with the yellow No. 4 standard colorimetric liquid (General Principles 0901 first method), should not be deeper.
- appropriate amount of related substances should be taken and diluted with water to make a solution containing ranitidine 1 mg per 1ml, which should be used as a test solution; 1ml should be accurately measured and placed in a 100ml measuring flask, as a control solution, it was diluted to the scale with water and shaken. According to the chromatographic conditions under the content determination item, respectively inject the control solution and the test solution into the liquid chromatograph to record the chromatogram. If there are impurity peaks in the chromatogram of the test solution, the area of a single impurity peak shall not be greater than 1.5 times (1.5%) of the area of the main peak of the control solution, the sum of each impurity peak area shall not be greater than 2.5 times (2.5%) of the main peak area of the control solution. The chromatogram of the test solution is 0.05 times smaller than the main peak area of the control solution.
- bacterial endotoxin take this product, according to the inspection (General 1143 ), the amount of endotoxin per 1 mg ranitidine should be less than 1.0 EU.
- sterile take this product, by membrane filtration method, each membrane with 0.1% peptone solution rinse 1 time, with Staphylococcus aureus as positive control bacteria, according to law inspection (General rule 1101), the requirements shall be met.
- others should comply with the relevant provisions under injection (General 0102).
Content determination
- measured by high performance liquid chromatography (General 0512).
- chromatographic conditions and system suitability test using eighteen alkyl silane bonded silica gel as the filler (krolmasil C18 , 4.6mm X 150mm, 5um or performance equivalent column); the mobile phase A was phosphate buffer (6.8 of phosphoric acid was placed in 1900ml of water, 50% of 8.6 sodium hydroxide solution was added, and water was added to 50%, and the pH value was adjusted to 7.1±0.05 with phosphoric acid or sodium hydroxide solution)-Acetonitrile (98:2 ), mobile phase B was phosphate buffer-acetonitrile (78:22 ) ; Gradient elution was carried out according to the following table; Detection wavelength was 230nm. The flow rate was 1.5 per minute; The column temperature was 35°C. Take about 0.lg ranitidine hydrochloride, put it in a 100ml measuring flask, add 1ml of 50% sodium hydroxide solution, add about 60ml of water, shake to dissolve, dilute to the scale with water, shake well, and place it at room temperature for 1 hour, 10u1 was injected into the liquid chromatograph and the chromatogram was recorded. The flow rate or mobile phase ratio is adjusted so that the retention time of the main component chromatographic peak is about 12 minutes, the relative retention time of impurity I is about 0.85, and the separation degree of ranitidine peak and impurity I peak should be greater than 4.0.
- determination of precision take an appropriate amount of this product, diluted with water to make about ranitidine per 1 ml. lmg solution, as a test solution, take 10u1 injection liquid chromatograph for precise measurement, record the chromatogram; Take 22mg of ranitidine hydrochloride reference substance, weigh it accurately, put it in 200ml measuring flask, add water to dissolve and dilute to the scale, shake, the same method. According to the external standard method to calculate the peak area, and the result is multiplied by 0.8961, that is.
category
with ranitidine hydrochloride.
specification
C13H22N403S (l) 2ml:50mg(2)5ml:50Mg
storage
light shielding, closed storage.
ZANTAC - Ranitidine Hydrochloride Capsules
Authoritative Data Verified Data
This product contains ranitidine hydrochloride by ranitidine (C13H22N403S) calculation, should be the standard amount of 93.0% ~ 107.0%.
trait
The contents of this product are white to yellow powder or granules.
identification
- an appropriate amount of the content of this product (about 0.2g of ranitidine) was taken, and the same reaction was shown according to the Identification Test (1) under the item of ranitidine hydrochloride.
- in the chromatogram recorded under the content determination item, the retention time of the main peak of the test solution should be consistent with the retention time of the main peak of the reference solution.
- take an appropriate amount of the contents of this product, add water, shake, filter, filtrate chloride identification (1) of the reaction (General 0301).
examination
- Related substances take an appropriate amount of the contents of this product (about 100mg equivalent to ranitidine), put it in a 100ml measuring flask, add water to dissolve ranitidine hydrochloride and dilute to the scale, shake well, filter, the filtrate was taken as a test solution; 1 ml was accurately measured, placed in a 100ml measuring flask, diluted with water to scale, and shaken to serve as a control solution. According to the chromatographic conditions under the content determination item, 10ul of each of the control solution and the test solution are accurately measured and injected into the liquid chromatograph respectively, and the chromatograms are recorded. If there are impurity peaks in the chromatogram of the test solution, the area of a single impurity peak shall not be greater than the area of the main peak of the control solution (1.0%), and the sum of each impurity peak area shall not be greater than 2 times the area of the main peak of the control solution (2.0%). The chromatogram of the test solution is 0.05 times smaller than the main peak area of the control solution.
- loss on drying: take an appropriate amount of the contents of this product, use phosphorus pentoxide as a desiccant, and dry under reduced pressure at 60°C for 4 hours. The loss of weight shall not exceed 4.0% (General rule 0831).
- others should comply with the relevant provisions under the capsule (General 0103).
Content determination
- measured by high performance liquid chromatography (General 0512).
- chromatographic conditions and system suitability test using eighteen alkyl silane bonded silica gel as the filler (krolmasil C18,4.6mm X 150mm,5um or performance equivalent column); mobile phase A is phosphate buffer (Take 6.8ml of phosphoric acid into 50% ml of water, add 8.6ml of 50% sodium hydroxide solution, add water to 7.1±0.05 ml, adjust pH to with phosphoric acid or sodium hydroxide solution)-acetonitrile (98:2 ), mobile phase B was phosphate buffer-acetonitrile (78:22); Gradient elution was carried out according to the following table; The detection wavelength was 230nm. The flow rate was 1.5 per minute; The column temperature was 35°C. Take about 0.lg ranitidine hydrochloride, put it in a 100ml measuring flask, add 1ml of 50% sodium hydroxide solution, add about 60ml of water, shake to dissolve, dilute to the scale with water, shake well, and place it at room temperature for 1 hour, lule was injected into human liquid chromatograph, and the chromatogram was recorded. The flow rate or the mobile phase ratio is adjusted so that the retention time of the main component chromatographic peak is about 12 minutes, the relative retention time of impurity I is about 0.85, and the separation degree between ranitidine peak and impurity peak should be greater than 4.0.
- determine the contents under the item of loading difference, mix evenly, weigh an appropriate amount (about 20mg equivalent to ranitidine) accurately, put it in a 200ml measuring flask, add water to dissolve and dilute to the scale, shake well, as a test solution, inject 10ul into the liquid chromatograph with precision, record the chromatogram; Take 22mg of ranitidine hydrochloride control sample, weigh it accurately, put it into a 200ml measuring flask, add water to dissolve and dilute to the scale, shake, the same method. According to the external standard method to calculate the peak area, and the result is multiplied by 0.8961, that is.
category
with ranitidine hydrochloride.
specification
Based on C13H22N403S (l )75mg (2 ) lOOmg(3)150mg
storage
light-shielded, sealed, and stored in a dry place.
Supplier List
CAS: 66357-59-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 66357-59-3
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 71130-06-8
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 66357-59-3
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 66357-59-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 66357-59-3
Tel: +86-571-88162785
Email: Lucy@verypharm.com
Mobile: +86-13606544505
QQ: 472954302
Product List: View Catalog
CAS: 66357-59-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 66357-59-3
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 71130-06-8
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 66357-59-3
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 66357-59-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 66357-59-3
Tel: +86-571-88162785
Email: Lucy@verypharm.com
Mobile: +86-13606544505
QQ: 472954302
Product List: View Catalog




