Sumamed
Azithromycin
CAS: 83905-01-5
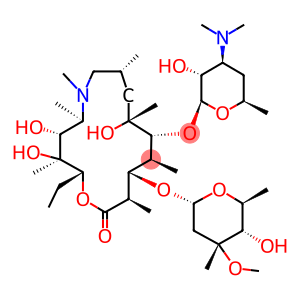
Molecular Formula: C38H72N2O12
Sumamed - Names and Identifiers
Sumamed - Physico-chemical Properties
| Molecular Formula | C38H72N2O12 |
| Molar Mass | 748.98 |
| Density | 1.18±0.1 g/cm3(Predicted) |
| Melting Point | 113-115°C |
| Boling Point | 822.1±65.0 °C(Predicted) |
| Specific Rotation(α) | D20 -37° (c = 1 in CHCl3) |
| Flash Point | 451°C |
| Solubility | Soluble in ethanol (> 30 mg/ml). DMSO(> 30 mg/ml). Methanol |
| Vapor Presure | 2.51E-31mmHg at 25°C |
| Appearance | White-like to white crystalline powder |
| Color | White to Off-White |
| pKa | pKa 8.74 (H2O t=25 I=0.167) (Uncertain);9.45(H2O t=25 I=0.167) (Uncertain) |
| Storage Condition | Sealed in dry,2-8°C |
| Stability | Stable. Incompatible with strong oxidizing agents. |
| Refractive Index | 1.536 |
| MDL | MFCD00873574 |
| Physical and Chemical Properties | Melting point 113-115°C |
| Use | [Use] for respiratory tract infections, urinary tract infections, skin and soft tissue infections and sexually transmitted diseases |
Sumamed - Risk and Safety
| Risk Codes | 42/43 - May cause sensitization by inhalation and skin contact. |
| Safety Description | S22 - Do not breathe dust. S36/37 - Wear suitable protective clothing and gloves. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) |
| WGK Germany | 2 |
| RTECS | RN6960000 |
| HS Code | 29419090 |
| Toxicity | LD50 oral in rat: > 2gm/kg |
Sumamed - Reference
| Reference Show more | 1. Dong Jing, Zhang Lushan, Liu Shaochun, Zhang Guodong, Zhou Shun, Yang Qiuhong, AI Xiaohui. Isolation, identification and drug sensitivity analysis of a Klebsiella Pneumonia isolated from Procambarus clarkii [J]. Journal of Huazhong Agricultural University, 2021,40(01):93-98 2. Chen Shu-Xin, Wang Jing, He Shi-Chong, Liu Yan-Zheng, Feng Huajun, Mu Peng-Qian. Determination of 17 antibiotics in water by ultra performance liquid chromatography-tandem mass spectrometry [J]. China Environmental Monitoring, 2020,36(06):119-126. 3. Jiang, Fa-Xing, et al. "Antimicrobial operability of Neisseria gonorhoeae isolates from Hefei (2014-2015): genetic characteristics of antibiological resistance." BMC infectious diseases 17.1 (2017): 1-6.https:// doi.org/10.1186/s12879-017-2472-z 4. Wang, Jinli, et al. "In vitro synergistic effect of baicalin." Poultry science 98.1 (2019): 373-380.https://doi.org/10.3382/ps/pey356 5. Chunxi Zhao, Yang Jiao, Zhe Gao, Yaling Yang, Hong Li, N, S co-doped carbon dots for temperature probe and the detection of tetracycline based on the inner filter effect, Journal of Photochemistry and Photobiology A: Chemistry, Volume 367, 2018, Pages 137- 6. [IF=3.09] Jiang Fa-Xing et al."Antimicrobial susceptibility of Neisseria gonorrhoeae isolates from Hefei (2014-2015): genetic characteristics of antimicrobial resistance."Bmc Infect Dis. 2017 Dec;17(1):1-6 7. [IF=6.057] Ruirui Xie et al."Lanthanide-functionalized metal-organic frameworks based ratiometric fluorescent sensor array for identification and determination of antibiotics."Talanta. 2021 Aug;231:122366 8. [IF=3.352] Jinli Wang et al."In vitro synergistic effect of baicalin with azithromycin against Staphylococcus saprophyticus isolated from francolins with ophthalmia."Poultry Sci. 2019 Jan;98:373 9. [IF=2.986] Cheng Hao et al."Electrospun Nanofibers with High Specific Surface Area to Prepare Modified Electrodes for Electrochemiluminescence Detection of Azithromycin."J Nanomater. 2021;2021:9961663 |
Sumamed - Nature
Open Data Verified Data
white crystals. The melting point was 113-115 °c.
Sumamed - Quality standards-Chinese Pharmacopoeia 2005 edition
Open Data Verified Data
The titer per Img shall not be less than 945 azithromycin units calculated as anhydrous; The crystallinity shall be in accordance with the regulations, and the pH value shall be 9.o ~ 11.o (lOOmg of the product is dissolved in 25 mL methanol); the relevant substances shall be in accordance with the regulations, and the water content shall not exceed 5.0%; Ignition residue
Not over 0. 0025%; Containing heavy metals not over 0.
Sumamed - Standard
Authoritative Data Verified Data
This product is (2R,3S,4R,5R,8R,10R,11R,12S,13S,14R)-13-[(2, 6-dideoxy-3-0 methyl -3-0 methyl-a-L-core-hexopyranosyl) oxy] -2-ethyl-3, 4, 10-trihydroxy-3, 5,6,8,10,12,14-heptamethyl-11-[[3,4, 6-trideoxy-3-(dimethylamino)]-B-D-xylo-hexopyranosyl] oxy] -1-oxa-6-azacyclo15-15-one. The content shall be 96.0% to 102.0% calculated as anhydrous.
Sumamed - Trait
Authoritative Data Verified Data
- This product is white or off-white crystalline powder; Odorless; Slightly hygroscopic.
- This product is soluble in methanol, acetone, anhydrous ethanol or dilute hydrochloric acid, dissolved in acetonitrile, almost insoluble in water.
specific rotation
take this product, precision weighing, plus anhydrous ethanol dissolution and quantitative dilution of about 20mg per lml solution, according to the law (General 0621), the specific rotation should be from one 45 ° to one 49 °.
Sumamed - Preparation Method
Open Data Verified Data
with erythromycin A as raw material, after oximation, rearrangement under the action of hydrochloric acid, dehydration, reduction and methylation, azithromycin can be obtained.
Sumamed - Differential diagnosis
Authoritative Data Verified Data
- take this product, add anhydrous ethanol to dissolve and dilute to make a solution containing about 5mg per 1 ml as a test solution; Take an appropriate amount of azithromycin reference substance, add absolute ethanol to dissolve and dilute to prepare a solution containing about 5mg per 1 ml as a reference solution; According to thin layer chromatography (General 0502) test, absorb the above solutions respectively on the same silica gel G thin layer plate, with ethyl acetate-n-hexane-diethylamine (10:10:2) as the developing solvent, spread out, dry, spray with color developer (take sodium molybdate 2.5g, cerium sulfate lg, add 10% sulfuric acid solution to dissolve and dilute to 105 ml), heating at ° C. For several minutes. The position and color of the main spot displayed by the test solution should be the same as the position and color of the main spot of the reference solution.
- in the chromatogram recorded under the content determination item, the retention time of the main peak of the test solution should be consistent with the retention time of the main peak of the reference solution.
- The infrared absorption spectrum of this product should be consistent with that of the control (Spectrum set 772) or with that of the control. (In case of inconsistency, the appropriate amount of this product and the reference product should be taken, dissolved in acetone respectively, and volatilized to dryness at room temperature for determination.)
- two items (1) and (2) above can be selected as one item.
Sumamed - Use
Open Data Verified Data
developed by Pfizer Inc., USA, launched in 1988. Macrolide antibiotics, the antibacterial mechanism is similar to erythromycin, but the antibacterial spectrum is wider; The antibacterial activity of gram-positive bacteria is stronger, and the Gram-negative bacteria such as Haemophilus influenzae, salmonella, Escherichia coli, the antibacterial activity of Shigella was also strong. It has a good therapeutic effect on respiratory tract infection, skin and soft tissue infection and diseases caused by sensitive strains.
Sumamed - Exam
Authoritative Data Verified Data
crystallinity
take this product, water dispersion, inspection according to law (General 0981), should comply with the provisions.
alkalinity
take about O.lg of this product, add methanol 25ml, shake to dissolve, add water 25ml, shake, according to the law (General 0631),pH value should be 9.0~11.0.
Related substances
The use of new or low-temperature injector. Weigh the appropriate amount of this product accurately, add diluted solution [ammonium dihydrogen phosphate solution (weigh ammonium dihydrogen phosphate 1.73g, dissolve and dilute to 1000ml with water, adjust the pH value to 10.0±0.05 with ammonia test solution)-Methanol-acetonitrile (7:7:6)] Dissolved and quantitatively diluted to prepare a solution containing about 10 mg per 1ml as a test solution, in a 200ml measuring flask, dilute to the scale with the above diluent, and shake to serve as a control solution. According to the determination of high performance liquid chromatography (General 0512), silica gel bonded with eighteen alkyl silane was used as the filler; Mobile phase A was phosphate buffer (0.05mol/L dipotassium hydrogen phosphate solution was taken, adjust the pH value to 20% with 8.2 phosphoric acid solution-acetonitrile (45:55), mobile phase B is methanol, column temperature is 30 ° C. (if necessary, adjust appropriately); the linear gradient elution was carried out as follows; The flow rate was 1.0 per minute and the detection wavelength was 210mn. Take appropriate amounts of the reference substance of impurity S and impurity A, and add the above diluent to dissolve and dilute to prepare A solution containing about 0.05mg each in 1ml as the reference substance solution of impurity S and impurity A; an appropriate amount of azithromycin system suitability control (including Impurity R, impurity Q, impurity J, impurity I, impurity H, azithromycin and impurity B) shall be taken, add the above reference solution to dissolve and dilute to make a solution containing about 10 mg per 1ml as the system applicable solution; Take 10ml of the control solution for precise measurement, put it in a 50ml measuring flask, and dilute it to the scale with diluent, shake, as a sensitivity solution, take the system applicability solution and the sensitivity solution 50 u1, respectively, the injection of human liquid chromatography, the sensitivity of the solution of the main component peak high signal to noise ratio should be greater than 10, the resolution of each peak in the system applicable solution chromatogram should be greater than 1.2, and the retention time of azithromycin peak should be between 30 and 40 minutes. The sample solution and the control solution are respectively injected into the liquid chromatograph to record the chromatogram. If there are impurity peaks in the chromatogram of the test solution, the peak area of impurity B shall not be more than 2 times (1.0%) of the main peak area of the control solution, and the impurity R, impurity Q, impurity J, impurity I, impurity S, impurity A, and impurity H are calculated as the corrected peak area (multiplied by the correction factor of 0.5, 0.4, 0.7, 1.6, 0.4, 1.4, 0.1, respectively) all shall not be greater than the main peak area of the control solution (0.5% ) , and the peak area of other individual impurities shall not be greater than the main peak area of the control solution (0.5% ) , the sum of the peak areas of each impurity shall not be greater than 4 times (2.0%) of the main peak area of the control solution (for injection) based on the corrected peak area. If there are impurity peaks in the chromatogram of the test solution, the peak area of impurity B shall not be more than 4 times (2.0%) of the main peak area of the control solution, and the impurity R, impurity Q, impurity J, impurity I, impurity S, impurity A, and impurity H are calculated as the corrected peak area (multiplied by the correction factor of 0.5, 0.4, 0.7, 1.6, 0.4, 1.4, 0.1, respectively) all shall not be greater than 2 times (1.0%) the main peak area of the control solution, and the peak area of other individual impurities shall not be greater than 2 times (1.0%) the main peak area of the control solution, the sum of the peak areas of each impurity shall not be greater than 8 times (4.0%) the area of the main peak of the control solution (for oral use) based on the corrected peak area. The peaks in the chromatogram of the test solution which are smaller than the main peak area of the sensitivity solution are ignored.
moisture
take this product, according to the determination of moisture (General 0832 first method 1), the water content shall not exceed 5.0%.
ignition residue
take l.Og of this product and determine it according to law (General rule 0841). The remaining residue shall not exceed 0.2%.
Heavy metals
The residue left in the item of burning residues shall be inspected according to law (General Principles 0821, Law 2) and shall not contain more than 10 ppm of heavy metals (for injection), not more than 25 parts per million (for oral use).
Sumamed - Content determination
Authoritative Data Verified Data
measured by high performance liquid chromatography (General 0512).
chromatographic conditions and system suitability test
silica gel bonded with eighteen alkyl silane as filler; Phosphate buffer (0.05mol/L dipotassium hydrogen phosphate solution, adjusted to pH 20% with 8.2 phosphoric acid solution)-acetonitrile (45:55) mobile phase; The detection wavelength was 210mn. Take the appropriate amount of azithromycin system applicable reference substance, add acetonitrile to dissolve and dilute the solution containing 10 mg per 1 ml, take 50u1 injection liquid chromatograph, and record the chromatogram should be consistent with the standard spectrum.
assay
take an appropriate amount of this product, weigh it accurately, add acetonitrile to dissolve and quantitatively dilute to prepare a solution containing about 1 mg per 1 ml as a test solution, A 50ul injection liquid chromatograph was used to record the chromatogram. An appropriate amount of azithromycin reference substance was taken and determined by the same method. According to the external standard method to calculate the peak area, that is.
Sumamed - Category
Authoritative Data Verified Data
macrolide antibiotics.
Sumamed - Storage
Authoritative Data Verified Data
sealed and stored in a cool and dry place.
Sumamed - Azithromycin for suspension
Authoritative Data Verified Data
This product contains azithromycin (C38H72N2012) should be labeled the amount of 90.0% to 110.0%.
trait
This product is granular or powder; Aromatic.
identification
take an appropriate amount of the fine powder of this product, and add ethanol to make a solution containing 5mg of azithromycin per lml, filter, and take the filtrate as the test solution; According to the identification under azithromycin (1) or (2) trials showing the same results.
examination
- alkalinity: take an appropriate amount of this product, add methanol (2.5ml per 10mg of azithromycin and toluene) to dissolve it, add water to make a solution containing 2mg of azithromycin per 1 ml, and shake well, 10 minutes after the determination according to law (General 0631), the pH value should be 9.0~11.0.
- Related substances take appropriate amount of fine powder of this product, add diluted solution [ammonium dihydrogen phosphate solution (weigh 1.73g of ammonium dihydrogen phosphate, dissolve with water and dilute to 1000ml, adjust pH value to 10.0±0.05 with ammonia test solution)-Methanol-acetonitrile (7:7:6)] azithromycin was dissolved and diluted to prepare a solution containing 10mg of azithromycin per 1 ml, filtered, and the continued filtrate was taken as the test solution; the Peak area of impurity B shall not be more than 4 times (2.0%) of the main peak area of the control solution as determined by the method under azithromycin, impurity H and impurity Q shall be calculated based on the corrected peak area (multiplied by the correction factor of 0.1 and 0.4 respectively) and shall not be greater than 2 times (1.0%) of the main peak area of the control solution, other single impurity peak area shall not be more than 2 times (1.0%) of the main peak area of the control solution, and the sum of each impurity peak area shall not be more than 8 times (4.0%) of the main peak area of the control solution calculated by the corrected peak area..
- take an appropriate amount of water from this product and determine the water content according to the method of determination of water content (General rule 0832, first method 1). The water content shall not exceed 2.0%.
- other than sedimentation volume ratio (single dose packaging), should comply with the relevant provisions under the item of oral suspension (General Principle 0123).
Content determination
take the contents under the item of difference in loading amount, mix evenly, weigh the appropriate amount accurately (about 0.lg equivalent to azithromycin), acetonitrile was added to dissolve and quantitatively dilute to prepare a solution containing about 1 mg of azithromycin per 1 ml, filtered, and the filtrate was taken as a test solution, which was obtained by measuring according to the method under the item of azithromycin.
category
Same as azithromycin.
specification
0.lg
storage
sealed and stored in a dry place.
Sumamed - Azithromycin tablets
Authoritative Data Verified Data
This product contains azithromycin (C38H72N2012) should be labeled the amount of 90.0% to 110.0%.
trait
This product is a white tablet or film-coated tablet, white or white after removing the coating.
identification
take the appropriate amount of fine powder of this product, and make a solution containing 5mg of azithromycin per 1 ml by adding fermentation, filter, and take the filtrate as the test solution. Or (2) trials showing the same results.
examination
- Related substances take appropriate amount of fine powder of this product, add diluted solution [ammonium dihydrogen phosphate solution (weigh 1.73g of ammonium dihydrogen phosphate, add water to dissolve and dilute to 1000ml, adjust pH value to 10.0±0.05 with ammonia test solution)-Methyl-acetonitrile (7:7:6)] azithromycin was dissolved and diluted into a solution containing 10 mg of azithromycin per 1 ml, filtered, and the filtrate was taken as the test solution; the Peak area of impurity B shall not be more than 4 times (2.0%) of the main peak area of the control solution as determined by the method under azithromycin, impurity H and impurity O shall not be more than 2 times (0.1) of the main peak area of the control solution calculated by the corrected peak area (multiplied by the correction factor 0.4 and 1.0% respectively), other single impurity peak area shall not be more than 2 times (1.0%) of the main peak area of the control solution, and the sum of each impurity peak area shall not be more than 8 times (4.0%) of the main peak area of the control solution calculated by the corrected peak area..
- dissolution of this product, according to the dissolution and release determination method (General 0931 second method), with phosphate buffer (pH 6.0)(0.1 mol/L disodium hydrogen phosphate solution 6000ml, add about 40ml of hydrochloric acid, adjust the pH value to 6.0 0.05) ml as the dissolution medium (0.lg and 0.125g specifications dissolution medium is 500ml), the speed is 100 rpm, according to the operation, after 45 minutes, take the appropriate amount of solution, filtration, precise amount of filtrate, quantitative dilution with dissolution medium to prepare a solution containing about 0.2mg of azithromycin per 1 ml, as a test solution; Appropriate amount of azithromycin reference substance, an appropriate amount of ethanol (about 1 ml of ethanol per 2mg) was added to dissolve the solution, and the solution was quantitatively diluted with the dissolution medium to prepare a solution containing about 0.2mg per 1 ml as a reference solution. According to the method under the content determination item, the dissolution amount of each tablet was calculated by the peak area according to the external standard method. The limit is 75% of the labeled amount and shall be in accordance with the provisions.
- others shall be in accordance with the relevant provisions under the item of tablets (General rule 0101).
Content determination
Take 10 tablets of this product, precision weighing, fine grinding, precision weighing appropriate amount (about equivalent to 0.lg of azithromycin), acetonitrile was added to dissolve and quantitatively dilute to prepare a solution containing about 1 mg of azithromycin per 1 ml, filtered, and the filtrate was taken as a test solution, which was obtained by measuring according to the method under the item of azithromycin.
category
Same as azithromycin.
specification
(1)0.lg (2)0.125g (3)0.25g (4)0.5g
storage
sealed and stored in a dry place.
Sumamed - Azithromycin capsules
Authoritative Data Verified Data
This product contains azithromycin (C38H72N2012) should be labeled the amount of 90.0% to 110.0%.
identification
take an appropriate amount of the contents of this product, add ethanol to make a solution containing 5mg of azithromycin per 1 ml, filter, and take the continued filtrate as the test solution. The same results were shown for the identification (1) or (2) tests under azithromycin.
examination
- relevant substances take appropriate amount of contents of this product, add diluted solution [ammonium dihydrogen phosphate solution (weigh 1.73g of ammonium dihydrogen phosphate, add water to dissolve and dilute to 1000ml, adjust pH value to 10.0±0.05 with ammonia test solution)-Methanol-acetonitrile (7:7:6)] azithromycin was dissolved and diluted to prepare a solution containing 10 mg of azithromycin per 1 ml, filtered, and the continued filtrate was taken as the test solution; the Peak area of impurity B shall not be more than 4 times (2.0%) of the main peak area of the control solution as determined by the method under azithromycin, impurity H and impurity Q shall be calculated based on the corrected peak area (multiplied by the correction factor of 0.1 and 0.4 respectively) and shall not be greater than 2 times (1.0%) of the main peak area of the control solution, other single impurity peak area shall not be more than 2 times (1.0%) of the main peak area of the control solution, and the sum of each impurity peak area shall not be more than 8 times (4.0%) of the main peak area of the control solution calculated by the corrected peak area..
- moisture the contents of this product shall not contain more than 0832 of moisture as determined by the method for moisture determination (General rule 6.0%, Method 1).
- dissolution of this product, according to the dissolution and release determination method (General 0931 second method), with phosphate buffer (pH 6.0)(0. 6000ml of 1 mol/L disodium hydrogen phosphate solution, add about 40ml of hydrochloric acid, adjust the pH value to 6.0±0.05)900ml as the dissolution medium (100g standard dissolution medium 500ml), the rotation speed is rpm, operate according to law, after 45 minutes, take the appropriate amount of the solution, filter, and take the filtrate as the test solution; Take the appropriate amount of azithromycin reference, precision weighing, and add the appropriate amount of ethanol (about 1 ml of ethanol per 2mg) the solution was dissolved and quantitatively diluted with a dissolution medium to prepare a solution containing about 0.25mg (0.28g of mg) per 1 ml as a control solution. According to the method under the content determination item, the dissolution amount of each particle was calculated by the peak area according to the external standard method. The limit is 75% of the labeled amount and shall be in accordance with the provisions.
- others should comply with the relevant provisions under the capsule (General 0103).
Content determination
take the contents under the item of difference in loading quantity, mix evenly, weigh accurately, grind finely, weigh accurately and take appropriate amount (about equivalent to azithromycin O.lg), add acetonitrile quantitative dilution to prepare a solution containing 1 mg of azithromycin per 1 ml, filter, take the filtrate as a test solution, according to the method under the item of azithromycin, then get.
category
Same as azithromycin.
specification
(1)0.125g (2) 0.25g
storage
sealed and stored in a cool and dry place.
Sumamed - Azithromycin granules
Authoritative Data Verified Data
This product contains azithromycin (C38H72N2012) should be labeled the amount of 90.0% to 110.0%.
trait
This product is suspended particles.
identification
take an appropriate amount of the fine powder of this product, add ethanol to make a solution containing 5mg of azithromycin per lml, filter, take the filtrate as the test solution, according to the identification under azithromycin (1) or (2) trials showing the same results.
examination
- alkalinity: take an appropriate amount of this product (about 20mg equivalent to azithromycin), add 5ml of methanol to dissolve, then add 5ml of water, mix well, and measure it according to law after 10 minutes (General rule 0631), the pH should be between 8.5 and 11.5.
- Related substances take appropriate amount of fine powder of this product, add diluted solution [ammonium dihydrogen phosphate solution (weigh 1.73g of ammonium dihydrogen phosphate, add water to dissolve and dilute to 1000ml, adjust pH value to 10.0±0.05 with ammonia test solution)-Methanol-acetonitrile (7:7:6)] azithromycin was dissolved and diluted to prepare a solution containing 10 mg of azithromycin per 1 ml, filtered, and the continued filtrate was taken as the test solution; the Peak area of impurity B shall not be more than 4 times (2.0%) of the main peak area of the control solution as determined by the method under azithromycin, impurity H and impurity Q shall be calculated based on the corrected peak area (multiplied by the correction factor of 0.1 and 0.4 respectively) and shall not be greater than 2 times (1.0%) of the main peak area of the control solution, other single impurity peak area shall not be more than 2 times (1.0%) of the main peak area of the control solution, and the sum of each impurity peak area shall not be more than 8 times (4.0%) of the main peak area of the control solution calculated by the corrected peak area..
- the moisture content of this product shall not exceed 0832 as determined by the method for determination of moisture (General rule 2.0%, first method 1).
- others should comply with the relevant provisions under The granule (General Principle 0104).
Content determination
take the contents under the item of difference in loading amount, mix evenly, weigh the appropriate amount accurately (about 0.lg equivalent to azithromycin), acetonitrile was added to dissolve and quantitatively dilute to prepare a solution containing about 1 mg of azithromycin per 1 ml, filtered, and the filtrate was taken as a test solution, which was obtained by measuring according to the method under the item of azithromycin.
category
Same as azithromycin.
specification
(1)0.lg (2)0.125g (3)0.25g (4)0.5g
storage
sealed and stored in a dry place.
Sumamed - Azithromycin for injection
Authoritative Data Verified Data
This product is a sterile preparation made of azithromycin with appropriate amount of citric acid or other suitable cosolvent. Azithromycin (C38H72N2012) should be included in 93.0%-107.0% of label load based on average loading.
trait
This product is white or off-white loose block or powder.
identification
take an appropriate amount of this product, add an appropriate amount of water (50mg of azithromycin and 1 ml of water) to dissolve, and dilute with ethanol to prepare a solution containing about 5mg of azithromycin per 1 ml as a test solution. The same results were shown for the identification (1) or (2) test under azithromycin.
examination
- the pH value of this product is appropriate, and the water is added to make a solution containing about 25mg of azithromycin per lml, which is determined according to law (General rule 0631). The pH value should be 5.5~7.5.
- clarity and color of solution take 5 bottles of this product and add water separately to make azithromycin 0.lg solution, the solution should be clear and colorless; If it is turbid, it should not be more concentrated than the 1# turbidity standard solution (General 0902 first method); If it is colored, no deeper than the yellow No. 1 Standard Colorimetric solution (General rule 0901 first method).
- the related substances may be newly manufactured or low-temperature injector used. After adding an appropriate amount of water (50mg of azithromycin and 1 ml of water) to dissolve the product, dilute the product with ammonium dihydrogen phosphate solution (1.73g), add water to dissolve and dilute, the pH was adjusted to 10.0±0.05 with ammonia test solution)-methanol-acetonitrile (7:7:6)] was quantitatively diluted to prepare a solution containing about 10 mg of azithromycin per 1 ml as a test solution. According to the method under azithromycin, if there are impurity peaks in the chromatogram of the test solution, it should be in accordance with the provisions of azithromycin (for injection).
- the moisture content of this product shall not exceed 0832 as determined by the method for determination of moisture (General rule 2.0%, first method 1).
- bacterial endotoxin this product, according to the law to check (General 1143), each 1 mg of azithromycin containing endotoxin should be less than 0.30EU.
- sterile take this product, dissolve and dilute with 0.9% sterile sodium chloride solution to prepare a solution containing 20mg of azithromycin per 1 ml, and treat it by membrane filtration. The drug loading of azithromycin per membrane is not more than 3.3g, wash with 0.1% sterile peptone aqueous solution (not less than 600ml per membrane), use Staphylococcus aureus as positive control bacteria, check according to law (General rule 1101), should comply with the regulations.
- others should comply with the relevant provisions under injection (General 0102).
Content determination
take the content under the difference of loading amount (about equivalent to 0.lg of azithromycin), weigh precisely, add appropriate amount of water (50mg of azithromycin and 1 ml of water) to dissolve, A solution containing about 1 mg of azithromycin per 1 ml was prepared by quantitative dilution with the diluent under the item of related substances, and as a test solution, it was obtained by measuring according to the method under the item of azithromycin.
category
Same as azithromycin.
specification
calculated as C38H72N2012 (1)0.lg (2)0.125g(3)0.25g (4)0.5g
storage
sealed and stored in a cool place.
Supplier List
CAS: 83905-01-5
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
CAS: 83905-01-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 83905-01-5
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
CAS: 83905-01-5
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 83905-01-5
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 83905-01-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 83905-01-5
Tel: 18888297294
Email: kenny@hychemchina.com
Mobile: 18888297294
QQ: 3440845085
CAS: 83905-01-5
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
CAS: 83905-01-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 83905-01-5
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
CAS: 83905-01-5
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 83905-01-5
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 83905-01-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 83905-01-5
Tel: 18888297294
Email: kenny@hychemchina.com
Mobile: 18888297294
QQ: 3440845085





