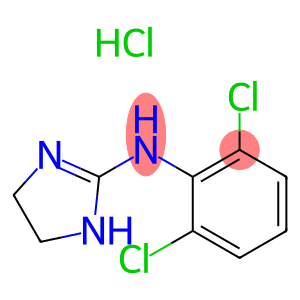
N-(2,6-二氯苯基)-4,5-二氢-1H-咪唑基-2-胺盐酸盐
clonidine hydrochloride
CAS: 4205-91-8
Molecular Formula: C9H10Cl3N3
N-(2,6-二氯苯基)-4,5-二氢-1H-咪唑基-2-胺盐酸盐 - Names and Identifiers
| Name | clonidine hydrochloride |
| Synonyms | DIXARIT katapresan Clonidine Hcl CLONIDINE HCL CLONIDINE HYDROCHLORIDE clonidine hydrochloride 2-(2,6-DICHLOROANILINO)-2-IMIDAZOLINE, HCL 2-(2,6-DICHLOROANILINO)-2-IMIDAZOLINE HYDROCHLORIDE 2-[2,6-DICHLOROANILINE]-2-IMIDAZOLINE HYDROCHLORIDE 2-(2,6-Dichloroanilino)-2-imidazoline hydrochloride 2-(2,6-DICHLOROPHENYLAMINO)-2-IMIDAZOLINE HYDROCHLORIDE |
| CAS | 4205-91-8 |
| EINECS | 224-121-5 |
| InChI | InChI=1/C9H9Cl2N3.ClH/c10-6-2-1-3-7(11)8(6)14-9-12-4-5-13-9;/h1-4,9,13-14H,5H2;1H |
| InChIKey | GLEWMLFXCSBZLK-UHFFFAOYSA-N |
N-(2,6-二氯苯基)-4,5-二氢-1H-咪唑基-2-胺盐酸盐 - Physico-chemical Properties
| Molecular Formula | C9H10Cl3N3 |
| Molar Mass | 266.55 |
| Melting Point | 312 °C |
| Boling Point | 319.3ºC at760mmHg |
| Water Solubility | Soluble in water (50 mg/ml), DMSO (75 mM), methanol, chloroform (slightly), and dehydrated alcohol. |
| Solubility | Soluble in water, ethanol, almost insoluble in chloroform, ether |
| Appearance | White crystalline powder |
| Color | white |
| Merck | 14,2390 |
| BRN | 4163525 |
| PH | pH(50g/l, 25℃) : 3.5~6.0 |
| Storage Condition | 2-8°C |
| MDL | MFCD00036705 |
| Use | Used as antihypertensive and smoking cessation drug |
N-(2,6-二氯苯基)-4,5-二氢-1H-咪唑基-2-胺盐酸盐 - Risk and Safety
| Hazard Symbols | T+ - Very toxic |
| Risk Codes | R25 - Toxic if swallowed R26 - Very Toxic by inhalation |
| Safety Description | S22 - Do not breathe dust. S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S28 - After contact with skin, wash immediately with plenty of soap-suds. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) |
| UN IDs | UN 2811 6.1/PG 1 |
| WGK Germany | 3 |
| RTECS | NJ2490000 |
| FLUKA BRAND F CODES | 10 |
| HS Code | 2933290000 |
| Hazard Class | 6.1(b) |
| Packing Group | III |
| Toxicity | LD50 in mice, rats (mg/kg): 328, 270 orally; 18, 29 i.v. (Walland) |
N-(2,6-二氯苯基)-4,5-二氢-1H-咪唑基-2-胺盐酸盐 - Standard
Authoritative Data Verified Data
This product is 2-[(2, 6-dichlorophenyl) imino] imidazolidine hydrochloride. The content of C9H9C12N3 • HC1 shall not be less than 99.0% based on dry product.
N-(2,6-二氯苯基)-4,5-二氢-1H-咪唑基-2-胺盐酸盐 - Trait
Authoritative Data Verified Data
- This product is white crystalline powder; Odorless.
- This product is soluble in water or ethanol, slightly soluble in three gas methane, and almost insoluble in ether.
N-(2,6-二氯苯基)-4,5-二氢-1H-咪唑基-2-胺盐酸盐 - Differential diagnosis
Authoritative Data Verified Data
- take about 1 mg of this product, add 2ml of water to dissolve, add 1ml of new 5% sodium nitrosoferricyanide solution, 2ml of sodium hydroxide solution and lg of sodium bicarbonate, and change to purple after shaking, after placement, the color was darker.
- take this product, add 0.01mol/L hydrochloric acid solution to make a solution containing about 0401 mg per ml, and measure it by UV-visible spectrophotometry (general rule), there is a maximum absorption at 272nm and 279nm wavelength, the absorbance is about 0.55 and 0.47 respectively.
- The infrared absorption spectrum of this product should be consistent with that of the control (Spectrum set 327).
- the aqueous solution of this product can identify the reaction of (1) (General rule 0301).
N-(2,6-二氯苯基)-4,5-二氢-1H-咪唑基-2-胺盐酸盐 - Exam
Authoritative Data Verified Data
acidity
take 0.10g of this product, Add 10ml of water, and measure it according to law (General rule 0631). The pH value should be 4.0~5.0.
clarity of the solution
take 0.10g of this product, Add 10ml of water to dissolve, and the solution should be clear.
Related substances
take this product, add methanol to dissolve and dilute to make a solution containing about 10 mg per lml, as a test solution; Take appropriate amount of precision, as a control solution, a solution containing about 50ug per 1 ml was prepared by dilution with methanol. According to the thin layer chromatography (General 0502) test, absorb the above two solutions each 3 u1, respectively, on the same silica gel G thin layer plate, with n-butanol-water-glacial acetic acid [(4:5:1), shake, after static stratification, take the upper layer of liquid filtration] as the developer, expand, dry, spray with dilute bismuth potassium iodide test solution, immediately gently spray 5% sodium nitrite solution. Test solution such as impurity spots, compared with the control solution of the main spot, not deeper.
loss on drying
take this product, dry to constant weight at 105°C, weight loss shall not exceed 0.5% (General rule 0831).
Heavy metals
take this product l. Add 2ml of acetate buffer solution (pH3.5) and appropriate amount of water to dissolve 25ml, and check according to law (General rule 0821 method 1), containing no more than 20 parts per million of heavy metals.
N-(2,6-二氯苯基)-4,5-二氢-1H-咪唑基-2-胺盐酸盐 - Content determination
Authoritative Data Verified Data
take this product about 0.15g, precision weighing, add glacial acetic acid 10ml and Mercury acetate test solution 3ml, warm to dissolve, cool, add crystal violet indicator solution 1 drop, with perchloric acid titration solution (0.1 mol/L) titration to the solution is blue-green, and the result of the titration is corrected with a blank test. Each 1 ml of perchloric acid titration solution (0.1 mol/L) corresponds to 26.66mg of C9H9Cl2N3. Hc1.
N-(2,6-二氯苯基)-4,5-二氢-1H-咪唑基-2-胺盐酸盐 - Category
Authoritative Data Verified Data
antihypertensive drugs.
N-(2,6-二氯苯基)-4,5-二氢-1H-咪唑基-2-胺盐酸盐 - Storage
Authoritative Data Verified Data
light shielding, sealed storage.
N-(2,6-二氯苯基)-4,5-二氢-1H-咪唑基-2-胺盐酸盐 - Clonidine hydrochloride tablets
Authoritative Data Verified Data
This product contains clonidine hydrochloride (C9H9C12N3 • HCI) should be 90.0% to 110.0% of the label.
trait
This product is white tablet.
identification
- take an appropriate amount of fine powder of this product (about 0.25mg of clonidine hydrochloride), add 5ml of water to dissolve clonidine hydrochloride, add 1ml of 30% sodium hydroxide solution, shake, and add 5ml of diethyl ether, after shaking and separating the diethyl ether solution, one drop of diethyl ether solution was taken after the solution was dispersed to about 0.2, and then the solution was stained with a newly prepared basic sodium nitrosoferricyanide solution (g of sodium nitrosoferricyanide), add 4ml of water and 1ml of sodium hydroxide solution, mix well) on the dry filter paper, and then add 1 drop of 8% sodium bicarbonate solution to generate purple spots.
- in the chromatogram recorded under the content determination item, the retention time of the main peak of the test solution should be consistent with the retention time of the main peak of the reference solution.
- This product water solution gasification product identification (1) of the reaction (General 0301).
examination
- Content uniformity take 1 tablet of this product, put it in 25ml measuring flask, add appropriate amount of mobile phase, fully shake for 30 minutes to dissolve clonidine hydrochloride, dilute it to the scale with mobile phase, shake well, filtration, take the filtrate, according to the content determination method under the item, should comply with the provisions (General 0941).
- others shall be in accordance with the relevant provisions under the item of tablets (General rule 0101).
Content determination
measured by high performance liquid chromatography (General 0512).
Chromatographic conditions and system suitability test using octane bonded silica gel as filler, 0.22% sodium octane Sulfonate-methanol-phosphoric acid [(500:500:1 ), the mobile phase was adjusted to pH 3.0 with 1 mol/L sodium hydroxide solution or phosphoric acid, and the detection wavelength was 220nm. The number of theoretical plates shall not be less than 3500 based on clonidine peak, and the tailing factor shall not exceed 1.5.
Determination of 20 tablets of this product, precision weighing, fine, precision weighing an appropriate amount (equivalent to clonidine hydrochloride 0.15mg ), placed in a 50ml measuring flask, plus the appropriate amount of mobile phase, fully shake for 30 minutes to dissolve clonidine hydrochloride, add the mobile phase to dilute to the scale, filter filter, take the filtrate as the test solution, take the precision amount of 5 u1, and inject the human liquid chromatograph, record the chromatogram; Take an appropriate amount of clonidine hydrochloride reference substance, precisely weigh, add mobile phase to dissolve and quantitatively dilute to prepare a solution containing about 3ug per 1 ml, and determine with the same method. According to the external standard method to calculate the peak area, that is.
category
Same as clonidine hydrochloride.
specification
(l)75ug (2)0.lmg
storage
light shielding, sealed storage.
N-(2,6-二氯苯基)-4,5-二氢-1H-咪唑基-2-胺盐酸盐 - Clonidine hydrochloride injection
Authoritative Data Verified Data
This product is a sterile aqueous solution of clonidine hydrochloride. Clonidine hydrochloride containing CC9H9C12N3 • HCl) shall be between 90.0% and 110.0% of the labeled amount.
trait
This product is a clear colorless liquid.
identification
- (1) 6ml of the product was taken, and evaporated to about 2ml on a water bath. The same reaction was shown according to the identification (1) Test under the item of clonidine hydrochloride.
- (2) take 5ml of this product, add 1 drop of lmol/ L hydrochloric acid solution, shake well, and measure by ultraviolet-visible spectrophotometry (General rule 0401), there is a maximum absorption at the wavelength of 272mn and 279mn.
examination
- the pH value should be 4.0 to 6.0 (General 0631).
- others should comply with the relevant provisions under injection (General 0102).
Content determination
- measured by high performance liquid chromatography (General 0512).
- chromatographic conditions and system suitability test using octane bonded silica gel as filler, 0.22% sodium octane Sulfonate-methanol-phosphoric acid [(500:500:1 ), the mobile phase was adjusted to pH 3.0 with 1 mol/ L sodium hydroxide solution or phosphoric acid, and the detection wavelength was 220nm. The number of theoretical plates shall not be less than 3500 based on clonidine peak, and the tailing factor shall not exceed 1.5.
- measure 2ml of the product, put it in a 100ml measuring flask, dilute to the scale with mobile phase, shake well, take 50ul of the product, inject it into the liquid chromatograph, record the chromatogram; in addition, the appropriate amount of clonidine hydrochloride reference substance was accurately weighed, dissolved and quantitatively diluted with mobile phase to prepare a solution containing about 3ug per lml, and the same method was used for determination. According to the external standard method to calculate the peak area, that is.
category
Same as clonidine hydrochloride.
specification
lml:0.15mg
storage
light shielding, closed storage.
N-(2,6-二氯苯基)-4,5-二氢-1H-咪唑基-2-胺盐酸盐 - Clonidine hydrochloride eye drops
Authoritative Data Verified Data
This product contains clonidine hydrochloride (C9H9C12N3 • HCl) should be 90.0% to 110.0% of the label.
trait
This product is a clear colorless liquid.
identification
- take 0.5ml of this product, add 2ml of water, shake well, according to the item (1) identification test under the item of clonidine hydrochloride, show the same reaction.
- take the solution of this product and add 0.01mol /L hydrochloric acid solution to make a solution containing 0.3mg per lml, and measure it according to ultraviolet-visible spectrophotometry (Tongcai 0401), there is a maximum absorption at the wavelength of 272nm and 279nm.
examination
- the pH value should be 5.0 to 7.0 (General 0631).
- others shall comply with the relevant provisions under Ophthalmic Preparations (General rule 0105).
Content determination
- measured by high performance liquid chromatography (General 0512).
- chromatographic conditions and system suitability test using octane bonded silica gel as filler, 0.22% sodium octane Sulfonate-methanol-phosphoric acid [(500:500:1 ), the mobile phase was adjusted to pH 3.0 with 1 mol/ L sodium hydroxide solution or phosphoric acid, and the detection wavelength was 220nm. The number of theoretical plates shall not be less than 3500 based on clonidine peak, and the tailing factor shall not exceed 1.5.
- precision measurement this product is quantitatively diluted with mobile phase to make a solution containing clonidine hydrochloride 2.5ug per lml, and 50 u1 is accurately measured and injected into human liquid chromatograph to record the chromatogram; in addition, the appropriate amount of clonidine hydrochloride reference substance was accurately weighed, dissolved and quantitatively diluted with mobile phase to prepare a solution containing about 2.5ug per 1 ml, which was determined by the same method. According to the external standard method to calculate the peak area, that is.
category
ophthalmic medication.
specification
5ml:12.5mg
storage
light shielding, closed storage.
N-(2,6-二氯苯基)-4,5-二氢-1H-咪唑基-2-胺盐酸盐 - Reference Information
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| background | in September 2010, FDA approved clonidine hydrochloride 0.1mg and 0.2mg sustained-release tablets (Kapvay,Shionogi Inc) alone or in combination with stimulants for the treatment of children with attention deficit hyperactivity disorder (ADHD) aged 6 to 17 years old. This product is the first clonidine preparation approved by the US FDA for the treatment of ADHD, and is the only adjuvant therapeutic drug for stimulant treatment of ADHD. |
| use | marked clonidine. α2-adrenergic agonist. Antihypertensive; analgesic for neuropathic pain. Used as an antihypertensive and smoking cessation drug |
| application | clonidine hydrochloride is also suitable for the treatment of hypertension. it can be used alone or in combination with other antihypertensive drugs. At present, there are also clonidine transdermal patches for the treatment of children's vocal and multiple movements combined with tic disorders. Clonidine can also be used for combined anesthesia during the operation period to strengthen the intensity and time of narcotic drugs and reduce the adverse effects of narcotic drugs. Reaction. |
| adverse reactions | most adverse reactions are mild and relieved with the medication process. Common: most common dry mouth (dose-related), lethargy, dizziness, mental depression, constipation and sedation, decreased sexual function and nocturia, itching, nausea, vomiting, insomnia, urticaria, angioedema and rubella, fatigue, orthostatic symptoms, tension and anxiety, hair loss, rash, anorexia and general malaise, weight gain, headache, fatigue, withdrawal syndrome, transient abnormal liver function. |
| preparation | a. synthesis of intermediate 1 188ml of formic acid and 10.0g 2,6-dichloroaniline are added to the reaction bottle, heated and stirred in an oil bath, stirred and refluxed at 95-100 ℃ for 5h, then cooled to room temperature, then cooled to 0 ℃, stirred and crystallized for 2h, pumped and dried to obtain 92.0g of white solid with a yield of 78.4%. B. synthesis of intermediate 2 70ml thionyl chloride and 92g of intermediate 1 are added to the reaction flask, stirred, cooled to 0 ℃ in an ice water bath, and 141ml of chlorine sulfoxide and 156ml of sulfonyl chloride are added. After adding, the temperature is naturally raised to room temperature, then heated to 40 ℃ and stirred for 20h, the system is dissolved and distilled to no fraction under reduced pressure, 300ml of dichloromethane is added, the temperature is lowered to 5 ℃ in ice water bath, 131g of dichloromethane solution of ethylene amine is added dropwise, after dropping, the temperature is naturally raised to room temperature, the reaction is stopped for 2 hours, the reaction is completed, and 200ml water is added to the system, stir for 30min, let stand for stratification, extract 100ml * 2 with dichloromethane, discard the aqueous phase, merge the organic phase, wash with water for 3 times, 10% sodium hydroxide for 2 times, saturated salt water for 1 time, collect dichloromethane layer and concentrate to obtain intermediate 2. Intermediate 2 300ml of anhydrous ethanol is added, activated carbon is used for 767 needle, stirred and heated to reflux, filtered while hot, 20ml of hydrogen chloride-ethanol dissolved solution is added dropwise to the filtrate, after dropping, kept warm and stirred for 30min, naturally cooled to room temperature, then cooled to 0 ℃, stirred and crystallized for 3h, filtered, leached with less ethanol, and dried to obtain clonidine hydrochloride, clonidine hydrochloride is dissolved in 300mL of water, stirred at room temperature for 1h, filtered to remove insoluble matter, cooled to below 10 ℃ in ice water bath, adjusted pH to 10 with 25% sodium hydroxide to generate white precipitate, filtered, and rinsed the filter cake with a small amount of water to obtain 60.3g of white-like solid. C. synthesis of clonidine hydrochloride 250ml ethanol and 63.3g intermediate 2 are added to a 500ml reaction bottle, stirred and heated until reflux. Remove the insoluble substance while hot, add 29.2g of hydrogen chloride-ethanol solution dropwise to the filtrate, after dropping, reflux stirring for 30min, then naturally cool to room temperature, then cool to 0 ℃-5 ℃, stir for crystallization for 3h, filter, and rinse the filter cake with a small amount of ethanol to obtain clonidine hydrochloride crude product. Add the solid body wet product obtained in the previous step into a 1 liter reaction bottle, add 250ml of ethyl acetate, and stir at room temperature for 1h. Then filter, the filter cake is rinsed with a small amount of ethyl acetate ester and dried to obtain 48.9g of white solid with 90.01% yield. |
| Production method | is obtained by formylation, chlorination and cyclization of 2, 6-dichloroaniline. Stir the obtained clonidine together with anhydrous ethanol and activated carbon, heat and reflux, filter out activated carbon, add the filtrate to the hydrogen chloride ethanol solution to form salt, cool and crystallize, filter, wash and crystallize with anhydrous ethanol, and dry to obtain clonidine hydrochloride. |
| category | toxic substances |
| toxicity classification | highly toxic |
| acute toxicity | oral-rat LD50: 126 mg/kg; Oral-mouse LD50: 139 mg/kg |
| flammability hazard characteristics | combustible; combustion produces toxic chloride and nitrogen oxide smoke |
| storage and transportation characteristics | warehouse ventilation and low temperature drying; Store separately from oxidants, alkalis and food additives |
| fire extinguishing agent | foam, water, carbon dioxide, sand |
Supplier List
CAS: 4205-91-8
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
CAS: 4205-91-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 4205-91-8
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 4205-91-8
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 4205-91-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 4205-91-8
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
CAS: 4205-91-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 4205-91-8
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 4205-91-8
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 4205-91-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025




