Ironsulfatehydratetechgroffwhitepowder
Iron(III) sulfate hydrate
CAS: 15244-10-7
Molecular Formula: Fe2(SO4)3 · xH2O
Ironsulfatehydratetechgroffwhitepowder - Names and Identifiers
| Name | Iron(III) sulfate hydrate |
| Synonyms | polymeric ferric sulfate Iron(III) sulfate hydrate Ferric sulfate test solution(ChP) Ironsulfatehydratetechgroffwhitepowder |
| CAS | 15244-10-7 |
| EINECS | 604-849-3 |
Ironsulfatehydratetechgroffwhitepowder - Physico-chemical Properties
| Molecular Formula | Fe2(SO4)3 · xH2O |
| Molar Mass | 399.88 (anhydrous ba |
| Density | 3,097 g/cm3 |
| Melting Point | 480°C(decomposition) |
| Boling Point | 330℃ at 760 mmHg |
| Water Solubility | Soluble in waterSoluble in water. Sparingly soluble in alcohol. |
| Appearance | Light gray-white powder or diamond crystal |
| Storage Condition | Room Temprature |
| Sensitive | Easily absorbing moisture |
| MDL | MFCD00011007 |
| Use | Iron sulfate, also known as high iron sulfate, is light yellow or white powder. Soluble in water and alcohol. The aqueous solution is acidic, and the basic salt can be formed when the dilute solution is heated to boiling. Sulfates with alkali metals or ammonium tend to form iron vanadium in the form of MFe(SO4)2 · 12H2O, such as ammonium ferric sulfate NH4Fe(SO4)2 · 12H2O, also known as Iron ammonium alum. Ferric sulfate has various hydrates containing 3, chemical book6,7,9,10,12 water, the most common being Fe2(SO4)3 · 9H2O, yellow crystals with a relative density of 2.1. After the hydrate is heated, anhydrous salt can be obtained. The hydrate can be prepared by oxidizing the ferrous salt with nitric acid or hydrogen peroxide in sulfuric acid solution. Purposes: convenient, efficient catalyst for the preparation of aromatic esters from the corresponding acid and alcohol. |
Ironsulfatehydratetechgroffwhitepowder - Risk and Safety
| Hazard Symbols | Xn - Harmful |
| Risk Codes | R22 - Harmful if swallowed R36/37 - Irritating to eyes and respiratory system. R41 - Risk of serious damage to eyes R38 - Irritating to the skin |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S39 - Wear eye / face protection. |
| UN IDs | UN 3077 |
Ironsulfatehydratetechgroffwhitepowder - Synthesis of isoamyl benzoate catalyzed by ferric sulfate hydrate
from hownet
Author:
Abstract:
synthesis of isoamyl benzoate with ferric sulfate hydrate as Catalyst, the results showed that the catalytic activity of the catalyst was higher, and the optimum process conditions for the synthesis of isoamyl benzoate were determined: the ratio of alcohol to acid was 3:1, the yield of Ester was 3.0 when the amount of catalyst was 4%, reaction time was 140 h, reaction temperature was 145 ~ 92.7% ℃.
Key words:
ferric sulfate hydrate catalysis synthesis isoamyl benzoate
DOI:
10.3969/j.issn.1007-2853.2000.02.005
cited:
year:
2000
Ironsulfatehydratetechgroffwhitepowder - Synthesis of butyrate catalyzed by solid ferric sulfate hydrate
from hownet
Author:
Abstract:
In this paper, the synthesis of butyric acid ester catalyzed by solid ferric sulfate hydrate is reported. The catalyst is characterized by easy operation, short reaction time, simple operation, no pollution and high yield of Ester, catalyst performance is stable, can be reused and other characteristics.
Key words:
butyrate ferric sulfate hydrate esterification catalyst solid ferric sulfate
DOI:
CNKI:SUN:LNHG.0.1995-03-006
cited:
year:
1995
Ironsulfatehydratetechgroffwhitepowder - Synthesis of acetate catalyzed by ferric sulfate hydrate
from VIP Journal Professional Edition
Author:
Abstract:
The results of the study on the synthesis of acetate with ferric sulfate hydrate as catalyst were reported, and the optimum conditions of the esterification reaction were discussed.
Key words:
ferric sulfate hydrate; Acetate; Esterification
DOI:
CNKI:SUN:LNHG.0.1997-05-017
cited:
year:
1997
Ironsulfatehydratetechgroffwhitepowder - Solvent effect of dehydration of alcohols to olefins catalyzed by ferric sulfate hydrate
from VIP Journal Professional Edition
Author:
Abstract:
Under the catalysis of ferric sulfate hydrate, the solvent effect, activation energy and activation entropy of 2-methyl-2-hexanol dehydration to olefin in different solvents were determined; the isokinetic constant β is determined to be equal to 395.8K. More reduction
Key words:
dehydration solvent effect ferric sulfate hydrate
DOI:
CNKI:SUN:JLDX.0.1989-01-019
cited:
year:
1989
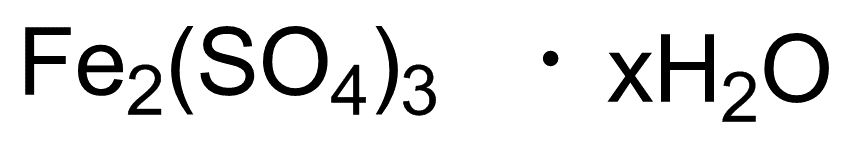
Supplier List
CAS: 15244-10-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 15244-10-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 15244-10-7
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 15244-10-7
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 15244-10-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 15244-10-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 15244-10-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 15244-10-7
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 15244-10-7
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 15244-10-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025




