Exemestane
Exemestane
CAS: 107868-30-4
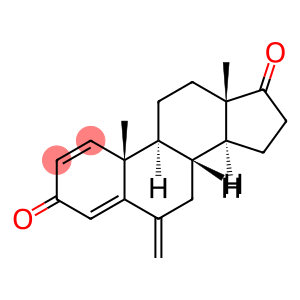
Molecular Formula: C20H24O2
Exemestane - Names and Identifiers
| Name | Exemestane |
| Synonyms | PNU155971 Exemestan ExeMestine Exemestone Exemestane ExeMestane (USP) Exemestane(Aromasin) ExeMestane(AroMasin) 6-Methylenandrosta-1,4-diene-3,17-dione 1,4-Androstadien-3,17-dione-6-Methylene-d2 6-methylideneandrosta-1,4-diene-3,17-dione (8ξ,9ξ,14ξ)-6-methylideneandrosta-1,4-diene-3,17-dione ExeMestane SynonyMs 6-Methylenandrosta-1,4-diene-3,17-dione 10,13-Dimethyl-6-methylidene-7,8,9,10,11,12,13,14,15,16-decahydrocyclopenta[a]phenanthrene- (8R,9S,10R,13S,14S)-10,13-Dimethyl-6-methylene-7,8,9,11,12,13,15,16-octahydro-6H-cyclopenta[a]phenanthrene-3,17(10H,14H)-dione (8R,9S,10R,13S,14S)-10,13-diMethyl-6-Methylene-7,8,9,10,11,12,13,14,15,16-decahydro-3H-cyclopenta[a]phenanthrene-3,17(6H)-dione |
| CAS | 107868-30-4 |
| EINECS | 643-090-2 |
| InChI | InChI=1/C20H24O2/c1-12-10-14-15-4-5-18(22)20(15,3)9-7-16(14)19(2)8-6-13(21)11-17(12)19/h6,8,11,14-16H,1,4-5,7,9-10H2,2-3H3/t14?,15?,16?,19-,20+/m1/s1 |
| InChIKey | BFYIZQONLCFLEV-DAELLWKTSA-N |
Exemestane - Physico-chemical Properties
| Molecular Formula | C20H24O2 |
| Molar Mass | 296.4 |
| Density | 1.13±0.1 g/cm3(Predicted) |
| Melting Point | 155.13°C |
| Boling Point | 453.7±45.0 °C(Predicted) |
| Flash Point | 169°C |
| Solubility | DMSO: ≥20mg/mL |
| Vapor Presure | 2.02E-08mmHg at 25°C |
| Appearance | powder |
| Color | white to off-white |
| Storage Condition | 2-8°C |
| Refractive Index | 1.572 |
| Physical and Chemical Properties | Melting Point: 155.13°C |
Exemestane - Risk and Safety
| Risk Codes | R60 - May impair fertility R61 - May cause harm to the unborn child R51 - Toxic to aquatic organisms |
| Safety Description | S53 - Avoid exposure - obtain special instructions before use. S22 - Do not breathe dust. S36/37 - Wear suitable protective clothing and gloves. S57 - Use appropriate container to avoid environmental contamination. |
| WGK Germany | 3 |
| HS Code | 29372900 |
Exemestane - Nature
Open Data Verified Data
white solid. mp 188~191 deg C; UV maximum absorption of 247nm(& epsilon;13750). Soluble in DMF, soluble in methanol, almost insoluble in water.
Exemestane - Preparation Method
Open Data Verified Data
androstin-4-ene-3, 17-= ketone and triethyl orthoformate are dissolved in tetrahydrofuran and anhydrous ethanol and reacted in the presence of p-toluenesulfonic acid, N-methylaniline and formaldehyde solution were then added and the reaction was continued. The resulting reaction product was dissolved in benzoic acid and anhydrous dioxane, dichlorodicyanobenzoquinone (DDQ) to give exemestane.
Exemestane - Standard
Authoritative Data Verified Data
This product is 6-methylidene-1, 4-dien-3, 17-Dione. The content of C20H2402 shall be 98.0% ~ 102.0% calculated as dry product.
Exemestane - Trait
Authoritative Data Verified Data
This product is white or white crystalline powder; Odorless.
This product is soluble in three gas methane, dissolved in ethyl acetate, acetone, methanol or ethyl alcohol, almost insoluble in water.
melting point
The melting point of this product (General 0612) is 192~196°C.
specific rotation
take about 0.5g of this product, accurately weigh it, put it in a 50ml measuring flask, add ethanol to dissolve and dilute to the scale, shake well, and determine it according to law (General rule 0621), the specific rotation was 88 ° to 298 °.
absorption coefficient
take about 25mg of this product, precision weighing, put it in 50ml measuring flask, add ethanol to dissolve and dilute to the scale, shake, take 2ml, put it in 100ml measuring flask, the absorbance was measured at a wavelength of 0401 Mn by ultraviolet-visible spectrophotometry (General 475) after being diluted to a scale with ethanol, and the absorption coefficient was 495.
Exemestane - Use
Open Data Verified Data
developed by Pharmacia & Upj ohn. It was approved by FDA in October 1997. Antineoplastic agents, second-generation aromatase inhibitors. Exemestane is the first oral hormonal agent that selectively targets and permanently binds to aromatase, an enzyme that can never remanufacture estrogens once binding of exemestane to the enzyme occurs. Can inhibit the supply of estrogen to cancer cells and help prevent cells from continuing to grow. For the treatment of metastatic breast cancer and as an adjuvant therapy for early breast cancer.
Exemestane - Differential diagnosis
Authoritative Data Verified Data
- take about 5mg of this product, add 3ml sulfuric acid to dissolve, place for 5 minutes, the solution gradient is orange-red, pour into lml water, the solution becomes brown-red, and there is flocculent precipitation.
- in the chromatogram recorded under the content determination item, the retention time of the main peak of the test solution should be consistent with the retention time of the main peak of the reference solution.
- take this product and dissolve it with ethanol to make a solution containing 10ug per 1 ml. According to ultraviolet-visible spectrophotometry (General 0401), there is a maximum absorption at the wavelength of 246mn.
- The infrared absorption spectrum of this product should be consistent with that of the control (Spectrum set 1156).
Exemestane - Exam
Authoritative Data Verified Data
clarity and color of ethanol solution
take 0.10g of this product and add 10ml of ethanol to dissolve. The solution should be clear and colorless.
Related substances
take an appropriate amount of this product, such as the mobile phase is dissolved and diluted to make a solution containing about 0.5mg per lml, as a test solution; Take 1ml for precision measurement, and put it in a 200ml measuring flask, dilute to the scale with mobile phase, shake, as a control solution, according to the chromatographic conditions under the content determination item, take 20 u1 of the test solution and the control solution, human liquid chromatograph was injected respectively, and the chromatogram was recorded to 2.5 times of the retention time of the main component peak. If there are impurity peaks in the chromatogram of the test solution, the area of a single impurity peak shall not be greater than the area of the main peak of the control solution (0.5% ) , the sum of each impurity peak area shall not be greater than 2 times (1.0%) of the main peak area of the control solution.
residual solvent
take about 0.2g of this product, weigh it accurately, place it in the top empty bottle, and add the internal standard solution accurately (take the appropriate amount of n-butanol, add N-methylpyrrolidone to dissolve and dilute the solution containing 0.5mg per 1 ml) 2ml, shake to dissolve, seal, as a test solution; Take toluene, cyclohexane, tetrahydrofuran, dioxane, dichloromethane, trichloromethane and methanol in appropriate amounts, precision weighing, plus the above internal standard solution dissolved and quantitatively diluted to make each 1 ml containing about 0.089mg, 0.388mg, 0.072mg, 0.038mg, 0.06mg, 0.006mg and 0.3mg of mixed solution, the precision of 2ml, in the top empty bottle, sealed, as a reference solution. Determined according to the residual solvent assay (General 0861 second method). The capillary column with polyethylene glycol (or similar polar) as the stationary liquid is used as the chromatographic column; The initial temperature is 30°C, maintained for 30 minutes, and the temperature is raised to 300°C at a rate of 20 * C per minute for 7 minutes; the inlet temperature was 200°C; The detector temperature was 220°C; The headspace bottle equilibrium temperature was 80°C and the equilibrium time was 20 minutes. Take the reference solution into the headspace, the separation degree between the peaks of each component shall meet the requirements. The test solution and the reference solution were respectively injected in the headspace, and the chromatogram was recorded, and the peak area was calculated according to the internal standard method. Toluene, cyclohexane, Tetrahydrofuran, dioxane, dichloromethane, chloroform and methanol residues shall be in accordance with the provisions.
loss on drying
take this product, dry to constant weight at 105°C, weight loss shall not exceed 0.5% (General rule 0831).
ignition residue
take l.Og of this product and check it according to law (General rule 0841). The residue left shall not exceed 0.1%.
Heavy metals
The residue left under the item of burning residual Tong shall be inspected according to law (General rule 0821, Law 2), and the content of heavy metals shall not exceed 10 parts per million.
Exemestane - Content determination
Authoritative Data Verified Data
measured by high performance liquid chromatography (General 0512).
chromatographic conditions and system suitability test
silica gel bonded with eighteen alkyl silane was used as a filler, and acetonitrile-water (35:65) was used as a mobile phase; The detection wavelength was 249nm; And the column temperature was 40°C. Take appropriate amount of exemestane control and impurity I control, add mobile phase to dissolve and dilute to make a mixed solution containing 2.5ug of exemestane and impurity I per 1 ml, as the system applicable solution, take 20ul injection liquid chromatograph and record the chromatogram. The resolution between exemestane peak and impurity I peak should be greater than 2.5.
assay
take about 25mg of this product, weigh it accurately, put it in 50ml measuring flask, add appropriate amount of mobile phase to dissolve and dilute to the scale, shake well, take 5ml accurately, put it in 100ml measuring flask, dilute to the scale with mobile phase, shake well, as a test solution, and inject 20ul into the liquid chromatograph with precision, record the chromatogram; Take the exemestane reference substance, and determine with the same method. According to the external standard method to calculate the peak area, that is.
Exemestane - Category
Authoritative Data Verified Data
antineoplastic agents.
Exemestane - Storage
Authoritative Data Verified Data
light shielding, sealed storage.
Exemestane - Exemestane Tablets
Authoritative Data Verified Data
This product contains exemestane (C20H2402) should be 93.0% to 107.0% of the label.
trait
This product is white or white-like tablets or film-coated tablets, white or white-like after removing the coating.
identification
- take an appropriate amount of fine powder of this product (about 5mg equivalent to exemestane), add about 10ml of ethanol, fully shake to dissolve exemestane, filter, and evaporate the filtrate to dryness on a water bath, the residue was dissolved by adding 3ml of sulfuric acid and allowed to stand for 5 minutes. The solution was graded to orange-red and poured into 1 ml of water.
- in the chromatogram recorded under the content determination item, the retention time of the main peak of the test solution should be consistent with the retention time of the main peak of the reference solution.
- take an appropriate amount of fine powder of this product and make it into a solution containing about 10ug of exemestane per lml, filter it, and then take the filtrate for determination by UV-Vis spectrophotometry (General rule 0401), there is an absorption maximum at a wavelength of 246nm.
examination
- relevant substances: Take appropriate amount of fine powder of this product, add mobile phase to dissolve and dilute to make a solution containing about 0.5mg of exemestane per 1ml, filter, and take the continued filtrate as the test solution; take 1ml of precision, 200ml children's bottle, diluted with mobile phase to scale, as a control solution. According to the chromatographic conditions under the item of centistokes determination, 20 u1 of the test solution and 20 u1 of the control solution are respectively injected into the liquid chromatograph, and the chromatogram is recorded to 2.5 times of the retention time of the main component peak. If there are impurity peaks in the chromatogram of the test solution, the area of a single impurity peak shall not be more than 2 times (1.0%) the area of the main peak of the control solution, the sum of each impurity peak area shall not be greater than 3 times (1.5%) of the main peak area of the control solution.
- dissolution this product, according to the dissolution and release determination method (General rule 0931-method), with 0.5% of sodium dodecyl sulfate solution as dissolution medium, speed of 100 rpm, operate according to law, after 30 minutes, take 10ml of solution, filter, Take 5ml of filtrate accurately, put it in a 10ml measuring flask, dilute it to the mark with 0.5% sodium dodecyl sulfate solution, shake it well, as a test solution. An appropriate amount of exemestane reference substance was added, and 0.5% sodium dodecyl sulfate solution was added to dissolve and quantitatively dilute to prepare a solution containing about 10% per 1 ml, which was used as the reference solution. The absorbance of the above two Solutions was measured at a wavelength of 0401 NM according to ultraviolet-visible spectrophotometry (general), and the elution amount of each tablet was calculated. The limit is 75% of the labeled amount and shall be in accordance with the provisions.
- others shall be in accordance with the relevant provisions under the item of tablets (General rule 0101).
Content determination
Take 20 tablets of this product, precision weighing, fine grinding, precision weighing appropriate amount (about equivalent to exemestane 25mg), put it in 50ml measuring flask, add appropriate amount of mobile phase, shake to dissolve exemestane, dilute to the scale with mobile phase, shake, filter, Take 5ml of filtrate accurately, put it in a 100ml measuring flask, dilute to the scale with mobile phase, shake well, as a test solution, precision measure 20 u1, according to the method under the determination of exemestane content, that is obtained.
category
Same as exemestane.
specification
25mg
storage
light shielding, sealed storage.
Exemestane - Exemestane capsules
Authoritative Data Verified Data
This product contains exemestane (C20H2402) should be 93.0% to 107.0% of the label.
trait
The contents of this product are white or off-white powder or granular powder.
identification
- take an appropriate amount of the contents of this product (about 5mg equivalent to exemestane), add about 10ml of ethanol, fully shake to dissolve exemestane, filter, and evaporate the filtrate to dryness on a water bath, the residue was dissolved by adding 3ml of sulfuric acid and allowed to stand for 5 minutes. The solution was graded to orange-red and poured into 1 ml of water.
- in the chromatogram recorded under the content determination item, the retention time of the main peak of the test solution should be consistent with the retention time of the main peak of the reference solution.
- take an appropriate amount of the contents of this product, add ethanol to make a solution containing about 10ug of exemestane per lml, filter, take the filtrate, and measure it by UV-Vis spectrophotometry (General 0401), there is an absorption maximum at a wavelength of 246nm.
examination
- relevant substances: Take appropriate amount of the contents of this product, add the mobile phase to dissolve and dilute to make a solution containing about 0.5mg of exemestane per 1ml, filter, and take the continued filtrate as the test solution; take 1ml of precision, 200ml flask, diluted with mobile phase to the scale, as a control solution. According to the chromatographic conditions under the content determination item, 20 u1 of the test solution and the control solution are accurately weighed and injected into the human liquid chromatograph respectively, and the chromatogram is recorded to 2.5 times of the retention time of the main component peak. If there are impurity peaks in the chromatogram of the test solution, the area of a single impurity peak shall not be more than 2 times (1.0%) the area of the main peak of the control solution, the sum of each impurity peak area shall not be greater than 3 times (1.5%) of the main peak area of the control solution.
- dissolution of this product, according to the dissolution and release determination method (General 0931 first method), with 0. ML of sodium dodecyl sulfate solution is the dissolution medium, and the rotation speed is 100 RPM. After 30 minutes, 10ml of the solution is taken, filtered, and 5ml of the continued filtrate is accurately measured and placed in a 10ml measuring flask, dilute to the mark with 0.5% sodium dodecyl sulfate solution and shake to obtain a test solution. In addition, an appropriate amount of exemestane reference product was accurately weighed, dissolved and quantitatively diluted with 0.5% sodium dodecyl sulfate solution to prepare a solution containing about 10ug per 1 ml as a reference solution. The absorbance of the above two Solutions was measured at a wavelength of 0401 Mn by ultraviolet-visible spectrophotometry (general), and the amount of dissolution of each particle was calculated. The limit is 75% of the labeled amount and shall be in accordance with the provisions.
- others should comply with the relevant provisions under the capsule (General 0103).
Content determination
take the contents under the difference of loading amount, mix evenly, accurately weigh an appropriate amount (about 25mg equivalent to exemestane), put it in a 50ml measuring flask, and add an appropriate amount of mobile phase, shake to dissolve exemestane, dilute to scale with mobile phase, shake well, filter, Take 5ml of continued filtrate precisely, put it in 100ml measuring flask, dilute to scale with mobile phase, shake well, as a test solution, 20UL was accurately measured and obtained according to the method under the item of exemestane content determination.
category
Same as exemestane.
specification
25mg
storage
light shielding, sealed storage.
Supplier List
CAS: 107868-30-4
Tel: +86-21-56795779
Email: charles7788@worldyachem.com
Mobile: +86-13651600618
QQ: 2850607228
Wechat: 13651600618
WhatsApp: +8613651600618
CAS: 107868-30-4
Tel: 13247110337
Email: 2205437118@qq.com
Mobile: 13247110337
QQ: 2205437118
Wechat: 13247110337
CAS: 107868-30-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 107868-30-4
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 107868-30-4
Tel: +86-21-56795779
Email: charles7788@worldyachem.com
Mobile: +86-13651600618
QQ: 2850607228
Wechat: 13651600618
WhatsApp: +8613651600618
CAS: 107868-30-4
Tel: 13247110337
Email: 2205437118@qq.com
Mobile: 13247110337
QQ: 2205437118
Wechat: 13247110337
CAS: 107868-30-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 107868-30-4
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog



