DITHIANON
dithianon
CAS: 3347-22-6
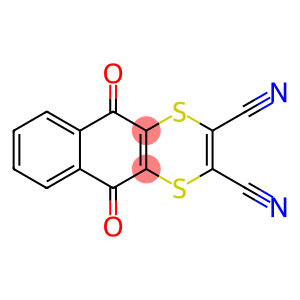
Molecular Formula: C14H4N2O2S2
DITHIANON - Names and Identifiers
DITHIANON - Physico-chemical Properties
| Molecular Formula | C14H4N2O2S2 |
| Molar Mass | 296.32 |
| Density | d18 1.55 |
| Melting Point | 225° |
| Boling Point | 402.1±45.0 °C(Predicted) |
| Flash Point | 197°C |
| Water Solubility | 0.5mg/L(temperature not stated) |
| Vapor Presure | 1.13E-06mmHg at 25°C |
| Appearance | neat |
| Merck | 13,3404 |
| BRN | 1325563 |
| Storage Condition | 0-6°C |
| Refractive Index | 1.6360 (estimate) |
| In vitro study | When exponentially aerobic growing cells of S. cerevisiae are submitted to acute Dithianon treatment they loss cell wall and membrane integrity, dying by necrosis, and this behavior is associated to a depletion of reduced proteic and non-proteic thiol groups. An important increase of cellular reactive oxygen species (ROS) associated to mitochondrial membrane potential modifications on Dithianon treated cells are also detected. In filamentous fungus, Dithianon inhibits mycelial growth and conidial germination. Studies on Ehrlich ascites carcinoma and yeast cells showed that Dithianon inhibits respiration and fermentation affecting several thiol enzymes of the glycolytic pathway as hexokinase, phosphofructokinase, and glyceraldeyde-3-phosphate dehydrogenase. Dithianon has in vitro cytotoxic effect and affect cell transforming activity of BLAB/c 3 T3 cells. |
| In vivo study | The activity of testosterone hydroxylase of liver microsomes derived from male mice is increased when they are treated with acute doses of Dithianon, while in females an inactivating effect is observed. |
DITHIANON - Risk and Safety
| Risk Codes | R22 - Harmful if swallowed R50/53 - Very toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment. |
| Safety Description | S24 - Avoid contact with skin. S60 - This material and its container must be disposed of as hazardous waste. S61 - Avoid release to the environment. Refer to special instructions / safety data sheets. |
| UN IDs | UN 3077 |
| WGK Germany | 3 |
| RTECS | QL0700000 |
| HS Code | 29349990 |
| Hazard Class | 6.1(b) |
| Packing Group | III |
| Toxicity | LD50 orally in rats, guinea pigs: 638, 110 mg/kg (Amadori, Heupt) |
DITHIANON - Reference Information
| NIST chemical information | information provided by: webbook.nist.gov (external link) |
| EPA chemical substance information | information provided by: ofmpeb.epa.gov (external link) |
| Application | dicyanoanthraquinone is a kind of bactericide with quinone structure and multi-site, inhibition of a series of fungal enzymes by reaction with sulfur-containing groups and interference with cellular respiration eventually leads to disease death. It has a good protective activity, but also has a certain therapeutic activity. |
| biological activity | Dithianon is a broad-spectrum anthraquinone fungicide with good adhesion to the surface of leaves and fruits. Dithianon is used to control several fungi on certain fruits and vegetables, such as Colletotrichum sp., Elsinoe ampelina, mold (Plasmopara viticola), phosphorescent bacteria (photophore viticola), etc. |
| Target | Reactive oxygen species (ROS) Colletotrichum sp.; elsinoe ampelina ; Plasmopara viticola; Phopsis viticola |
| toxic substance data | information provided by: pubchem.ncbi.nlm.nih.gov (external link) |
Last Update:2024-04-09 02:00:04
Supplier List
Spot supply
Product Name: Anthraquinone Impurity 14 Visit Supplier Webpage Request for quotationCAS: 3347-22-6
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Dithianon Visit Supplier Webpage Request for quotationCAS: 3347-22-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: dithianon Request for quotationCAS: 3347-22-6
Tel: +86-17551318830
Email: 3785839865@qq.com
Mobile: 13776910623
QQ: 3785839865
Product Name: DITHIANON Request for quotation
CAS: 3347-22-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 3347-22-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: dithianon Request for quotation
CAS: 3347-22-6
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 3347-22-6
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
Spot supply
Product Name: Anthraquinone Impurity 14 Visit Supplier Webpage Request for quotationCAS: 3347-22-6
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Dithianon Visit Supplier Webpage Request for quotationCAS: 3347-22-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: dithianon Request for quotationCAS: 3347-22-6
Tel: +86-17551318830
Email: 3785839865@qq.com
Mobile: 13776910623
QQ: 3785839865
Product Name: DITHIANON Request for quotation
CAS: 3347-22-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 3347-22-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: dithianon Request for quotation
CAS: 3347-22-6
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 3347-22-6
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
View History




