DERMATANSULFATEPORCINE
CHONDROITIN SULFATE B SODIUM SALT
CAS: 54328-33-5
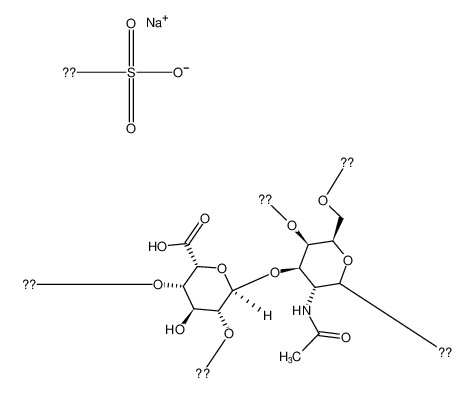
Molecular Formula:
DERMATANSULFATEPORCINE - Names and Identifiers
DERMATANSULFATEPORCINE - Physico-chemical Properties
| Molar Mass | 1526.03 |
| Storage Condition | Room Temprature |
| Refractive Index | -68 |
DERMATANSULFATEPORCINE - Risk and Safety
| Safety Description | S22 - Do not breathe dust. S24/25 - Avoid contact with skin and eyes. |
DERMATANSULFATEPORCINE - RP-HPLC determination of chondroitin sulfate
from VIP
Author:
pan , Tao Liping , Li Aizhen , Chen Xinzhi
Abstract:
A RP-HPLC method was established for the simultaneous determination of total chondroitin sulfate and its components A,B,C,D. A C18 column was used with the mobile phase of acetonitrile-0.5% phosphoric acid-Isopropanol (15:85:1) at the detection wavelength of 209nm and the column temperature of 40 ℃. The linear range of chondroitin sulfate was 0.4~2 mg/mL (r = 0.9997). The average recovery was 99.3% with RSD of 1.16%.
Key words:
chondroitin sulfate high performance liquid chromatography assay
DOI:
10.3969/j.issn.1001-8255.2007.09.019
cited:
year:
2007
DERMATANSULFATEPORCINE - Production device for refining chondroitin sulfate B
Application (patent) number:
202120212321
applicant (patent):
Binzhou College
inventor:
DERMATANSULFATEPORCINE - Application of chondroitin sulfate B and vaccine containing chondroitin sulfate B
from Palm Bridge research
Application (patent) number:
CN201510008740.4
application date:
2015-01-08
Public/Announcement Number:
CN104491859A
Public/announcement date:
2015.04.08
applicant (patent):
Institute of Medical Biology, Chinese Academy of Medical Sciences
inventor:
Wang Chen Yun , Wang , Sun Jing , Li Yanhan , Ummini , Li Jianfang , ha , Yun Hu , Shi Jiandong
National and provincial code:
CN530102
Abstract:
The present invention provides an application of chondroitin sulfate B and a vaccine agent containing chondroitin sulfate B, wherein chondroitin sulfate B is used as an adjuvant in a vaccine, that is, 5 to 105 μg of chondroitin sulfate B is contained in each single vaccine dose. Chondroitin sulfate B as a vaccine adjuvant, toxic side effects, in the immune dose range is safe and reliable, can effectively induce antigen-specific humoral immune response, and the induced humoral immune response effect is better than the adjuvant group, chondroitin sulfate B performance is stable, high biological potency, no toxic effect, can be added to a variety of traditional vaccines and genetic engineering vaccines as vaccine adjuvants.
Supplier List
CAS: 54328-33-5
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
CAS: 54328-33-5
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 54328-33-5
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 54328-33-5
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 54328-33-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 54328-33-5
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
CAS: 54328-33-5
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 54328-33-5
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 54328-33-5
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 54328-33-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025




