68401-82-1
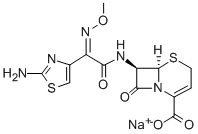
ceftizoxime sodium
CAS: 68401-82-1
Molecular Formula: C13H14N5NaO5S2
68401-82-1 - Names and Identifiers
| Name | ceftizoxime sodium |
| Synonyms | Ceftix Cefizox Ceftizon Eposerin Ceftizoxime sodium ceftizoxime sodium Ceftizoxime, sodium salt Sodium 7-[2-(2-aminothiazol-4-yl)-2-methoxyiminoacetamido]-3-cephem-4-carboxylate (6r,7r)-7-[[(2z)-(2-amino-4-thiazolyl)(methoxyimino)acetyl]amino]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid sodium salt sodium (6R,7R)-7-{[(2Z)-2-(2-amino-1,3-thiazol-4-yl)-2-(methoxyimino)acetyl]amino}-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylate 5-Thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid, 7-[[(2Z)-(2-amino-4-thiazolyl)(methoxyimino)acetyl]amino]-8-oxo-, monosodium salt, (6R,7R)- 5-Thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid, 7-[[(2-amino-4-thiazolyl)(methoxyimino)acetyl]amino]-8-oxo-, monosodium salt, [6R-[6α,7β(Z)]]- |
| CAS | 68401-82-1 |
| EINECS | 629-729-8 |
| InChI | InChI=1/C13H13N5O5S2.Na/c1-23-17-7(5-4-25-13(14)15-5)9(19)16-8-10(20)18-6(12(21)22)2-3-24-11(8)18;/h2,4,8,11H,3H2,1H3,(H2,14,15)(H,16,19)(H,21,22);/q;+1/p-1/b17-7-;/t8-,11-;/m1./s1 |
68401-82-1 - Physico-chemical Properties
| Molecular Formula | C13H14N5NaO5S2 |
| Molar Mass | 407.39 |
| Melting Point | 227℃ |
| Water Solubility | Soluble in water |
| Solubility | DMSO: soluble1mg/mL |
| Appearance | solid |
| Color | white to faint yellow |
| Storage Condition | -20°C |
| Physical and Chemical Properties | White to yellowish crystalline powder. |
68401-82-1 - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | 26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. |
| WGK Germany | 3 |
| RTECS | XI0368000 |
| Toxicity | LD50 in mice, rats (mg/kg): ~6000 i.v. (Fukuhara) |
68401-82-1 - Standard
Authoritative Data Verified Data
This product is (6R,7R)-7-[2-(2-aminothiazol-4-yl)-2-(methoxyimino) acetamido]-8-oxo-5-thia-1-azabicyclo [4.2.0] oct-2-ene-2-carboxylic acid sodium salt. The content of ceftizoxime (C13H13N5O5S2), calculated as anhydrous, shall not be less than 90.0%.
68401-82-1 - Trait
Authoritative Data Verified Data
- This product is white to light yellow crystalline, crystalline or granular powder; Odorless or slightly odorous; Slightly hygroscopic.
- This product is very soluble in water, very slightly soluble in methanol, and almost insoluble in ethanol and acetone.
specific rotation
take this product, precision weighing, add water to dissolve and quantitatively dilute to make a solution containing about 10mg per lml, and determine according to law (General rule 0621), the specific rotation is from +125 ° to +145 °.
absorption coefficient
take this product, precision weighing, add water to dissolve and quantitatively dilute to make a solution containing about 10% per lml, according to UV-visible spectrophotometry (General 0401), the absorbance was measured at a wavelength of 235nm, and the absorption coefficient was 410 to 450.
68401-82-1 - Preparation solution concentration reference
| 1mg | 5mg | 10mg | |
|---|---|---|---|
| 1 mM | 2.467 ml | 12.334 ml | 24.668 ml |
| 5 mM | 0.493 ml | 2.467 ml | 4.934 ml |
| 10 mM | 0.247 ml | 1.233 ml | 2.467 ml |
| 5 mM | 0.049 ml | 0.247 ml | 0.493 ml |
68401-82-1 - Differential diagnosis
Authoritative Data Verified Data
- Take appropriate amount of this product and ceftizoxime reference, add phosphate buffer (pH7.0 )5ml to shake to dissolve, dilute with 75% ethanol to prepare a solution containing about 5mg of ceftizoxime per 1 ml as the test solution and the control solution; Take the appropriate amount of ceftizoxime control and cefradine control, phosphate buffer (pH 7.0) was added to dissolve, and a solution containing about 5mg of ceftizoxime and 5mg of cefradine per 1 ml in 75% ethanol was prepared as a system suitability solution. According to the thin layer chromatography (General 0502) test, absorb the above three solutions each 2 u1, respectively, on the same silica gel GF254 thin layer plate, with ethyl acetate-ether-two gas methane-formic acid (5:4:5:6) for the development of the solvent, expand, dry, set the UV lamp (254mn) under the inspection or set the iodine vapor in the color. The system applicable solution should show two clearly separated spots, and the position and color of the main spot displayed by the test solution should be the same as the position and color of the main spot of the reference solution.
- in the chromatogram recorded under the content determination item, the retention time of the main peak of the test solution should be consistent with the retention time of the main peak of the reference solution.
- The infrared absorption spectrum of this product should be consistent with that of the control (Spectrum set 723).
- This product shows the reaction of sodium salt identification (1) (General rule 0301).
- two items (1) and (2) above can be selected as one item.
68401-82-1 - Exam
Authoritative Data Verified Data
crystallinity
take this product, determination according to law (General 0981), should comply with the provisions.
pH
take this product, add water to make a solution containing ceftizoxime O.lg per lml, and determine it according to law (General rule 0631). The pH value should be 6.0~8.0.
clarity and color of solution
take 5 parts of this product, each 0.6g, respectively, after adding water 5ml to dissolve, the solution should be clear and colorless; If it is turbid, compare with No. 1 turbidity standard solution (General rule 0902 first method), shall not be more concentrated; If the color is developed, it shall not be deeper in comparison with the yellow or yellow-green standard colorimetric solution No. 6 (General Principles 0901 first method).
Related substances
Take appropriate amount of this product, precision weighing, add pH 7.0 phosphate buffer (take potassium dihydrogen phosphate 3.63g, disodium hydrogen phosphate 14.33g, add water to dissolve and dilute to 1000ml) dissolve and dilute to prepare a solution containing about 0.5mg of ceftizoxime per 1 ml as a test solution, quantitative dilution with pH 7.0 phosphate buffer to prepare a solution containing about 5ug of ceftizoxime per 1 ml, as a control solution, according to the chromatographic conditions under the content determination item, 20ul of the test solution and the control solution were respectively injected into the human liquid chromatograph, and the chromatogram was recorded to 3 times of the retention time of the main component peak. If there are impurity peaks in the chromatogram of the test solution, the area of a single impurity peak shall not be greater than 0.5 times (0.5%) the area of the main peak of the control solution, and the sum of the areas of each impurity peak shall not be greater than the area of the main peak of the control solution (1.0%)
ceftizoxime polymer
measured by size exclusion chromatography (General 0514).
chromatographic conditions and system suitability test
with dextran gel G-10(40 ~ 120um) as the filler, the inner diameter of the glass column is 1.0~1.4, the length of the column is 30 ~ 40cm, mobile phase A was 7.0 mol/L phosphate buffer at pH 0.075 [0.O75mol/L disodium hydrogen phosphate solution -0.075mol/L sodium dihydrogen phosphate solution (61:39 )]; Mobile phase B is water, flow rate is 1.5ml per minute, detection wavelength is 254nm, take 2000~200u1 of 100 mg /ml Blue dextran solution and inject it into human Liquid Chromatograph. Use mobile phase A and B as mobile phase respectively, and record chromatogram. The number of theoretical plates was not less than 2000 based on the Blue dextran 300 peak. The tailing factor should be less than 2.0, and the retention time ratio of the Blue dextran 2000 peak in the two mobile phase systems should be between 0.93 and 1.07, the ratio of the retention time of the main peak of the control solution to the polymer peak in the test solution and the Blue dextran 2000 peak in the corresponding chromatographic system should be between 0.93 and 1.07, weigh about 0.2g of ceftizoxime sodium, put it in a 10ml measuring flask, add 2000 mg /ml Blue dextran solution to dissolve and dilute to the mark, shake well, take 100~200u1 and inject it into human liquid chromatograph, and measure with mobile phase A. The peak height of high polymer and the valley height ratio between monomer and high polymer should be greater than 2.0, the relative standard deviation of peak area shall not be more than 100 for 5 consecutive injections of 2000 ~ 5.0% control solution.
preparation of control solution
The ceftizoxime reference substance 12.5mg was accurately weighed, placed in a 500ml measuring flask, dissolved in water, diluted to the scale, and shaken to obtain the product.
assay
take about 0.3g of this product, weigh it accurately, put it in a 10ml measuring flask, add water to dissolve and dilute it to the scale, shake it well. Immediately take 100~200u1 injection precision liquid chromatography. The mobile phase A was used as the mobile phase, and the chromatogram was recorded. In addition, 100~200u1 of the control solution was injected into the human liquid chromatograph, and the mobile phase B was used as the mobile phase for measurement, and the chromatogram was recorded. The amount of ceftizoxime-containing polymer, calculated by the area of the ceftizoxime peak by the external standard method, shall not exceed 0.10%.
moisture
take this product, according to the moisture determination method (General 0832 first method 1), the water content shall not exceed 8.5%.
visible foreign body
take 5 parts of this product, each 2. 0g, plus particle inspection water dissolution, inspection according to law (General rule 0904), should comply with the provisions. (For aseptic dispensing)
insoluble particles
Take 3 parts of this product, and add the particle inspection water to make a solution containing 60mg per 1 ml, and check according to law (General rule 0903 ), no more than 6000 particles with lOum and more than 10um and no more than 600 particles with 25um and more than 25um in each lg sample. (For aseptic dispensing)
bacterial endotoxin
take this product, check according to law (General rule 1143), the amount of endotoxin per 1 mg ceftizoxime should be less than 0.10EU. (For injection)
sterile
take this product, dissolve and dilute with 0.1% sterile peptone aqueous solution to prepare a solution containing 40mg per 1 ml, and treat it by membrane filtration, wash with 0.1% sterile peptone solution (not less than 600ml per membrane), take Escherichia coli as positive control bacteria, check according to law (General rule 1101 ) , should comply with the regulations. (For aseptic dispensing)
68401-82-1 - Content determination
Authoritative Data Verified Data
measured by high performance liquid chromatography (General 0512).
chromatographic conditions and system suitability test
silica gel bonded with OCTA alkyl silane as filler; Acetonitrile (9:1)-pH 3.6 buffer solution (citric acid 1.42g, disodium hydrogen phosphate 2.31g, dissolved in water and diluted to ML) mobile phase; The detection wavelength was 254mn. Weigh 5mg of ceftizoxime control, put it in a 10ml measuring flask, add 0.1 mol/L sodium hydroxide solution 1ml, 30 minutes, diluted with pH 7.0 phosphate buffer to the scale, shake, A mixed solution containing about 4% of the degradation product of ceftizoxime base (its relative retention time is about 0.8) was prepared, and 20u1 was injected into the human liquid chromatograph, and the chromatogram was recorded, ceftizoxime peak retention time is about 8 minutes, ceftizoxime peak and its relative retention time of about 0.8 alkali degradation product peak separation degree should be greater than 3.0.
assay
take about 20mg of this product, accurately weigh, put it in a 200ml measuring flask, add pH 7.0 phosphate buffer solution and dilute to the scale, shake, as a test solution, the chromatogram was recorded by injection of 20u1 into human liquid chromatograph, and an appropriate amount of ceftizoxime reference substance was taken for determination by the same method. The content of C13H13N5O5S2 in the sample was calculated by the peak area according to the external standard method.
68401-82-1 - Category
Authoritative Data Verified Data
B-lactam antibiotics, cephalosporins.
68401-82-1 - Storage
Authoritative Data Verified Data
sealed and stored in a cool dark dry place.
68401-82-1 - Ceftizoxime Sodium for Injection
Authoritative Data Verified Data
This product is a sterile powder of ceftizoxime sodium. The content of ceftizoxime (C13H13N505S2) shall not be less than 90.0%, calculated as anhydrous; The content of ceftizoxime (C13H13N505S2) shall be 90.0% to 110.0% of the labeled amount, calculated as average loading.
trait
This product is white to light yellow crystalline, crystalline or granular powder.
identification
take this product, according to ceftizoxime sodium under the identification test, showed the same results.
examination
- the clarity and color of the solution take 5 bottles of this product, and add water according to the labeled amount to make each lml containing 0.lg solution, the solution should be clear and colorless; If it is turbid, it should not be more concentrated than the 1# turbidity standard solution (General 0902 first method); If it is colored, no deeper color shall be compared with the yellow or yellow-green standard colorimetric solution No. 6 (General rule 0901, Method 1).
- insoluble particles this product is taken, and the labeled amount is added to the particle inspection water to make a solution containing 60mg per 1 ml, which is inspected according to law (General rule 0903 ), and the labeled amount is l. The conversion below Og is that no more than 6000 particles with lOum and more than 10um and no more than 600 particles with 25um and more than 25um per g sample; The labeled amount is l.Og or more (including l. (Og) no more than 6000 particles with lOuw and more than 10um and no more than 600 particles with 25um and more than 25um in each sample container.
- the pH value, related substances, ceftizoxime polymer, moisture, bacterial endotoxin and sterility shall be checked according to the method under the item of ceftizoxime sodium.
- others should comply with the relevant provisions under injection (General 0102).
Content determination
The content under the charge amount difference item was obtained by measuring according to the method under the ceftizoxime sodium item.
category
with ceftizoxime sodium.
specification
Based on C13H13N505S2 (l) 0.5g (2 ) 0.75g(3)1.0g (4)1.5g (5) 2.og
storage
sealed and stored in a cool dark dry place.
Supplier List
CAS: 68401-82-1
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
CAS: 68401-82-1
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 68401-82-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 68401-82-1
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 68401-82-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 68401-82-1
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
CAS: 68401-82-1
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 68401-82-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 68401-82-1
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 68401-82-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025




