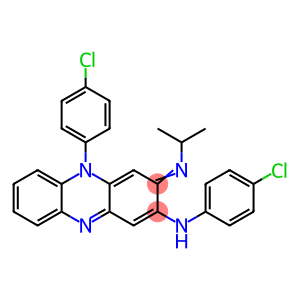
4-chloro-N-(5-(4-chlorophenyl)-3,5-dihydro-3-isopropyliminophenazin-2-yl)aniline
clofazimine
CAS: 2030-63-9
Molecular Formula: C27H22Cl2N4
4-chloro-N-(5-(4-chlorophenyl)-3,5-dihydro-3-isopropyliminophenazin-2-yl)aniline - Names and Identifiers
| Name | clofazimine |
| Synonyms | b-663 clofazimine b663(pharmaceutical) N,5-Bis(4-chlorophenyl)-3,5-dihydro-3-(isopropyliaino)phenazin-2-amine 2-phenazinamine,3,5-dihydro-n,5-bis(4-chlorophenyl)-3-((1-methylethyl)imino) 3-(p-chloranilino)-10-(p-chlorphenyl)-2,10-dihydro-2-(isopropylimino)-phenaz (3E)-N,5-bis(4-chlorophenyl)-3-(propan-2-ylimino)-3,5-dihydrophenazin-2-amine N,10-Bis[4-chlorophenyl]-2,10-dihydro-2-[[1-methylethyl] imino]-3-phenazinamine 4-chloro-N-(5-(4-chlorophenyl)-3,5-dihydro-3-isopropyliminophenazin-2-yl)aniline 3-(p-Chloroanilino)-10-(p-Chlorophenyl)-2,10-dihydro-2-(iso-propylimi-no)phenazine |
| CAS | 2030-63-9 |
| EINECS | 217-980-2 |
| InChI | InChI=1/C27H22Cl2N4/c1-17(2)30-24-16-27-25(15-23(24)31-20-11-7-18(28)8-12-20)32-22-5-3-4-6-26(22)33(27)21-13-9-19(29)10-14-21/h3-17,31H,1-2H3/b30-24- |
4-chloro-N-(5-(4-chlorophenyl)-3,5-dihydro-3-isopropyliminophenazin-2-yl)aniline - Physico-chemical Properties
| Molecular Formula | C27H22Cl2N4 |
| Molar Mass | 473.4 |
| Density | 1.1342 (rough estimate) |
| Melting Point | 210-212° |
| Boling Point | 616.26°C (rough estimate) |
| Water Solubility | 10mg/L(temperature not stated) |
| Solubility | Practically insoluble in water, soluble in methylene chloride, very slightly soluble in ethanol (96 per cent). It shows polymorphism (5.9). |
| Appearance | neat |
| Color | Yellow to Amber to Dark red |
| Merck | 14,2373 |
| pKa | 8.37; also reported as 8.51(at 25℃) |
| Storage Condition | 2-8°C |
| Refractive Index | 1.6300 (estimate) |
| Physical and Chemical Properties | Brick Red crystalline powder. Melting Point 212-213 °c. Soluble in chloroform, dimethylformamide, ethanol-soluble, insoluble in water. |
| Use | Mainly used for the treatment of dapsone-resistant Mycobacterium leprae infection |
| In vitro study | Clofazimine stimulates oxygen consumption and superoxide production by neutrophils. Clofazimine acts on neutrophils and activates phospholipase A2, which leads to increased release of lysophosphatidylcholine and arachidonic acid from neutrophil cell membranes. Clofazimine inhibits mitogen-induced stimulation of peripheral blood mononuclear cells. Clofazimine acts on macrophages to stabilize lysosomal membranes and inhibit the metabolism of leprosy bacilli in mouse peritoneal macrophages. According to the increase in the release of 3h-radiolabeled arachidonic acid and lysophosphatidylethanolamine ([3H] LPE) in bacterial membrane phospholipids, Clofazimine (5 mg/mL) the activity of phospholipase A2 can be increased in a dose-related manner by acting on Staphylococcus aureus. Clofazimine inhibits 90% of 20 strains of Mycobacterium tuberculosis, MICs <1.0 μg/mL. It should be noted that Clofazimine inhibits 2227 strains of Mycobacterium tuberculosis, MICs 0.06 μg/mL. Clofazimine (1 μg/mL) dose-dependently inhibited the activity of j774a.1 macrophages. |
| In vivo study | Clofazimine (20 mg/kg) prevented death and acted on the lungs and spleen of C57BL / 6 mice infected with M. Tuberculosis H37Rv, resulting in a significant reduction in the number of CFU. Liposomal Clofazimine (L-CLF) (50 mg/kg) acts on acutely infected mice with Mycobacterium tuberculosis Erdman, dose-dependently reducing CFU in the spleen, liver, and lungs by log 2 to 3. Clofazimine (500 μg twice daily) resulted in the highest concentration of Clofazimine in the spleen and liver of mice, while the concentration of Clofazimine in the lungs was significantly reduced. Clofazimine (20 mg/kg) was effective in reducing the bacterial load in the liver, spleen and lungs of C57BL / 6 mice experimentally infected with Mycobacterium avium strain tmc724. |
4-chloro-N-(5-(4-chlorophenyl)-3,5-dihydro-3-isopropyliminophenazin-2-yl)aniline - Risk and Safety
| Risk Codes | R22 - Harmful if swallowed R36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S36 - Wear suitable protective clothing. S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. |
| WGK Germany | 3 |
| RTECS | SG1578000 |
| HS Code | 35040000 |
| Toxicity | LD50 orally in mice, rats, and guinea pigs: >4 g/kg (Stenger) |
Supplier List
Spot supply
Product Name: clofazimine Visit Supplier Webpage Request for quotationCAS: 2030-63-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Clofazimine Request for quotation
CAS: 2030-63-9
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 2030-63-9
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Multiple SpecificationsSpot supply
Product Name: N,5-Bis(4-Chlorophenyl)-3,5-Dihydro-3-(Isopropylimino)Phenazin-2-Amine Visit Supplier Webpage Request for quotationCAS: 2030-63-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: clofazimine Visit Supplier Webpage Request for quotationCAS: 2030-63-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Clofazimine Request for quotation
CAS: 2030-63-9
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 2030-63-9
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Multiple SpecificationsSpot supply
Product Name: N,5-Bis(4-Chlorophenyl)-3,5-Dihydro-3-(Isopropylimino)Phenazin-2-Amine Visit Supplier Webpage Request for quotationCAS: 2030-63-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


