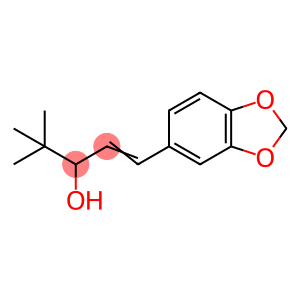
4,4-dimethyl-1-(3,4-methylenedioxyphenyl)-1-penten-3-ol
Stiripentol
CAS: 49763-96-4
Molecular Formula: C14H18O3
4,4-dimethyl-1-(3,4-methylenedioxyphenyl)-1-penten-3-ol - Names and Identifiers
| Name | Stiripentol |
| Synonyms | bcx2600 Diacomit 256-480-9 Stiripentol STIRIPENTOL 1-(1,3-BENZODIOXOL-5YL)-4,4-DIMETHYL-1-PENTEN-3-OL 1-(1,3-benzodioxol-5-yl)-4,4-dimethyl-1-penten-3-o 1-(1,3-benzodioxol-5-yl)-4,4-dimethylpent-1-en-3-ol 4,4-dimethyl-1-(3,4-methylenedioxyphenyl)-1-penten-3-o 4,4-dimethyl-1-(3,4-methylenedioxyphenyl)-1-penten-3-ol (1E)-1-(1,3-benzodioxol-5-yl)-4,4-dimethylpent-1-en-3-ol (RS)-(E)-4,4-dimethyl-1-[3,4(methylenedioxy)-phenyl]-1-penten-3-ol |
| CAS | 49763-96-4 |
| EINECS | 256-480-9 |
| InChI | InChI=1/C14H18O3/c1-14(2,3)13(15)7-5-10-4-6-11-12(8-10)17-9-16-11/h4-8,13,15H,9H2,1-3H3/b7-5+ |
| InChIKey | IBLNKMRFIPWSOY-FNORWQNLSA-N |
4,4-dimethyl-1-(3,4-methylenedioxyphenyl)-1-penten-3-ol - Physico-chemical Properties
| Molecular Formula | C14H18O3 |
| Molar Mass | 234.29 |
| Density | 1.139±0.06 g/cm3(Predicted) |
| Melting Point | 73-74°C |
| Boling Point | 365.4±11.0 °C(Predicted) |
| Flash Point | 174.8°C |
| Solubility | DMSO: ≥20mg/mL |
| Vapor Presure | 5.54E-06mmHg at 25°C |
| Appearance | powder |
| Color | white to beige |
| pKa | 14.45±0.20(Predicted) |
| Storage Condition | -20°C |
| Refractive Index | 1.578 |
| Use | This product is for scientific research only and shall not be used for other purposes. |
| In vitro study | Stiripentol (STP) is an anticonvulsant agent, which can inhibit N-demethylation of CLB to N-desmethylclobazam (NCLB) mediated by CYP3A4 (noncompetitively) and CYP2C19 (competitively). The inhibition of CLB demethylation by Stiripentol (STP) is best described by a noncompetitive inhibition model with apparent K i =1.6 μM for the cDNA-expressing CYP3A4 and by a competitive inhibition model with K i =0.52 μM for the cDNA-expressing CYP2C19. Formation of OH-NCLB from NCLB by cDNA-expressing CYP2C19 is competitively inhibited by Stiripentol (STP) with a K i =0.14 μM. |
| In vivo study | In mice treating with Stiripentol (STP) monotherapy, the difference between BT 1 (39.67±1.09°C) and BT 2 (41.32±1.05°C) reaches statistical significance (t=3.097, p<0.05). The difference in BT 2 between Stiripentol (STP) monotherapy and CLB monotherapy is statistically significant (t=2.615, p<0.05). In mice treating with Stiripentol (STP)+CLB combination therapy, the difference between BT 1 (40.18±0.58°C) and BT 2 (43.03±0.49°C) reaches statistical significance (t=10.44, p<0.01). |
4,4-dimethyl-1-(3,4-methylenedioxyphenyl)-1-penten-3-ol - Risk and Safety
| Hazard Symbols | Xn - Harmful |
| Risk Codes | 22 - Harmful if swallowed |
| WGK Germany | 3 |
| RTECS | SB3393000 |
4,4-dimethyl-1-(3,4-methylenedioxyphenyl)-1-penten-3-ol - Reference Information
| introduction | stinopentol (Stiripentol) is a new drug for the treatment of epilepsy listed in 2007, with the chemical name 4, 4-dimethylene-1-(3,4-methylenedioxyphenyl)-1-pentene-3-ol [4, 4-Dimethylene-1-(3,4-methylenedioxyphenyl)-1-penten-3-ol]; French researchers reported that the addition of stipentanol (Slipanto) to sodium valproate and clobazin can effectively treat severe myoclonic epilepsy in infants and young children; stipentanol (Slipanto) is an inhibitor of cytochrome P450. When used in combination with clobazin and valproate, stipranol (Slipanto) has a therapeutic effect on severe myoclonic epilepsy (SMEI). The University of Washington School of Medicine in Missouri, USA, believes that this combination drug experiment is difficult to determine that stivalol (Slipanto) reduces the frequency of seizures in patients, because the mechanism of action of stivalol (Slipanto) is Increase the blood concentration of other drugs such as clobazin and sodium valproate. |
| Use | The mechanism of action of stipranol (Slipanto) is relatively unique, which can not only increase the release of GABA, but also inhibit the activity of certain enzymes, Thereby increasing the blood concentration of other antiepileptic drugs. Stilpentanol (Slipanto) was approved in the European Union in 2001 for the adjuvant treatment of severe infantile epileptic myoclonic seizures. |
| Effects and uses | The mechanism of action of stipranol (Slipanto) is relatively unique, which can not only increase the release of GABA, but also inhibit the activity of certain enzymes, Thereby increasing the blood concentration of other antiepileptic drugs. Stilpentanol (Slipanto) was approved in the European Union in 2001 for the adjuvant treatment of severe infantile epileptic myoclonic seizures. |
| Adverse Reactions | Adverse reactions of stentopentol (Slipanto) treatment are mainly caused by its synergistic effect on other drugs. The combination with sodium valproate may increase the incidence of nausea and vomiting of sodium valproate. |
| Preparation | A method for preparing stentopentanol (Slipanto) includes the following steps:(1) 120g of piperonal is put into a mixed solvent of 0.2L ethanol and 2.4L of distilled water, 160g of solid NaOH, 2.6g of benzyltrimethylammonium chloride and 120g of methyl tert-butanone are added, and the reaction is stirred at 40°C. TLC detection reaction (developing agent: petroleum ether-ethyl acetate (6:1)), the reaction was complete after 16h. The reaction liquid is cooled to room temperature, filtered under reduced pressure, the filter cake is recrystallized with 0.12L absolute ethanol, and dried to obtain yellow needle-like crystals 4,4-dimethyl -1-[(3,4-methylenedioxy)-phenyl] -1-pentene -3-ketone (172.5g,93.2%),m.p.93.7~95.1 ℃. ESI-MS (m/z):233.1[M + H]+,255.1[M + Na]+. (2) Put the 4,4-dimethyl -1-[(3,4-methylenedioxy)-phenyl] -1-pentene -3-ketone obtained in step (1) into 0.8L ethanol, under stirring at 25~40 ℃, add 14.8g of sodium borohydride in batches within 10 minutes, and add sodium borohydride in batches to avoid uneven reaction caused by high local concentration, add 0.08L acetone and 0.12L distilled water, stir at 50 ℃ for 5 minutes, and bubble is generated. After the bubble disappears, continue to add water, and solid is precipitated. After the solid is precipitated completely, cool to room temperature, filter under reduced pressure, recrystallize the filter cake with 0.15L anhydrous ethanol, and dry to obtain white crystal stipenol (Slipanto) in total (91.1g,93.7%). |
| biological activity | Stiripentol (STP) is an anticonvulsant, which can inhibit CYP3A4 (non-competitive) and CYP2C19 (competitive) Mediated CLB to N-desmethylclobazam (NCLB) N-demethylation, its Ki values are 1.59±0.07 and 0.516±0.065 μM respectively, IC50 values are 1.58 and 3.29 μM respectively. |
| target | IC50: 1.58 μM (CYP3A4), 3.29 μM (CYP2C19) Ki: 1.59±0.07 μM (CYP3A4), 0.516±0.065 μM (CYP2C19) |
Last Update:2024-04-09 21:11:58
Supplier List
Featured ProductsSpot supply
Product Name: Stiripentol Visit Supplier Webpage Request for quotationCAS: 49763-96-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Stiripentol Request for quotation
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Stiripentol Visit Supplier Webpage Request for quotationCAS: 49763-96-4
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Product Name: STIRIPENTOL Request for quotation
CAS: 49763-96-4
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 49763-96-4
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Spot supply
Product Name: Stiripentol Visit Supplier Webpage Request for quotationCAS: 49763-96-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Featured ProductsSpot supply
Product Name: Stiripentol Visit Supplier Webpage Request for quotationCAS: 49763-96-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Stiripentol Request for quotation
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Stiripentol Visit Supplier Webpage Request for quotationCAS: 49763-96-4
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Product Name: STIRIPENTOL Request for quotation
CAS: 49763-96-4
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 49763-96-4
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Spot supply
Product Name: Stiripentol Visit Supplier Webpage Request for quotationCAS: 49763-96-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History




