23674-86-4
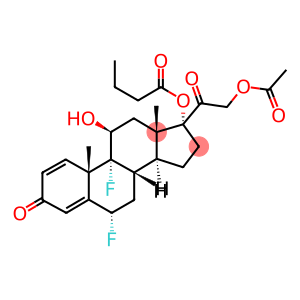
Difluprednate
CAS: 23674-86-4
Molecular Formula: C27H34F2O7
23674-86-4 - Names and Identifiers
23674-86-4 - Physico-chemical Properties
| Molecular Formula | C27H34F2O7 |
| Molar Mass | 508.55 |
| Density | 1.1869 (estimate) |
| Melting Point | 191-194° |
| Boling Point | 600.3±55.0 °C(Predicted) |
| Specific Rotation(α) | D22 +31.7° (c = 0.5 in dioxane) |
| Flash Point | 316.9°C |
| Solubility | Dioxane (Slightly), Methanol (Slightly) |
| Vapor Presure | 6.46E-17mmHg at 25°C |
| Appearance | Solid |
| Color | White |
| pKa | 12.56±0.70(Predicted) |
| Storage Condition | 2-8°C |
| Refractive Index | 1.544 |
23674-86-4 - Risk and Safety
| WGK Germany | 2 |
| RTECS | TU3831500 |
23674-86-4 - Preparation solution concentration reference
| 1mg | 5mg | 10mg | |
|---|---|---|---|
| 1 mM | 1.966 ml | 9.832 ml | 19.664 ml |
| 5 mM | 0.393 ml | 1.966 ml | 3.933 ml |
| 10 mM | 0.197 ml | 0.983 ml | 1.966 ml |
| 5 mM | 0.039 ml | 0.197 ml | 0.393 ml |
Last Update:2024-01-02 23:10:35
23674-86-4 - Reference Information
| overview | it is very important to quickly resolve inflammation and pain after ophthalmology. According to statistics, there are about 5 million eye surgeries in the United States every year, mainly cataract surgery and lens replacement. Despite advanced small incision techniques and improved lens emulsification, many patients still have a certain degree of inflammation after surgery. Surgical trauma alters the blood-aqueous barrier, causing protein leakage within the aqueous humor. If inflammation is not treated in time, it can interfere with the recovery of vision and even lead to serious complications, including pain, discomfort, photophobia, corneal edema and cystoid macular edema (CME). Clinically, CME is one of the blinding macular diseases. Whether it can be diagnosed and treated in time has a great influence on the prognosis of vision. Because the incidence of patients is difficult to predict, steroid hormones or non-steroidal anti-inflammatory drugs are commonly used for local prevention and treatment in the United States after surgery. On June 23, 2008, FDA approved the listing of 0.05% diflupredniside ophthalmic emulsion (Difluprednate,Durezol) developed by SirionTherapeutics company for the treatment of postoperative local inflammation and pain. Diflupredniside (difluoroprednisolone acetate butyrate) is a difluorinated derivative of prednisolone with potent anti-inflammatory activity. The molecular formula is C27H34F2O7 and the relative molecular mass is 508.56. Its structure is similar to that of other corticosteroids. Fluorination is at the carbon 6 position and carbon 9 position, which is the effective part. |
| Preparation | Difluoroprexide: 6 α, 9α-fluoro-11β-hydroxy-4-ene-pregnant-3-, 20-diketon-17-butyro-20-acetate. Add 200mg of intermediate 8 and 5ml to 50ml of polytetrafluoroethylene reaction bottle, cool to -25 ℃, add 2ml of HF pyridine solution, keep warm and stir for 4 hours, TLC monitor the reaction, adjust the pH to 7.5 with 30% sodium hydroxide solution, then pickling, washing with saturated salt water, dividing out DCM layer, drying anhydrous sodium sulfate and concentrating DCM to obtain brownish yellow oily liquid, column chromatography to obtain 160mg white solid with 80% yield, purity ≥ 99.0%. |
| Pharmacological effects and applications | Inflammatory reactions triggered by many stimulating factors can delay wound healing. Corticosteroid drugs can inhibit the occurrence of these inflammatory reactions and reduce inflammation-related edema, cellulose deposition, capillary swelling, leukocyte migration, capillary proliferation, fibroblast proliferation, collagen precipitation and scar formation. At present, the mechanism of action on ophthalmic corticosteroids is not obvious. However, it is generally believed that corticosteroids act by inducing phospholipase A2 inhibitors. Arachidonic acid is released from membrane phospholipids by phospholipase A2. The hypothesis is that these proteins control the biosynthesis of acting inflammatory mediators such as prostaglandins and interleukins by blocking the release of arachidonic acid, a common precursor of inflammatory mediators. The drug was originally used for skin, and as an ophthalmic drug, it can quickly and effectively pass through the corneal epithelium. It is then rapidly deacetylated into an active metabolite, difluoroprednisone butyrate (DFB), a highly active corticosteroid receptor agonist. This active ingredient will be converted into inactive metabolites. Unlike other commonly used steroidal eye drops, diflupenolate does not use benzalkalamine as a preservative. This tetravalent ammonium is a detergent that destroys cell walls by emulsifying cell membrane liposomes. Benzalkamine can destroy the stability of the tear membrane, have toxic effects on the corneal and conjunctival epithelium, and cause immune allergic reactions. In order to avoid the occurrence of these adverse reactions, diflupenib is used as a preservative with sorbic acid, which has little irritation and damage to eye tissues. |
| drug interaction | antibiotics can be used during surgery and after surgery, and a concomitant drug administration plan can be formulated two weeks before the test. If necessary, low doses of oral aspirin can prevent platelet aggregation. During surgery or after surgery, when performing fundus examination, pupil dilators can be used (on the 1st and 15th days, respectively). If the IOP is increased during the study, the researcher may decide to give intraocular pressure lowering drugs. |
| Adverse reactions | Side effects related to glucocorticoid eye drugs include: increased intraocular pressure (which may cause optic nerve damage), visual acuity and visual field defects, Subcapsular cataract formation, continued eye infection caused by pathogens such as herpes simplex virus, and will cause thinning of the sclera and cornea and perforation of the eyeball. The probability of adverse reactions in the subjects in clinical trials is 5% ~ 15%, including corneal edema, congestion of conjunctiva and ciliary body, eye pain, photophobia, turbidity of posterior capsule, flash of anterior chamber, conjunctival edema and eyelid inflammation. Other adverse effects including decreased visual acuity, punctate keratitis, ocular inflammation, and iritis occurred in the subjects with a probability of 1% to 5%. Less than 1% of the subjects had discomfort or irritation at the medication site, corneal pigmentation and keratopathy, scleral adventitis, eye itching, eyelid irritation and crusting, foreign body sensation, increased lacrimation, macular edema, scleral hyperemia and uveitis. The occurrence of the above adverse reactions may be related to the results of the operation. Overall, there were fewer adverse symptoms in the treatment group than in the placebo group. There were no serious adverse reactions in the treatment group, and there were few systemic non-ophthalmic adverse reactions. Most systemic symptoms occurred in a small number of patients, less than 2% of the number of patients in the treatment group, and only the incidence of constipation was 2.1% of the total number. During the study, one patient in the placebo group died of stroke, regardless of medication. |
| Pharmacokinetics | Preclinical pharmacokinetic and toxicity studies have shown that 0.05% diflopprednate ophthalmic emulsion administered 4 times a day has a non-toxic effect on the eyes. Pre-clinical studies on aqueous humor concentration showed that the bioavailability of emulsion was 40% higher than that of suspension. After single-or multiple-dose administration in rabbits, difloprexide is acetylated and distributed in the cornea, conjunctiva, iris, ciliary body, aqueous humor, retina, choroid, without accumulation in the blood. In addition, in animal experiments on uveitis and postoperative inflammation, the anti-inflammatory strength of diflopranide ophthalmic emulsion can be changed by changing the dosage of the drug. When used for mild inflammation, 0.05% concentrations of difloprexide are equivalent to 0.1% betamethasone sodium phosphate, and when used for severe inflammation, the effect is better than betamethasone. Difluprednate is deacetylated in vivo and converted into an active metabolite, 6-difluprednisone-butyrate (DFB). Clinical pharmacokinetic studies have shown that continuous administration through the eye, the amount of blood absorbed by the drug is very small. The drug 0.01% or 0.05% 2 drops each time, 4 times a day, the drug concentration in plasma failed to reach the detectable value (50ng/mL) after 7 days. |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: Difluprednate Visit Supplier Webpage Request for quotationCAS: 23674-86-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Difluprednate Visit Supplier Webpage Request for quotationCAS: 23674-86-4
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: Difluprednate Request for quotation
CAS: 23674-86-4
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 23674-86-4
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: Difluprednate Request for quotation
CAS: 23674-86-4
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 23674-86-4
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
Product Name: Difluprednate Visit Supplier Webpage Request for quotation
CAS: 23674-86-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 23674-86-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Difluprednate Visit Supplier Webpage Request for quotationCAS: 23674-86-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Difluprednate Visit Supplier Webpage Request for quotationCAS: 23674-86-4
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: Difluprednate Request for quotation
CAS: 23674-86-4
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 23674-86-4
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: Difluprednate Request for quotation
CAS: 23674-86-4
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 23674-86-4
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
Product Name: Difluprednate Visit Supplier Webpage Request for quotation
CAS: 23674-86-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 23674-86-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History




