1702-17-6
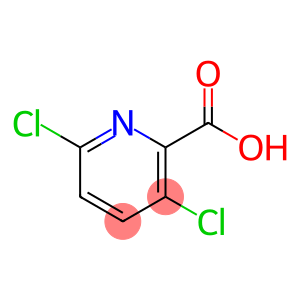
3,6-dichloropyridine-2-carboxylic acid
CAS: 1702-17-6
Molecular Formula: C6H3Cl2NO2
1702-17-6 - Names and Identifiers
| Name | 3,6-dichloropyridine-2-carboxylic acid |
| Synonyms | benzalox cirtoxin campaign Clopyralid acide3,6-dcp 3,6-dichlorpicolinic acid 3,6-dichloro-picolinicaci 3,6-dichloropicolinic acid acide3,6-dichloropicolinique 3-chloropyridine-2-carboxylic acid 3,6-dichloro-2-pyridinecarboxylicaci acidedichloro-3,6picolinique(french) 3,6-dichloropyridine-2-carboxylic acid 3,6-dichloro-pyridine-2-carboxylic acid |
| CAS | 1702-17-6 |
| EINECS | 216-935-4 |
| InChI | InChI=1/C6H4ClNO2/c7-4-2-1-3-8-5(4)6(9)10/h1-3H,(H,9,10) |
1702-17-6 - Physico-chemical Properties
| Molecular Formula | C6H3Cl2NO2 |
| Molar Mass | 192 |
| Density | 1.4313 (rough estimate) |
| Melting Point | 151-152°C |
| Boling Point | 0°C |
| Flash Point | 0°C |
| Water Solubility | 1.0 g/L |
| Vapor Presure | 0.000599mmHg at 25°C |
| Appearance | White or yellowish powder or crystal |
| Color | White to Light yellow to Light orange |
| Merck | 14,2400 |
| BRN | 473755 |
| pKa | 2.02±0.25(Predicted) |
| Storage Condition | Keep in dark place,Sealed in dry,Room Temperature |
| Refractive Index | 1.6100 (estimate) |
| MDL | MFCD00078655 |
| Use | For the control of broad-leaved weeds in rapeseed fields |
1702-17-6 - Risk and Safety
| Risk Codes | R41 - Risk of serious damage to eyes R51/53 - Toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S30 - Never add water to this product. S61 - Avoid release to the environment. Refer to special instructions / safety data sheets. S39 - Wear eye / face protection. |
| UN IDs | UN 3077 |
| WGK Germany | 3 |
| RTECS | TJ7550700 |
| TSCA | Yes |
| Hazard Class | 9 |
| Packing Group | III |
| Toxicity | LD50 in male, female rats (mg/kg): >5000, 4300 orally; LC50 (96 hr) to rainbow trout: 103.5 mg/l (Brown, Uprichard) |
1702-17-6 - Upstream Downstream Industry
| Raw Materials | Chlorine |
1702-17-6 - Reference Information
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| background and overview | rape is the main oil crop in Qinghai province. after agricultural restructuring in the past two years, the planting area has reached 200,000 hm2. Perennial Compositae weeds such as spiny cabbage and chicerae are common in Qinghai Province, and the harm is serious. The rhizome has strong reproductive ability, deep root system, strong drug resistance, and difficulty in prevention and removal, which has caused serious damage to agricultural production, especially spring rape production. harm. As there are fewer herbicides to control broad-leaved weeds in rape fields, 50% high-tech SC and artificial weeding are generally used to control prickling and chicory in rape fields. In addition, there are many kinds of weeds in corn fields. The main weeds include crabgrass, barnyard grass, purslane, amaranth, etc., which are one of the main factors affecting corn yield. The initial use of nitrosulfurone for weeding, and now its single agent and its compound preparations in China in a large number of registration, the corn field broad-leaved weeds Abutilon, Abutilon and grass weeds, barnyard grass and other effects are better, at the same time, the agent on the difficult to remove weeds of purslane, the shortcomings of the poor effect of amaranth is also increasingly obvious. dichloropyridinic acid (clopyralid) is a kind of systemic plant hormone herbicide, which is a product of Dow company in the United States. its trade name is "Longquan". its weeding effect is good and its selectivity is strong. In 1987, it entered the U.S. market for corn and sugar beet fields to control annual or perennial broad-leaved weeds. After the weeds are applied, they are absorbed by the leaves or roots of plants, and then conducted in plants, causing out-of-control and disorderly cell division. Growth, which eventually leads to the destruction of vascular bundles, can also inhibit cell division and growth, and is used to control 1-year or perennial broad-leaved weeds. At present, dichloropyridyl acid aqua and granules are mainly used to control broad-leaved weeds such as Rape and Enchicera, and the effect is obvious. At present, the domestic registration is mainly used to control a variety of broad-leaved weeds in corn, rape and wheat fields, but the agent is almost ineffective against gramineous weeds. |
| pharmacological action | after being applied to weeds, it is absorbed by the leaves or roots of plants, and then conducted in plants, causing uncontrolled cell division and disorderly growth, finally leading to the destruction of vascular bundles, and also inhibiting cell division and growth. |
| determination method | a solid phase extraction-gas chromatography method for the determination of dichloropyridyl acid in rape plants, rapeseed and soil was established by pre-column derivatization method. 1) pre-column derivatization reaction: transfer the extract to a plug test tube with ethyl acetate, blow dry with a nitrogen blow meter, add 1mL of methanol, 6 drops of concentrated H2SO4, cover the grinding plug, react in a water bath at 50 ℃ for 30 min, add 10 mL of distilled water after cooling, shake for 1min, extract with 4mL of n-hexane, and take 1mL of n-hexane phase through SP E. 2)SPE purification: after the Florsil solid phase extraction column is activated by 10 mLV (n-hexane) ∶V (ethyl acetate) = 19:1, then 10 mL n-hexane is used for equilibrium, the above 1 mL n-hexane phase is passed through the column, the flow rate is controlled to be 1~2 mL/min, eluted with 10 mLV (n-hexane):V (ethyl acetate) = 19:1, and the eluent is collected in a 10 mL volumetric flask, after nitrogen drying, add 1 mL n-hexane for determination. 3) chromatographic conditions: μ- ECD detector; Chromatographic column SE-30 capillary column, 30 m × 0.53mm (I. d.)× 0.5 μm; The inlet temperature is 240 ℃; The detector temperature is 260 ℃; The column temperature is 130 ℃; The column flow rate is 3 mL/min; The injection volume is 2 μL. 4) results: the retention time of dichloropyridinic acid was about 4.5 min. the average addition recovery rate of blank rape plants was 82.8% ~ 99.1%, RSD was 1.0% ~ 11%. The average addition recovery rate of rapeseed was 85.0% ~ 94.9 %,RSD was 2.8% ~ 8.5%. The average addition recovery rate of soil was 102.3% ~ 109.7%, RSD was 3.5% ~ 9.8%, and the detection limit (LOD) of the method was 1.87 μg/kg, the quantitative limit (LOQ) is 4.0 μg/kg, and this method can fully meet the requirements of pesticide residue analysis. |
| use | 1. control of spring rape field prickles and chicera: perennial chrysanthemum weeds such as prickles and chicera are common in Qinghai province, causing serious harm, with strong root reproduction ability, deep root system, strong drug resistance and difficulty in prevention and removal, causing serious harm to agricultural production, especially spring rape production. 30% Dichloropyridyl acid AS and 75% Longquan SG have the same control effect on Raspberry and Sonchus in different ecological regions, both of which are more than 30 percentage points higher than that of high Turk SC, and the control effect is excellent. The amount of 30% dichloropyridyl acid can be reduced to 450 ml/hm2 when the density of fields with small density occurs in spiny cabbage and chestnut, and the fields with high density can be selectively sprayed without the need for whole field medication to save costs. 2. The effect of controlling broad-leaved weeds in summer corn fields: Based on the results of two-year experiments, the 75% of dichloropyridyl acid soluble powder has excellent imitation effect on summer corn Asteraceae weeds such as Acantha, Spiceera, Bidens bipinnata, and Artemisia mugwort, which is better than that of the control agent 72% 2,4-D butyl emulsifiable concentrate treatment; the imitation effect on Phyllanthus, Tongquancao, mother grass, and Mo Shangcai is generally to excellent; there is a certain imitation of quinoa and amaranth weeds (amaranth, amaranth, etc.). The recommended dose in the field is 225~315g/hm2, and spray is applied at the 3-5 leaf stage of corn and 3-5 leaf stage of weeds. used to control broadleaf weeds in rape fields |
| study on digestion dynamics | in order to systematically study the residue digestion dynamics of dichloropidinic acid pesticide in rape and soil and promote safe production, a residue analysis method of dichloropidinic acid in rape and soil was established by gas chromatography, and the residue digestion dynamics of dichloropidinic acid in rape and soil were studied. rape and soil samples were extracted with ethyl acetate + petroleum ether 1:1 solution respectively, and were determined by gas chromatography-electron capture detector (GC-ECD) after methylation. dichloropyridinic acid was quantified by area external standard method. The results showed that the average recovery rate of dichloropyridinic acid in rape and soil was 86.3%-110.1%. The relative standard deviation was 2.4%-10.6%. The residual amount of dichloropyridinic acid decreased with the extension of time after application. The digestion dynamic curve conforms to the first-order kinetic equation. Its half-lives in rape and soil are 2.8d and 3.5d respectively, which are easily degradable pesticide compounds. |
| synthesis route | method 1: using methylpyridine as raw material, carrying out photocatalytic chlorination, fractionation after reaction, extracting the fraction at 100-104 ℃ at 2mmHg(266.644Pa) to obtain 3-chloro -2-(trichloromethyl) pyridine, further photocatalytic chlorination, and then crystallization treatment with n-hexane to obtain 3, 6-dichloro-2-(trichloromethyl) pyridine; then 3, 6-dichloro-2-(trichloromethyl) pyridine and concentrated nitric acid are mixed and refluxed for a period of time. After the reaction, it is cooled and crystallized. Filtration and benzene recrystallization to obtain dichloropyridine formic acid with high yield and purity. The specific reaction process is as follows: Method 2: Using tetrachloropyridine formonitrile as raw material, the dechlorination reaction is carried out with hydrazine hydrate as a reducing agent, and then dichloropilinic acid is obtained by hydrolysis and neutralization. The specific reaction process is as follows: Method 3: Use excess hydrazine hydrate to reduce 3,4,5,6-tetrachloropilinic acid in an aqueous solution of sodium carbonate at a certain temperature. The intermediate product 3,5, 6-trichloro-4-hydrazinyl pyridinic acid salt is obtained without adding hydrazine hydrate, dichloropyridinic acid is obtained by acidification with concentrated hydrochloric acid. The specific reaction process is as follows: Method 4: Using 3,4,5,6-tetrachloropilinic acid as raw material, electrolytic reduction in alkaline solution to obtain dichloropilinic acid. During the electrolysis process, the anode is deoxygenated, and the cathode is prepared with dichloropilinic acid. Cao Yong has no diaphragm electrolytic cell, the cathode material is silver, and the anode material is graphite, stainless steel or nickel and alloys. The specific reaction process is as follows: |
| main reference | [1] Wang Xiaojing et al. Study on the digestion dynamics of dichloropyridyl acid in rape and soil. Henan Agricultural Sciences. 2013,42(12):94-97. [2] Gao Xingxiang et al. The compounding of nitrosulpenone and dichloropridyl acid was applied to the determination of weeding effect in corn fields. Corn Science. 2015,23(3):143~148. [3] Wang xiumei et al. study on residue dynamics of dichloropyridyl acid in rape and soil. journal of Jilin agricultural university. 2006,28(4):430-432. [4] Wu Xiangwei et al. Determination of dichloropyridyl acid in rape and soil by pre-column derivatization-solid phase extraction/gas chromatography. Analytical laboratory. 2009,28(12):35-38. [5] Guo liangzhi et al. Effect of dichloropyridyl acid on control of spring rape fields of prickles and sorrel. Weed scientific 2009,1:53-54. [6] Zhu Jianyi et al. Pharmacological test of dichloropridyl acid on control of broad-leaved weeds in summer corn fields and winter rape fields. Scientific and technological innovation of plant protection and modern agricultural construction. [7] Ma Chunan et al. Progress in synthesis of 3, 6-dichloropyridine formic acid. Journal of Chemical Engineering. 2011, 62(9): 2398-2405. |
| toxic substance data | information provided by: pubchem.ncbi.nlm.nih.gov (external link) |
Last Update:2024-04-09 15:16:48
Supplier List
Spot supply
Product Name: Clopyralid Visit Supplier Webpage Request for quotationCAS: 1702-17-6
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: Clopyralid Visit Supplier Webpage Request for quotationCAS: 1702-17-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Clopyralid Request for quotation
CAS: 1702-17-6
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 1702-17-6
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: 3,6-dichloropyridine-2-carboxylic acid Request for quotation
CAS: 1702-17-6
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 1702-17-6
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
Multiple Specifications
Product Name: Clopyralid Visit Supplier Webpage Request for quotationCAS: 1702-17-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Clopyralid Visit Supplier Webpage Request for quotationCAS: 1702-17-6
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: Clopyralid Visit Supplier Webpage Request for quotationCAS: 1702-17-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Clopyralid Request for quotation
CAS: 1702-17-6
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 1702-17-6
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: 3,6-dichloropyridine-2-carboxylic acid Request for quotation
CAS: 1702-17-6
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 1702-17-6
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
Multiple Specifications
Product Name: Clopyralid Visit Supplier Webpage Request for quotationCAS: 1702-17-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History
Raw Materials for 1702-17-6




