13076-17-0
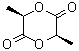
D(+)-LACTIDE
CAS: 13076-17-0
Molecular Formula: C6H8O4
13076-17-0 - Names and Identifiers
13076-17-0 - Physico-chemical Properties
| Molecular Formula | C6H8O4 |
| Molar Mass | 144.13 |
| Density | 1.186±0.06 g/cm3(Predicted) |
| Melting Point | 96.5-97.5 °C |
| Boling Point | 285.5±15.0 °C(Predicted) |
| Solubility | Soluble (130g/L) (25°C) |
| Appearance | White powder or crystal |
| Color | White to Off-White |
| Storage Condition | Sealed in dry,Room Temperature |
| MDL | MFCD00082566 |
13076-17-0 - Reference
| Reference Show more | 1. [IF=6.331] Cijun Shuai et al."A stereocomplexation constructed between poly (D-lactide) grafted hydroxyapatite and poly (L-lactide): Toward bioactive composite scaffold with enhanced interfacial bonding."J Mater Chem B. 2021 Dec;: |
13076-17-0 - Preparation and properties of L-lactide and poly (L-lactic acid)
from VIP
Author:
Lou ling , Yin Jingbo , high Battle Group , Liang Qi Zhi , donglisong , Chen Xuesi , landscape
Abstract:
oligomeric L-lactic acid was obtained from L-lactic acid by high temperature catalytic reaction, and then monomer L-lactide (LL A) was prepared by high temperature pyrolysis and vacuum distillation, the theoretical yield was over 80%. Poly (L-lactide, PLLA) was obtained by ring-opening polymerization of LL A Using stannous octoate as catalyst and its viscosity average molecular weight reached 4 0.
Key words:
L-lactide polylactic acid biodegradable material
DOI:
10.3321/j.issn:1000-7555.2003.02.017
cited:
year:
2003
13076-17-0 - Synthesis and biodegradability of starch/DL-lactide graft copolymer
from VIP
Author:
Abstract:
Starch/DL-lactide graft copolymer was synthesized in the presence of anhydrous LiCl with starch as graft backbone and DL-lactide as graft monomer. The effects of grafting ratio, reaction time and reaction temperature on monomer conversion (C%), grafting rate (G%) and grafting efficiency (GE%) were studied. When the molar ratio of DL-lactide to starch structural units is 10:1, the reaction temperature is 80~85 ℃, the reaction time is 4H, G% and GE% can reach 37.3%,179.7% and 68.0%, respectively. The graft copolymer was characterized by differential scanning calorimetry (DSC), infrared spectroscopy and X-ray diffraction, starch and DL-lactide were reacted to form starch/DL-lactide graft copolymer. The results of waterproof experiment show that the product can reduce the water absorption of paperboard from 41.1% to 1.0% under given conditions. The degradation experiments show that the graft copolymer can be completely degraded by acid, alkali and microorganism.expand
Key words:
starch; DL-lactide; graft copolymerization; Biodegradation
DOI:
10.3321/j.issn:1000-3304.2000.06.018
cited:
year:
2000
13076-17-0 - Study on Hydrolysis behavior of biodegradable polymer-polycaprolactone/polyoxyethylene/polylactide terpolymer
from titanium academic
Author:
Chen Hongli , beijianzhong , Royal Kingdom
Abstract:
PCL/PEO/PLA terpolymer was prepared by poly (ethylene glycol) ether (PEG)-initiated ring-opening polymerization of & epsilon; Caprolactone and L-lactide using stannous isooctate as catalyst. The in vitro degradation behavior of the polymer in pH 7 4 phosphate buffer solution at 37 ℃ was studied. The molecular weight, molecular weight distribution, composition, water absorption and crystallinity of the polymer during the hydrolytic degradation were studied by GPC,1H NMR,DSC and XRD techniques. The results show that the water absorption of the copolymer increases with the content of Polyether component, and the decrease of the content of Polyether component increases with the increase of the weight loss rate of the hydrolyzed material. In addition, the study also shows that when the content of lactide is high, the crystal structure of the polymer is mainly formed by PLLA. Since the hydrolytic degradation of the polymer first occurs in the amorphous region and the crystalline region edge, with the degradation of the copolymer, the formation of crystalline PLLA oligomer, resulting in the molecular weight of the copolymer is bimodal distributionexpand
Key words:
PCL/PEO/PLA terpolymer biodegradation crystallinity hydrophilic hydrolysis mechanism
DOI:
10.3321/j.issn:1000-3304.2000.05.022
cited:
year:
2000
13076-17-0 - Reference Information
| Use | D-( )-lactide is a cyclic diester of lactic acid, which is a by-product. When the body breaks down carbohydrates to obtain energy, muscle cells and red blood cells will produce this by-product. The lactide can be polymerized into polylactic acid using a suitable catalyst, resulting in a material with useful properties. |
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
Supplier List
CAS: 13076-17-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 13076-17-0
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 13076-17-0
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 13076-17-0
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 13076-17-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 13076-17-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 13076-17-0
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 13076-17-0
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 13076-17-0
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 13076-17-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025




