1,3,5-Triazine
S-triazine (1,3,5)
CAS: 290-87-9
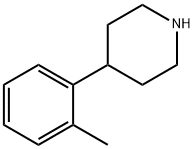
Molecular Formula: C12H17N
1,3,5-Triazine - Names and Identifiers
| Name | S-triazine (1,3,5) |
| Synonyms | Triazine 1,3,5-Triazine S-triazine (1,3,5) 4-(2-methylphenyl)piperidine |
| CAS | 290-87-9 |
| EINECS | 206-028-1 |
| InChI | InChI:1S/C3H3N3/c1-4-2-6-3-5-1/h1-3H |
1,3,5-Triazine - Physico-chemical Properties
| Molecular Formula | C12H17N |
| Molar Mass | 175.27008 |
| Density | 0.958g/cm3 |
| Melting Point | 77-83℃ (dec.) |
| Boling Point | 282.2°C at 760 mmHg |
| Flash Point | 127.3°C |
| Vapor Presure | 0.00341mmHg at 25°C |
| Refractive Index | 1.52 |
| Use | The application of the triazine ring has three modifiable sites, which can react with the nucleophile to obtain a series of derivatives. At present, there are mainly 13 triazine herbicides on the market, atrazine is at the top of the list with more than $0.5 billion in sales, and prozin, cyclo-azinone and oxazin are among the more than $billion in sales. Recently, the compound annual growth rate of the old products in the past 5 years was 6.4% ~ 8.6%; The compound annual growth rate of triazine fluoxamine in the past 5 years was more than 5.0%. Because of the market growth of these varieties, the market growth of triazine herbicides is still in the forefront of the global pesticide market. |
1,3,5-Triazine - Risk and Safety
| Hazard Symbols | Xn - Harmful |
| Risk Codes | R22 - Harmful if swallowed R37/38 - Irritating to respiratory system and skin. R41 - Risk of serious damage to eyes |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S39 - Wear eye / face protection. |
1,3,5-Triazine - Introduction
S-triazine (1,3,5) is an organic compound with the chemical formula C3H3N3. The following is a description of the nature, use, manufacture and safety information of S-triazine (1,3,5):
Nature:
- S-triazine (1,3,5) is a colorless crystalline solid that is soluble in water and most organic solvents.
-It has relatively high thermal stability and chemical inertness.
- S-triazine (1,3,5) is a weak base that reacts with acids to form salts.
Use:
- S-triazine (1,3,5) is widely used as an intermediate in the chemical industry and agriculture.
-It is a raw material for the synthesis of many important organic compounds, such as dyes, glyphosate (a common herbicide) and fire retardants.
- S-triazine (1,3,5) is also used to prepare high-performance polymers, metal surface treatment agents and batteries.
Method:
- S-triazine (1,3,5) is mainly prepared by the reaction of ammonium cyanide and ammonia carbonate. First, ammonium cyanide and ammonia carbonate are heated to a high temperature to form ammonium triazazide carbonyl. The triaza isoxazidocarbonyl ammonium is then converted to the S-triazine (1,3,5) by dehydrating cleavage.
Safety Information:
- S-triazine (1,3,5) has low toxicity and is a relatively safe compound.
-However, during handling and preparation, it is still necessary to pay attention to personal protective measures, such as wearing gloves and respiratory protection devices, to avoid contact and inhalation of the substance.
-in the storage process, it should be stored in a dry, well ventilated place, and with oxidants, acids and other substances stored separately to prevent dangerous accidents.
In General, S-triazine (1,3,5) has a wide range of uses, and when used and handled correctly, it has a low safety risk.
Nature:
- S-triazine (1,3,5) is a colorless crystalline solid that is soluble in water and most organic solvents.
-It has relatively high thermal stability and chemical inertness.
- S-triazine (1,3,5) is a weak base that reacts with acids to form salts.
Use:
- S-triazine (1,3,5) is widely used as an intermediate in the chemical industry and agriculture.
-It is a raw material for the synthesis of many important organic compounds, such as dyes, glyphosate (a common herbicide) and fire retardants.
- S-triazine (1,3,5) is also used to prepare high-performance polymers, metal surface treatment agents and batteries.
Method:
- S-triazine (1,3,5) is mainly prepared by the reaction of ammonium cyanide and ammonia carbonate. First, ammonium cyanide and ammonia carbonate are heated to a high temperature to form ammonium triazazide carbonyl. The triaza isoxazidocarbonyl ammonium is then converted to the S-triazine (1,3,5) by dehydrating cleavage.
Safety Information:
- S-triazine (1,3,5) has low toxicity and is a relatively safe compound.
-However, during handling and preparation, it is still necessary to pay attention to personal protective measures, such as wearing gloves and respiratory protection devices, to avoid contact and inhalation of the substance.
-in the storage process, it should be stored in a dry, well ventilated place, and with oxidants, acids and other substances stored separately to prevent dangerous accidents.
In General, S-triazine (1,3,5) has a wide range of uses, and when used and handled correctly, it has a low safety risk.
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: s-Triazine Visit Supplier Webpage Request for quotationCAS: 290-87-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 1,3,5-triazine-2,4,6-trithiol triso. S. sol. Request for quotation
CAS: 290-87-9
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 290-87-9
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: 1,3,5-Triazine Visit Supplier Webpage Request for quotationCAS: 290-87-9
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: 1,3,5-Triazine Request for quotationCAS: 290-87-9
Tel: 15335994747
Email: 3521670276@qq.com
Mobile: 15335994747
QQ: 3521670276
Wechat: A2020071098
Product Name: 1,3,5-Triazine Request for quotation
CAS: 290-87-9
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 290-87-9
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: S-triazine (1,3,5) Request for quotation
CAS: 290-87-9
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 290-87-9
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
Spot supply
Product Name: s-Triazine Visit Supplier Webpage Request for quotationCAS: 290-87-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 1,3,5-triazine-2,4,6-trithiol triso. S. sol. Request for quotation
CAS: 290-87-9
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 290-87-9
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: 1,3,5-Triazine Visit Supplier Webpage Request for quotationCAS: 290-87-9
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: 1,3,5-Triazine Request for quotationCAS: 290-87-9
Tel: 15335994747
Email: 3521670276@qq.com
Mobile: 15335994747
QQ: 3521670276
Wechat: A2020071098
Product Name: 1,3,5-Triazine Request for quotation
CAS: 290-87-9
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 290-87-9
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: S-triazine (1,3,5) Request for quotation
CAS: 290-87-9
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 290-87-9
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
View History





