加拉明
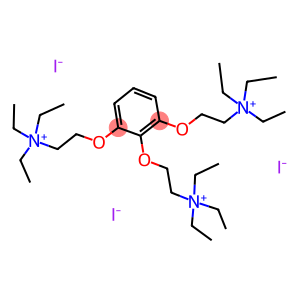
Gallamine triethiodide
CAS: 65-29-2
Molecular Formula: C30H60I3N3O3
加拉明 - Names and Identifiers
加拉明 - Physico-chemical Properties
| Molecular Formula | C30H60I3N3O3 |
| Molar Mass | 891.53 |
| Density | 1.4288 (estimate) |
| Melting Point | 235 °C (dec.) (lit.) |
| Boling Point | 502.6°C at 760 mmHg |
| Flash Point | 125.9°C |
| Solubility | DMSO 178 mg/mL Water 178 mg/mL Ethanol <1 mg/mL |
| Vapor Presure | 3.13E-10mmHg at 25°C |
| Appearance | powder |
| Color | Crystals from Me2CO (aq) |
| Merck | 13,4364 |
| Storage Condition | 2-8°C |
| Refractive Index | 1.501 |
| MDL | MFCD00011832 |
| Physical and Chemical Properties | White or milky white crystalline powder, odorless, slightly bitter, hygroscopic. Mp235 °c. Soluble in water (1:6), slightly soluble in ethanol (1:500), acetone, benzene and chloroform, insoluble in ether. LD50 (mouse, oral) 425mg/kg. |
加拉明 - Risk and Safety
| Hazard Symbols | Xn - Harmful |
| Risk Codes | R22 - Harmful if swallowed R36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36 - Wear suitable protective clothing. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) |
| WGK Germany | 3 |
| RTECS | BS1100000 |
| HS Code | 29239000 |
| Toxicity | LD50 oral in rabbit: 100mg/kg |
加拉明 - Reference
| Reference Show more | 1. Xu Shiliang, Wang Shizheng, Xu Rui, et al. Comparison of the effect of several kinds of attractant on snake feeding [J]. Feed industry, 2020, 26 (2):15-19. |
加拉明 - Introduction
Galaiodide ammonium
Last Update:2022-10-16 17:27:43
加拉明 - Reference Information
| Introduction | galanium iodide is a synthetic non-depolarizing muscle relaxant containing three quaternary ammonium groups. The muscle relaxant effect is similar to that of the Carine, but without blocking the ganglion and releasing histamine, but has a strong atropine-like effect, which can obviously relieve the tension of the vagus nerve and accelerate the heart rate, slightly elevated blood pressure and increased cardiac output. |
| preparation | Under the action of potassium hydroxide, pyrogallic acid is formed into sodium salt, and then reacted with diethylaminoethyl chloride and ethyl iodide in sequence. The synthesis reaction formula of the ammonium salt of the iodine is shown in the figure below: Fig. 1 The synthesis reaction formula of the ammonium salt of the salt |
| biological activity | Gallamine triethiode is a cholinergic receptor inhibitor with an IC50 of 68.0±8.4 μm. |
| Target | Value |
| AChR | 68.0 μM |
| Use | biochemical research, with muscle relaxation effect. |
| production method | Under the action of sodium acetate, pyrogallic acid is formed into sodium salt, and then reacted with diethylaminoethyl chloride and ethyl iodide in sequence. |
| toxic substance data | information provided by: pubchem.ncbi.nlm.nih.gov (external link) |
Last Update:2024-04-09 02:00:10
Supplier List
Spot supply
Product Name: Phloroglucinol Impurity 208 Visit Supplier Webpage Request for quotationCAS: 65-29-2
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Gallamine Triethiodide Visit Supplier Webpage Request for quotationCAS: 65-29-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Gallamine triethiodide Request for quotation
CAS: 65-29-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 65-29-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: Gallamine triethiodide Visit Supplier Webpage Request for quotationCAS: 65-29-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Phloroglucinol Impurity 208 Visit Supplier Webpage Request for quotationCAS: 65-29-2
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Gallamine Triethiodide Visit Supplier Webpage Request for quotationCAS: 65-29-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Gallamine triethiodide Request for quotation
CAS: 65-29-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 65-29-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: Gallamine triethiodide Visit Supplier Webpage Request for quotationCAS: 65-29-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



