亚胺唑
Imibenconazole
CAS: 86598-92-7
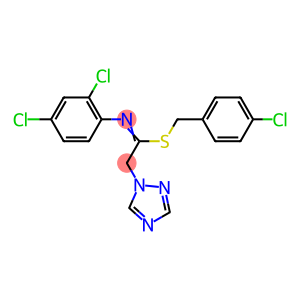
Molecular Formula: C17H13Cl3N4S
亚胺唑 - Names and Identifiers
亚胺唑 - Physico-chemical Properties
| Molecular Formula | C17H13Cl3N4S |
| Molar Mass | 411.74 |
| Density | 1.42±0.1 g/cm3(Predicted) |
| Melting Point | 89.5-90° |
| Boling Point | 567.4±60.0 °C(Predicted) |
| Flash Point | 4℃ |
| Water Solubility | 1.7 mg l-1 (20 °C) |
| Vapor Presure | 8.5 x l0-8 Pa (25 °C) |
| pKa | 1.81±0.10(Predicted) |
| Storage Condition | 0-6°C |
| Physical and Chemical Properties | Pure product is white crystal. M. p.89.5 ~ 90 ℃, vapor pressure 0.85 × 10-4Pa(25 ℃). Solubility: acetone 1063g/L, benzene 580g/L, xylene 250g/L, methanol 120g/L, water 1.79g/L. It is stable under weak alkaline conditions, stable to light, and unstable under strong acid and alkali. |
亚胺唑 - Risk and Safety
| Risk Codes | R11 - Highly Flammable R20/21/22 - Harmful by inhalation, in contact with skin and if swallowed. R36 - Irritating to the eyes |
| Safety Description | S16 - Keep away from sources of ignition. S36/37 - Wear suitable protective clothing and gloves. |
| UN IDs | UN 1648 3 / PGII |
| WGK Germany | 2 |
| RTECS | XZ4803030 |
| Toxicity | LD50 in male, female rats, male, female mice (mg/kg): 2800, 3000, >5000, >5000 orally; in male, female rats (mg/kg): >2000, >2000 dermally; in honey bees (mg/bee): >125 orally; LC50 (48 hr) in carp: 1.02 ppm; LC50 (6 hr) in water fleas: >102 ppm (Ogawa) |
亚胺唑 - Reference Information
| toxicity | Acute Oral LD502800mg/kg in male rats and 3000mg/kg in female rats; Mice (male and female) acute oral LD50>5000mg/kg. Rats (male and female) Acute percutaneous LD50>2000mg/kg. It has mild irritation to the eyes of rabbits. It has mild irritation to guinea pig skin and no sensitization to rabbit skin. Carp lc501.02 mg/L (48h), Daphnia lc50102 mg/L (6h). Oral LD50>25 μg/Bee. |
| Use | a novel triazole fungicide. Bactericidal spectrum is wide, with the absorption and resistance to rain, both therapeutic and preventive activity. It is effective by inhibiting the growth of germ tube and mycelium. For the prevention and control of apple scab, rust, powdery mildew, coal spot disease, coal spot disease, pear scab, rust, peach scab, and grape, orange, watermelon, peanut, tea, tobacco diseases. Spray with 115% -1000 times diluted with 4000 wettable powder. |
| production method | 1-(1,2, 4-triazol-1-ylacetoxy-2 ',4'-dichlorophenylimino)-1,2, 4-triazole and p-chlorobenzylmercaptan were refluxed in methyl isobutyl ketone for 5H, cooled after reaction, water and benzene were added, and the organic layer was separated, add 1mol/L NaOH, wash with water, dry with anhydrous sodium sulfate, and dissolve under reduced pressure to obtain iminazole. |
Last Update:2024-04-09 15:16:54
Supplier List
Spot supply
Product Name: Metconazole Impurity 2 (Imibenconazole) Visit Supplier Webpage Request for quotationCAS: 86598-92-7
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: IMIBENCONAZOLE Request for quotation
CAS: 86598-92-7
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 86598-92-7
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Spot supply
Product Name: Imibenconazole Visit Supplier Webpage Request for quotationCAS: 86598-92-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Metconazole Impurity 2 (Imibenconazole) Visit Supplier Webpage Request for quotationCAS: 86598-92-7
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: IMIBENCONAZOLE Request for quotation
CAS: 86598-92-7
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 86598-92-7
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Spot supply
Product Name: Imibenconazole Visit Supplier Webpage Request for quotationCAS: 86598-92-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


