乌拉地尔盐酸盐
Urapidil hydrochloride
CAS: 64887-14-5
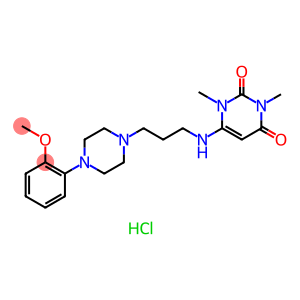
Molecular Formula: C20H30ClN5O3
乌拉地尔盐酸盐 - Names and Identifiers
| Name | Urapidil hydrochloride |
| Synonyms | URAPIDIL HCL URAPIDIL HYDROCHLORIDE Urapidil hydrochloride URAPIDIL HYDROCHLORIDE A1 ADRENOCEPTOR A NTAG 6[[3-[4-(O-METHOXYPHENYL)-1-PIPERAZINYL]PROPYL]AMINO]-1,3-DIMETHYLURACIL HYDROCHLORIDE 6-[[3-[4-(2-Methoxyphenyl)-1-piperazinyl]propyl]amino]-1,3-dimethyluracil hydrochloride 6-[[3-[4-(2-METHOXYPHENYL)-1-PIPERAZINYL]PROPYL]AMINO]-1,3-DIMETHYL-2,4(1H,3H)-PYRIMIDINEDIONE 6-[3-[4-(2-methoxyphenyl)piperazin-1-yl]propylamino]-1,3-dimethylpyrimidine-2,4-dione hydrochloride 6-[3-[4-(2-methoxyphenyl)piperazin-1-yl]propylamino]-1,3-dimethyl-pyrimidine-2,4-dione hydrochloride 6-[[3-[4-(2-METHOXYPHENYL)-1-PIPERAZINYL]PROPYL]AMINO]-1,3-DIMETHYL-2,4(1H,3H)-PYRIMIDINEDIONE HYDROCHLORIDE |
| CAS | 64887-14-5 |
| EINECS | 636-348-0 |
| InChI | InChI=1/C20H29N5O3.ClH/c1-22-18(15-19(26)23(2)20(22)27)21-9-6-10-24-11-13-25(14-12-24)16-7-4-5-8-17(16)28-3;/h4-5,7-8,15,21H,6,9-14H2,1-3H3;1H |
乌拉地尔盐酸盐 - Physico-chemical Properties
| Molecular Formula | C20H30ClN5O3 |
| Molar Mass | 423.94 |
| Melting Point | 156-1580C |
| Boling Point | 549°C at 760 mmHg |
| Flash Point | 285.8°C |
| Solubility | H2O: soluble |
| Vapor Presure | 4.22E-12mmHg at 25°C |
| Appearance | solid |
| Color | white |
| Storage Condition | Keep in dark place,Inert atmosphere,Room temperature |
乌拉地尔盐酸盐 - Risk and Safety
| Hazard Symbols | Xn - Harmful |
| Risk Codes | R22 - Harmful if swallowed R36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36 - Wear suitable protective clothing. |
| WGK Germany | 3 |
| RTECS | YQ9862000 |
乌拉地尔盐酸盐 - Preparation solution concentration reference
| 1mg | 5mg | 10mg | |
|---|---|---|---|
| 1 mM | 2.359 ml | 11.794 ml | 23.588 ml |
| 5 mM | 0.472 ml | 2.359 ml | 4.718 ml |
| 10 mM | 0.236 ml | 1.179 ml | 2.359 ml |
| 5 mM | 0.047 ml | 0.236 ml | 0.472 ml |
Last Update:2024-01-02 23:10:35
乌拉地尔盐酸盐 - Reference Information
| hypertension drug | urapidil hydrochloride is a selective α-receptor blocker, and its hypotensive effect has both central and peripheral mechanisms, but mainly outside the week dual mechanism. Peripherally, it blocks the postsynaptic Alpha 1 receptor, inhibits the vasoconstrictive action of catecholamines, thereby reducing peripheral vascular resistance and cardiac load; Centrally, by exciting the 5-hydroxytryptamine-1a receptor, regulates the activity of the circulatory center, prevents the increase of blood pressure and heart rate caused by sympathetic reflex. Dual-mechanism action: rapid depressurization, safe dual-channel metabolism. At present, urapidil has been consistently recommended by the authoritative guidelines at home and abroad, and is the first-line treatment for hypertensive emergencies. In 2018, the guidelines for the diagnosis and treatment of cardiovascular diseases during pregnancy issued by the European Society of Cardiology ESC, and in 2017, the expert consensus on the diagnosis and treatment of hypertension in emergency department in China issued by the emergency physicians of Chinese Medical Association, recommended urapidil for the treatment of acute aortic dissection, acute Cerebral ischemic stroke, acute cerebral hemorrhage, subarachnoid hemorrhage, hypertensive encephalopathy, acute heart failure, perioperative hypertension, pheochromocytoma, emergency stress hypertension, pediatric hypertensive emergency. |
| indication | urapidil hydrochloride is suitable for various types of hypertension, hypertensive crisis, severe and very severe hypertension, refractory hypertension, and control of perioperative hypertension, including patients with hypertension such as liver, renal insufficiency, coronary heart disease, diabetes and pulmonary hypertension caused by chronic obstructive pulmonary disease. Also has a role in prostate hypertrophy. |
| dosage | injection: 5ml:25mg;10ml:50mg. Tablets: 30mg:60mg. Sustained-release tablets: 30mg. Oral: Daily 30~180mg, 2 times, the general initial dose of 30mg each time, 2 times a day, after the appropriate adjustment according to the condition. Intravenous injection: the goods 12.5~25mg added to 10ml sodium chloride injection or glucose injection, intravenous injection, blood pressure changes, 5~10 minutes after the injection can be repeated if necessary 12.5~25mg. Intravenous drip: in order to maintain the efficacy or slow blood pressure, the product can be added to 25mg sodium chloride injection or glucose injection in intravenous drip, drip speed is generally 100~400g/min. Congestive heart failure, blood pressure increased significantly, first with the product 12.5~25mg plus 10ml sodium chloride injection or glucose injection intravenous injection. General situation can be directly intravenous drip, drop rate of 100~400g/min, according to the condition of the adjustment of dose and drop speed. Can be 1 times a day or 24 hours of continuous use, treatment is generally 3~6 days, depending on the condition can be longer than 6 days. |
| adverse reactions | 1. After the use of urapidil, individual cases may appear Head Pain, dizziness, Nausea, Vomit, sweating, irritability, Fatigue, palpitations, arrhythmia, upper facial pressure or Dyspnea and other symptoms, the reason is mostly caused by blood pressure drop too fast, usually in a few minutes can disappear, the patient does not need to stop. Excessive reduction of blood pressure, can raise the lower limbs, add blood volume can be improved. 2. Allergic reactions are rare (such as itching, skin redness, rash, etc.). 3. Platelet count decreased in very few cases when the drug was taken orally, but the causal relationship has not been confirmed by serum immunological studies. |
| note | 1) patients with stenosis of Aortic Isthmus or arterial and venous shunts (except dialysis shunts) injection was contraindicated. It is contraindicated for vasodilators, pregnant women and lactating women. 2) Head Pain, dizziness, Fatigue, palpitations, gastrointestinal reactions, allergic reactions, orthostatic hypotension and excessive blood pressure decline were observed. 3) can not be mixed with basic drugs, because of its acidic nature may cause solution turbidity or formation of flocs. 4) combination with angiotensin converting enzyme is not recommended. |
| Reference Preparation | T akeda's urapidil hydrochloride injection was approved for marketing in Austria: 27.35mg urapidil hydrochloride (equivalent to 25mg urapidil), 10ml:54.7mg urapidil hydrochloride (equivalent to 50mg urapidil). Subsequently, TAKEDA's urapidil injection has been approved in several EU countries (Germany, Switzerland, Poland, France, Spain, etc.). In 2012, CFDA approved the original urapidil hydrochloride injection of Takeda for marketing, Specification: 5ml:25mg, Origin is Austria. The reference for urapidil injection is Ebrantil(5ml:25mg, 10ml:50mg), which is listed in the European Union and imported by the original research and import of Takeda co., Ltd. |
| biological activity | Urapidil HCl is the hydrochloride salt form of Urapidil, an alpha1-adrenergic receptor antagonist and a 5-HT1A receptor agonist, the pIC50 was 6.13 and 6.4, respectively. |
| Target | Value |
| Use | for the treatment of various types of hypertension, hypertension |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: Urapidil HCl Visit Supplier Webpage Request for quotationCAS: 64887-14-5
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Urapidil hydrochloride Visit Supplier Webpage Request for quotationCAS: 64887-14-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Urapidil Hydrochloride Visit Supplier Webpage Request for quotation
CAS: 64887-14-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 64887-14-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Urapidil HCl Visit Supplier Webpage Request for quotationCAS: 64887-14-5
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Urapidil hydrochloride Visit Supplier Webpage Request for quotationCAS: 64887-14-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Urapidil Hydrochloride Visit Supplier Webpage Request for quotation
CAS: 64887-14-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 64887-14-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


