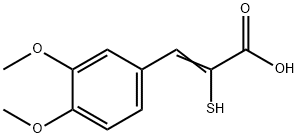
β-(3,4-Dimethoxy-phenyl)-α-mercapto-acrylsaeure
(2Z)-3-(3,4-dimethoxyphenyl)-2-sulfanylprop-2-enoic acid
CAS: 97852-94-3
Molecular Formula: C11H12O4S
β-(3,4-Dimethoxy-phenyl)-α-mercapto-acrylsaeure - Names and Identifiers
| Name | (2Z)-3-(3,4-dimethoxyphenyl)-2-sulfanylprop-2-enoic acid |
| Synonyms | AURORA 1235 AKOS BBS-00007608 3,4-DIMETHOXY-ALPHA-MERCAPTOCINNAMIC ACID β-(3,4-Dimethoxy-phenyl)-α-mercapto-acrylsaeure 2-Propenoic acid, 3-(3,4-dimethoxyphenyl)-2-mercapto- (2Z)-3-(3,4-dimethoxyphenyl)-2-sulfanylprop-2-enoic acid |
| CAS | 97852-94-3 |
| InChI | InChI=1/C11H12O4S/c1-14-8-4-3-7(5-9(8)15-2)6-10(16)11(12)13/h3-6,16H,1-2H3,(H,12,13)/b10-6- |
β-(3,4-Dimethoxy-phenyl)-α-mercapto-acrylsaeure - Physico-chemical Properties
| Molecular Formula | C11H12O4S |
| Molar Mass | 240.28 |
| Density | 1.285g/cm3 |
| Boling Point | 414.2°C at 760 mmHg |
| Flash Point | 204.3°C |
| Vapor Presure | 1.33E-07mmHg at 25°C |
| Refractive Index | 1.61 |
β-(3,4-Dimethoxy-phenyl)-α-mercapto-acrylsaeure - Introduction
(2Z)-3-(3,4-dimethoxyphenyl)-2-sulfanylprop-2-enoic acid is a chemical substance commonly abbreviated as DMTS. Its properties are as follows:
1. Appearance: DMTS is a light yellow solid.
2. Solubility: DMTS has good solubility in general organic solvents.
3. Melting point: The melting point of DMTS is about 75-80 degrees Celsius.
4. density: the density of DMTS is about 1.25 g/cm.
5. chemical formula:(2Z)-(2Z)-3-(3,4-dimethoxyphenyl)-2-sulfanylprop-2-enoic acid chemical formula is C12H14O4S, which contains a thiol group and two methoxy groups.
DMTS has several applications in the chemical and pharmaceutical fields:
1. Intermediate: DMTS can be used as an intermediate for the synthesis of other organic compounds.
2. Metal coordination compound: DMTS is a ligand that can form coordination compound with metal, which can be used to synthesize metal complexes.
3. drug research: due to the special nature of its thiol group, DMTS can be used as a thiol protecting group or a reagent to excite fluorescence in drug research, so as to design small molecule probes.
Method of preparing DMPT:
DMTS can be synthesized by a variety of methods, common preparation methods include:
1. By thiolation reaction: 3,4-dimethoxyphenylacetone is reacted with sodium thioacetate under alkaline conditions to produce DMTS.
2. Halogenation by Sulfonyl Reaction: 3,4-dimethoxyphenylacetone is reacted with sulfuryl chloride to produce DMTS.
Safety Information:
DMTS is less dangerous, but the following should be noted:
1. transport and storage to avoid contact with oxidants.
2. Wear appropriate personal protective equipment such as lab gloves and safety glasses during operation.
3. Avoid inhalation or contact with skin. If inhaled or exposed, wash or seek medical attention.
4. When using or handling DMPT, please follow the correct laboratory procedures and safety operation guidelines.
Please note that the above information is for reference only. When using and handling chemicals, it is important to follow relevant safety procedures and guidelines.
1. Appearance: DMTS is a light yellow solid.
2. Solubility: DMTS has good solubility in general organic solvents.
3. Melting point: The melting point of DMTS is about 75-80 degrees Celsius.
4. density: the density of DMTS is about 1.25 g/cm.
5. chemical formula:(2Z)-(2Z)-3-(3,4-dimethoxyphenyl)-2-sulfanylprop-2-enoic acid chemical formula is C12H14O4S, which contains a thiol group and two methoxy groups.
DMTS has several applications in the chemical and pharmaceutical fields:
1. Intermediate: DMTS can be used as an intermediate for the synthesis of other organic compounds.
2. Metal coordination compound: DMTS is a ligand that can form coordination compound with metal, which can be used to synthesize metal complexes.
3. drug research: due to the special nature of its thiol group, DMTS can be used as a thiol protecting group or a reagent to excite fluorescence in drug research, so as to design small molecule probes.
Method of preparing DMPT:
DMTS can be synthesized by a variety of methods, common preparation methods include:
1. By thiolation reaction: 3,4-dimethoxyphenylacetone is reacted with sodium thioacetate under alkaline conditions to produce DMTS.
2. Halogenation by Sulfonyl Reaction: 3,4-dimethoxyphenylacetone is reacted with sulfuryl chloride to produce DMTS.
Safety Information:
DMTS is less dangerous, but the following should be noted:
1. transport and storage to avoid contact with oxidants.
2. Wear appropriate personal protective equipment such as lab gloves and safety glasses during operation.
3. Avoid inhalation or contact with skin. If inhaled or exposed, wash or seek medical attention.
4. When using or handling DMPT, please follow the correct laboratory procedures and safety operation guidelines.
Please note that the above information is for reference only. When using and handling chemicals, it is important to follow relevant safety procedures and guidelines.
Last Update:2024-04-09 18:58:34