tetrafluorostannane
TIN(IV) FLUORIDE
CAS: 7783-62-2
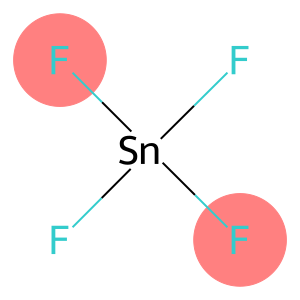
Molecular Formula: F4Sn
tetrafluorostannane - Names and Identifiers
| Name | TIN(IV) FLUORIDE |
| Synonyms | TIN FLUORIDE TIN(IV) FLUORIDE STANNIC FLUORIDE tetrafluorostannane STANNIC TETRAFLUORIDE |
| CAS | 7783-62-2 |
| EINECS | 232-016-0 |
tetrafluorostannane - Physico-chemical Properties
| Molecular Formula | F4Sn |
| Molar Mass | 194.7 |
| Density | 4,78 g/cm3 |
| Melting Point | 705°C (subl.) |
| Boling Point | 705°C |
| Water Solubility | hydrolyzes in H2O [MER06] |
| Appearance | Powder |
| Specific Gravity | 4.78 |
| Color | white |
| Exposure Limit | ACGIH: TWA 2 mg/m3NIOSH: IDLH 100 mg/m3; TWA 2 mg/m3 |
| Merck | 14,8776 |
| Storage Condition | Room Temprature |
| Sensitive | Moisture Sensitive |
tetrafluorostannane - Risk and Safety
| Risk Codes | 20/21/22 - Harmful by inhalation, in contact with skin and if swallowed. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S27 - Take off immediately all contaminated clothing. S36 - Wear suitable protective clothing. S36/39 - |
| UN IDs | UN2923 |
| WGK Germany | 3 |
| Hazard Note | Corrosive |
| Hazard Class | 8 |
| Packing Group | II |
tetrafluorostannane - References
| Overview | STANNIC FLUORIDE is a hygroscopic white monoclinic crystal. Very soluble in water, at room temperature, slow hydrolysis, heating that is rapid hydrolysis. Preparation Method: obtained by reacting SnO with elemental F2 at 500 °c. |
| Application | the application of STANNIC FLUORIDE is exemplified as follows: 1) a low-temperature hydrogen peroxide activator is prepared. 2) preparation of a modified graphene composite lithium ion battery anode material, 3) preparation of mesoporous HAP/zirconia/lithium oxide artificial bone ceramic. |
| physical properties | Tin tetrafluoride is a hygroscopic white monoclinic crystal. Very soluble in water, at room temperature, slow hydrolysis, heating that is rapid hydrolysis. |
| preparation | Tin tetrafluoride can be obtained by reacting SnO with elemental F2 at 500 °c. |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: Tin(IV) fluoride Visit Supplier Webpage Request for quotationCAS: 7783-62-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: TIN(IV) FLUORIDE Visit Supplier Webpage Request for quotation
CAS: 7783-62-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 7783-62-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Tin(IV) fluoride Visit Supplier Webpage Request for quotationCAS: 7783-62-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: TIN(IV) FLUORIDE Visit Supplier Webpage Request for quotation
CAS: 7783-62-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 7783-62-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History

