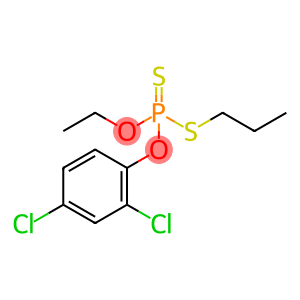
o-ethyl-o-(2,4-dichlorophenyl)-s-n-propyl-dithiophosphate
Prothiofos
CAS: 34643-46-4
Molecular Formula: C11H15Cl2O2PS2
o-ethyl-o-(2,4-dichlorophenyl)-s-n-propyl-dithiophosphate - Names and Identifiers
o-ethyl-o-(2,4-dichlorophenyl)-s-n-propyl-dithiophosphate - Physico-chemical Properties
| Molecular Formula | C11H15Cl2O2PS2 |
| Molar Mass | 345.25 |
| Density | 1.3000 |
| Melting Point | 25°C |
| Boling Point | 126.5 °C |
| Water Solubility | 0.07 mg l-1(20 °C) |
| Vapor Presure | 3.0×10-4 Pa (20 °C) |
| Appearance | liquid |
| BRN | 1998314 |
| Storage Condition | APPROX 4°C |
| Physical and Chemical Properties | Pure product is colorless liquid. B. p.164 ~ 167 ℃/24Pa, vapor pressure <9.999 × 10-4Pa (20 ℃), relative density 1.293 (20 ℃), refractive index n20D1.5680. It is soluble in toluene, cyclohexanone and other organic solvents, and has a solubility of 1.7mg/L in water at 20 ℃. The half-life in isopropanol/water (1:1) mixture is 26h when the pH value is 11.4 at 37 ℃ and 160d when the pH value is 2. |
o-ethyl-o-(2,4-dichlorophenyl)-s-n-propyl-dithiophosphate - Risk and Safety
| Risk Codes | R22 - Harmful if swallowed R50/53 - Very toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment. |
| Safety Description | S60 - This material and its container must be disposed of as hazardous waste. S61 - Avoid release to the environment. Refer to special instructions / safety data sheets. |
| UN IDs | UN 3082 |
| WGK Germany | 3 |
| RTECS | TD5680000 |
o-ethyl-o-(2,4-dichlorophenyl)-s-n-propyl-dithiophosphate - Reference Information
| NIST chemical information | information provided by: webbook.nist.gov (external link) |
| EPA chemical substance information | information provided by: ofmpeb.epa.gov (external link) |
| toxicity | The acute oral LD50 of male rats was 1730mg/kg as determined by Tokyo Dental College, female rats were 1800mg/kg, male mice were 940mg/kg, female mice were 960mg/kg, male rabbits were 750mg/kg, male cats were> 500mg/kg. Acute percutaneous LD50 was 4170mg/kg in male rats, 4100mg/kg in female rats, 1650mg/kg in male mice, and 1600mg/kg in female mice. Subcutaneous injection of LD50 rats> 3000mg/kg, mice 1250 ~ 1550mg/kg. The Elberfeld toxicology study determined that the acute oral LD50 of male rats was 0.25 mg/kg, female rats were 947mg/kg, male mice were 1036mg/kg, and female mice were 1051mg/kg. The LC50 of carp was 9.5mg/L (48h), and the LC50 of Daphnia was 0.13mg/L (3H). The oral LD50 was 100 ~ 200mg/kg for canines and 200mg/kg for quails. The LD50 of honey bee was 3.4 μg. |
| Use | asymmetric organophosphorus pesticides, with a broad spectrum of biological activity, no absorption. It is highly effective for lepidopteran larvae, especially for the production of cross-resistant aphids, thrips, Veratrum, rolled leaf insects and worms, the multi-resistant strains of Musca domestica have good killing activity, the same high efficiency on mosquitoes and Nicotiana, effective on coleopteran pests, on the leaf cicadaceae, Miridae and arthroidae pests weak effect. It has obvious activity against the larval stage of underground pests, and is also promising for the control of important underground pests such as golden needle worm, ground tiger and termites. Emulsifiable concentrates are usually sprayed 1000 times on water, with a safe interval of 21d for vegetables and 45d for citrus. |
| production method | PrSP(O)Cl2 and powdered P2S5 were heated at 130 °c for 3H to obtain PrSP(S). Cl2. It was dissolved in benzene and treated with ethanol and triethylamine at 0-5 °c to give PrS(EtO)O(S)Cl at -167 °c. Then it is reacted with 2, 4-dichlorophenol and carbonic acid potassium in methyl ethyl ketone and refluxed for 3H to obtain propylsulfur and phosphorus. The reaction can also be carried out in a reactor equipped with a stirrer, a thermometer and a reflux condenser, and the phosphorus pentasulfide, 2, 4-dichlorophenol, catalyst and organic solvent are reacted at a lower temperature for 2-3H, then, acid binding agent and bromopropane were added, the temperature was raised and reacted for 2H. After completion of the reaction, the reaction was cooled and filtered, washed with water until neutral, and dissolved under reduced pressure to obtain propylenophos bromide. |
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: Prothiofos Request for quotation
CAS: 34643-46-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 34643-46-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Multiple Specifications
Product Name: Prothiofos Visit Supplier Webpage Request for quotationCAS: 34643-46-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: Prothiofos Request for quotation
CAS: 34643-46-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 34643-46-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Multiple Specifications
Product Name: Prothiofos Visit Supplier Webpage Request for quotationCAS: 34643-46-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History

