meadjohnson1999
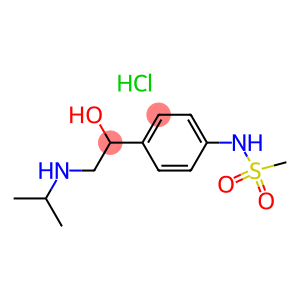
(+)-sotalol hydrochloride
CAS: 959-24-0
Molecular Formula: C12H21ClN2O3S
meadjohnson1999 - Names and Identifiers
| Name | (+)-sotalol hydrochloride |
| Synonyms | SOTALEX SOTACOR Sotalol HCl meadjohnson1999 Sotalol Hydrochloride (+)-sotalol hydrochloride isopropylaminohydroxyethylmethanesulfonanilidehydrochloride 4-(2-isopropylamino-1-hydroxyaethyl)methanesulfonailidhydrochlorid 4-(1-hydroxy-2-isopropylaminoethyl)methanesulphonanilide hydrochloride n-isopropyl-beta-(4-methanesulfonamidophenyl)ethanolaminehydrochloride 4'-(1-hydroxy-2-(isopropylamino)ethyl)-methanesulfonanilidmonohydrochlorid 4'-(1-hydroxy-2-(isopropylamino)ethyl)methanesulfonanilidemonohydrochloride |
| CAS | 959-24-0 |
| EINECS | 213-496-0 |
| InChI | InChI=1/C12H20N2O3S.ClH/c1-9(2)13-8-12(15)10-4-6-11(7-5-10)14-18(3,16)17;/h4-7,9,12-15H,8H2,1-3H3;1H |
meadjohnson1999 - Physico-chemical Properties
| Molecular Formula | C12H21ClN2O3S |
| Molar Mass | 308.82 |
| Melting Point | 218-220°C |
| Water Solubility | Soluble in phosphate buffered saline, DMSO, ethanol, water, and methanol. |
| Solubility | H2O: 20mg/mL |
| Appearance | powder |
| Color | white to off-white |
| Storage Condition | 2-8°C |
| Use | Used as cardiovascular drugs, beta blockers |
| In vitro study | Sotalol hydrochloride (MJ 1999) is an adrenergic β-antagonist that is used in the treatment of life-threatening arrhythmias. Sotalol hydrochloride is a competitive beta adrenoceptor antagonist devoid of membrane-stabilizing activity and intrinsic sympathomimetic activity that has no preferential actions on beta 1 or beta 2 responses. Sotalol hydrochloride causes concentration-dependent increases in the contractility of isolated ventricular tissue that is not blocked by previous beta or alpha blockade or catecholamine depletion. Sotalol hydrochloride consistently reduces the heart rate to a greater degree than propranolol and causes significantly less cardiac suppression than propranolol at a given heart rate. Sotalol hydrochloride is not only a beta blocker but a class III antiarrhythmic drug. Its possible antifibrillatory activity was therefore investigated in both the ventricles and atria of dog heart in situ, since vulnerability to fibrillation is not the same in both these parts of the myocardium. |
meadjohnson1999 - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36 - Wear suitable protective clothing. |
| WGK Germany | 3 |
| RTECS | PB0826000 |
| HS Code | 2935904000 |
| Toxicity | LD50 in male mice, rats (mg/kg): 2600, 3450 orally; 670, 680 i.p.; LD50 orally in rabbits: 1000 mg/kg; LD50 i.p. in dogs: 330 mg/kg (Lish) |
meadjohnson1999 - Standard
Authoritative Data Verified Data
This product is 4 '-(l-hydroxy-2-isopropylaminoethyl) methanesulfonylaniline hydrochloride. The content of C12H20N203S • HC1 shall be between 98.5% and 101.5% calculated on the dry product.
meadjohnson1999 - Trait
Authoritative Data Verified Data
- This product is white or off-white crystalline powder; Odorless.
- This product is soluble in water or methanol, soluble in ethanol, almost insoluble in chloroform.
absorption coefficient
take this product, precision weighing, add 0.1 mol/L sodium hydroxide solution is dissolved and quantitatively diluted to prepare a solution containing about 10ug per 1 ml, and the absorbance is measured at the wavelength of 249nm by ultraviolet-visible spectrophotometry (General rule 0401), the absorption coefficient is 456 to 504.
meadjohnson1999 - Category
Authoritative Data Verified Data
- in the chromatogram recorded under the content determination item, the retention time of the main peak of the test solution should be consistent with the retention time of the main peak of the reference solution.
- the solution under the term of absorption coefficient was taken and measured by ultraviolet-visible spectrophotometry (General rule 0401), and the maximum absorption was found at a wavelength of Nm.
- The infrared absorption spectrum of this product should be consistent with that of the control (Spectrum set 1199).
- the aqueous solution of this product was chloride identification (1) of the reaction (General 0301).
meadjohnson1999 - Exam
Authoritative Data Verified Data
acidity
take 0.4g of this product, add 20ml of water to dissolve, and then measure it according to law (General rule 0631). The pH value should be 4.5~6.0.
clarity and color of solution
take this product l. After adding 10ml of water to dissolve, the solution should be clear and colorless; If it is turbid, it should not be more concentrated compared with No. 1 turbidity standard solution (General rule 0902 first method); If it is colored, it shall not be deeper in comparison with the yellow No. 1 Standard Colorimetric solution (General rule 0901 first method).
sulfate
take 2.0g of this product and check it according to law (General rule 0802). Compared with the control solution made of 0.01% of standard potassium sulfate solution, it should not be more concentrated ().
Related substances
take this product, add the dilution solvent under the item of content determination to dissolve and dilute to make a solution containing about 2mg per 1 ml, as the test solution; Take the appropriate amount of precision, A solution containing 10ug per 1 ml was prepared as a control solution by quantitative dilution with a dilution solvent. According to the chromatographic conditions under the content determination item, 20 u1 of the test solution and the control solution are respectively injected into the liquid chromatograph, and the chromatogram is recorded to 3 times of the retention time of the main component peak. If there are impurity peaks in the chromatogram of the test solution, the area of a single impurity peak shall not be greater than 0.6 times (0.3%) of the area of the main peak of the control solution; the sum of each impurity peak area shall not be greater than the main peak area of the control solution (0.5%).
loss on drying
take this product, dry to constant weight at 105°C, weight loss shall not exceed 0.5% (General rule 0831).
ignition residue
take l.Og of this product and check it according to law (General rule 0841). The residue left shall not exceed 0.1%.
Heavy metals
The residue left under the item of taking the ignition residue shall not contain more than 10 parts per million of heavy metal when examined by law (General Principles 0821, Law II).
Iron Salt
take l.Og of this product, add 25ml of water to dissolve, and then check according to law (General rule 0807). Compared with the control solution made of 0.001% of standard iron solution, it shall not be deeper ().
meadjohnson1999 - Content determination
Authoritative Data Verified Data
measured by high performance liquid chromatography (General 0512).
chromatographic conditions and system suitability test
silica gel bonded with octanoalkyl silane was used as a filler; 0.2% sodium octane sulfonate solution (adjusted to pH 3.0 with phosphoric acid)-acetonitrile (79:21) was used as a mobile phase; The detection wavelength was 228mn. The number of theoretical plates shall not be less than 2000 calculated by sotalol peak, and the separation degree between sotalol peak and adjacent impurity peaks shall meet the requirements.
assay
take an appropriate amount of this product, weigh it accurately, add Diluted solvent [water (adjust pH to 3.0 with phosphoric acid)-acetonitrile (79:21)] dissolve and quantitatively dilute to make about 0.lmg solution was used as the test solution, and 20 u1 was accurately measured. Human liquid chromatograph was used to record the chromatogram. According to the external standard method to calculate the peak area, that is.
meadjohnson1999 - Category
Authoritative Data Verified Data
B adrenergic receptor antagonists.
meadjohnson1999 - Storage
Authoritative Data Verified Data
light-shielded, sealed, and preserved in a dry place.
meadjohnson1999 - Sotalol Hydrochloride Tablets
Authoritative Data Verified Data
This product contains Sotalol Hydrochloride (C12H20N203S • HCI) should be 95.0% ~ 105.0% of the label amount.
trait
This product is white or off-white.
identification
- Take appropriate amount of fine powder of this product (equivalent to 250mg of Sotalol Hydrochloride), put it in a 50ml measuring flask, and shake with 25ml of methanol for 10 minutes to dissolve Sotalol Hydrochloride, dilute to the scale with methanol, filter, take the filtrate as a test solution; Take an appropriate amount of Sotalol Hydrochloride control, dissolve and dilute with methanol to make a solution containing 2mg per 1 ml, according to the thin layer chromatography (General 0502) test, take the above two solutions of 10 u1, respectively, on the same silica gel GF254 thin layer plate, with methanol-chloroform (3:7) as the developing solvent, after deployment, dry, put the UV light (254nm) under the view. The position and color of the main spot displayed by the test solution should be the same as that of the reference solution.
- in the chromatogram recorded under the content determination item, the retention time of the main peak of the test solution should be consistent with the retention time of the main peak of the reference solution.
- the solution of the test article under the item of dissolution was taken and measured by ultraviolet-visible spectrophotometry (General rule 0401), and there was a maximum absorption at a wavelength of MN.
- two items (1) and (2) above can be selected as one item.
examination
- Related substances take an appropriate amount of fine powder of this product (about 50mg equivalent to Sotalol Hydrochloride) and place it in a 25ml measuring flask, add the diluted solvent under the content determination item to dissolve Sotalol Hydrochloride and dilute to the scale, shake well, filter, and take the filtrate as the test solution, A solution containing 10ug of Sotalol Hydrochloride per 1 ml was prepared as a control solution by quantitative dilution with a dilution solvent. If there are impurity peaks in the chromatogram of the test solution, the single impurity peak area shall not be more than 0.6 times (0.3%) of the main peak area of the control solution; the sum of each impurity peak area shall not be greater than the main peak area of the control solution (0.5%).
- the dissolution of this product, according to the dissolution and release determination method (General rule 0931 second method), water 900ml as the dissolution medium, the speed of 50 rpm, according to the law, after 30 minutes, the solution is filtered, the appropriate amount of the filtrate is taken out with precision, and the appropriate amount of the filtrate is diluted quantitatively with water to prepare a solution containing about 10ug of Sotalol Hydrochloride per 1 ml, according to UV-visible spectrophotometry (General rule 0401), measure absorbance at the wavelength of 228nm; Take the Sotalol Hydrochloride reference substance, water was added to dissolve and quantitatively diluted to prepare a solution containing about 10ug per 1 ml, which was determined by the same method, and the dissolution amount of each tablet was calculated. The limit is 80% of the labeled amount and shall be in accordance with the provisions.
- others shall be in accordance with the relevant provisions under the item of tablets (General rule 0101).
Content determination
Take 20 tablets of this product, precise weighing, fine grinding, precise weighing appropriate amount (about equivalent to Sotalol Hydrochloride 50mg ), put it in 50ml measuring flask, add dilute solvent [water (adjust pH to 3.0 with phosphoric acid)-acetonitrile (79:21)] to dissolve Sotalol Hydrochloride and dilute to the scale, shake, filter, precision take the appropriate amount of filtrate, diluted with a quantitative dilution of solvent made per lml containing about 0. The 1 mg solution was obtained as a test solution by measurement according to the method described in the item of Sotalol Hydrochloride content measurement.
category
Same as Sotalol Hydrochloride.
specification
80mg
storage
light shielding, sealed storage.
Supplier List
CAS: 959-24-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 959-24-0
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 959-24-0
Tel: +86-571-88162785
Email: Lucy@verypharm.com
Mobile: +86-13606544505
QQ: 472954302
Product List: View Catalog
CAS: 959-24-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 959-24-0
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 959-24-0
Tel: +86-571-88162785
Email: Lucy@verypharm.com
Mobile: +86-13606544505
QQ: 472954302
Product List: View Catalog


