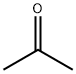
ketone,dimethyl
Acetone
CAS: 67-64-1
Molecular Formula: C3H6O
ketone,dimethyl - Names and Identifiers
| Name | Acetone |
| Synonyms | Acetone (CH3)2CO Propanone 2-Propanon 2-Propanone -Ketopropane ketonepropane dimethylketal Methyl ketone ketone propane GRAM STAIN NO 3 Dimethyl ketone pyroacetic acid chevron acetone ketone,dimethyl ACETONE ALCOHOL pyroacetic ether beta-ketopropane GRAMS DECOLORIZER |
| CAS | 67-64-1 |
| EINECS | 200-662-2 |
| InChI | InChI=1/C3H6O/c1-3(2)4/h1-2H3 |
ketone,dimethyl - Physico-chemical Properties
| Molecular Formula | C3H6O |
| Molar Mass | 58.08 |
| Density | 0.791g/mLat 25°C(lit.) |
| Melting Point | -94°C(lit.) |
| Boling Point | 56°C760mm Hg(lit.) |
| Flash Point | 1°F |
| JECFA Number | 139 |
| Water Solubility | soluble |
| Solubility | Miscible with water and with ethanol (96 per cent). |
| Vapor Presure | 184 mm Hg ( 20 °C) |
| Vapor Density | 2 (vs air) |
| Appearance | Liquid |
| Specific Gravity | 0.79 (25/25℃) |
| Color | Colorless, invisible vapor |
| Odor | Characteristic pungent odor detectable at 33 to 700 ppm (mean = 130 ppm) |
| Exposure Limit | TLV-TWA 1780 mg/m3 (750 ppm), STEL2375 mg/m3 (ACGIH); 10 h–TWA 590mg/m3 (250 ppm); IDLH 20,000 ppm(NIOSH). |
| Merck | 14,66 |
| BRN | 63580 |
| pKa | 19.3(at 25℃) |
| PH | 5-6 (395g/l, H2O, 20°C) |
| Storage Condition | Store at +5°C to +30°C. |
| Explosive Limit | 2.6-12.8%(V) |
| Refractive Index | n20/D 1.359(lit.) |
| Physical and Chemical Properties | Appearance and properties: colorless, transparent and easy to flow liquid, aromatic odor, volatile. melting point (℃): -94.6 boiling point (℃): 56.5 relative density (water = 1): 0.80 relative vapor density (Air = 1): 2.00 saturated vapor pressure (kPa): 53.32(39.5 ℃) heat of combustion (kJ/mol): 1788.7 critical temperature (℃): 235.5 critical pressure (MPa): 4.72 logarithm of octanol/water partition coefficient: -0.24 flash point (℃): -20 ignition temperature (℃): 465 Upper Explosive limit%(V/V): 2.5 lower explosive limit%(V/V): 13.0 solubility: miscible with water, miscible with ethanol, ether, chloroform, oil, most organic solvents such as hydrocarbons |
| Use | Is the basic organic raw materials and low boiling point solvents. It is an important raw material for the manufacture of acetic anhydride, chloroform, organic glass, epoxy resin, polyisoprene rubber and the like, and is also used as a solvent and an extracting agent. |
ketone,dimethyl - Risk and Safety
| Risk Codes | R11 - Highly Flammable R36 - Irritating to the eyes R66 - Repeated exposure may cause skin dryness or cracking R67 - Vapors may cause drowsiness and dizziness R39/23/24/25 - R23/24/25 - Toxic by inhalation, in contact with skin and if swallowed. |
| Safety Description | S9 - Keep container in a well-ventilated place. S16 - Keep away from sources of ignition. S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) S36/37 - Wear suitable protective clothing and gloves. S7 - Keep container tightly closed. |
| UN IDs | UN 1090 3/PG 2 |
| WGK Germany | 3 |
| RTECS | AL3150000 |
| FLUKA BRAND F CODES | 3-10 |
| TSCA | Yes |
| HS Code | 29141100 |
| Hazard Class | 3 |
| Packing Group | II |
| Toxicity | LD50 in rats: 10.7 ml/kg orally (Smyth) |
ketone,dimethyl - Upstream Downstream Industry
| Raw Materials | Benzene Propylene Cumene |
| Downstream Products | acetylene 2-Furaldehyde Acetone cyanohydrin 2-Methyl-2-butanol malonic acid 2,4-Pentanedione diketene Benzalacetone 4,4'-Isopropylidenediphenol |
ketone,dimethyl - Nature
Open Data Verified Data
colorless transparent liquid with special odor. Soluble in water and methanol, ethanol, ether, chloroform, pyridine and other organic solvents. Flammable, volatile, more active chemical properties. The vapor can be mixed with air to form an explosive mixture, and the explosion limit is 2. ss%~ 12.8% (volume). The carbonyl groups of acetone can react with a variety of nucleophiles. It can react with ammonia derivatives, hydrocyanic acid, alkynes and organometallic compounds. Acetone also allows the reaction of a hydrogen. Steam and air can form an explosive mixture, in the case of open flame, high heat, easy to burn explosion. A strong reaction with an oxidizing agent can occur.
ketone,dimethyl - Preparation Method
Open Data Verified Data
Technical grade acetone was dehydrated with anhydrous copper sulfate, potassium carbonate or 4A type molecular sieve, and then subjected to rectification to obtain reagent acetone.
ketone,dimethyl - Use
Open Data Verified Data
Common analytical reagents and solvents. Chromatographic analysis of standard substances.
ketone,dimethyl - Safety
Open Data Verified Data
LD50 rat oral: 10.7 ml/kg. In case of high fever, the internal pressure of the container increases, and there is a risk of cracking and explosion. Mixing with oxidant, reductant, alkali and edible chemicals is strictly prohibited. Flush with plenty of water. Store in a cool, ventilated warehouse. Keep away from fire and heat source. The bin temperature should not exceed 30 ℃. Protection from direct sunlight. Keep the container sealed. Should be stored separately from the oxidant.
ketone,dimethyl - Toxicity and use limits
toxicity
- ADI GMP is limited (FAO/WHO,2001).
- ld503000 mg/kg (mouse, oral).
- The acute lethal dose to a human is 50ml/human.
usage limit
- FDA & sect;173.210(2000): the limit in the spice oleoresin is 30mg/kg.
- FEMA(mg,/kg): Soft drink 5; Cold drink 5; Candy 8; Baked food 8; Pudding 5; Smear food 5.
maximum allowable amount of food additive
The Chinese name of the additive is allowed to use the Chinese name of the food additive function maximum allowable usage (g/kg) maximum allowable residue (g/kg)
- acetone food food flavors the maximum allowable amount and the maximum allowable residual amount of each flavor component used in the preparation of flavors in GB 2760 shall not exceed
- acetone food processing aids for food industry/processing aids for food industry should generally be made before the final product, except for the residues in food.
ketone,dimethyl - Reference Information
| FEMA | 3326 | ACETONE |
| relative polarity | 0.355 |
| olfactory Threshold | 42ppm |
| Henry's Law Constant | 2.27 at 14.9 °C, 3.03 at 25 °C, 7.69 at 35.1 °C, 11.76 at 44.9 °C (Betterton, 1991) |
| NIST chemical information | information provided by: webbook.nist.gov (external link) |
| EPA chemical substance information | information provided by: ofmpeb.epa.gov (external link) |
| Overview | acetone is also called dimethyl ketone, dimethyl ketone, dimethyl ketone, or acetic ketone, is the simplest ketone in the family of saturated fatty ketones. Melting Point -95 ° C, boiling point 56 ° C, density: 25 when the specific gravity of 0.788, a special odor of colorless flammable liquid. At room temperature for colorless transparent liquid, volatile, flammable, aromatic odor. With water, methanol, ethanol, ether, chloroform and pyridine can be miscible, can dissolve oil, fat, resin and rubber, can dissolve cellulose acetate and nitrocellulose, is an important solvent. Acetone on the human body has no special toxicity, but after inhalation can cause Head Pain, bronchitis and other symptoms. If inhaled in large quantities, there may be loss of consciousness. It is mainly used for degreasing, dehydration, fixation and so on in daily life. It is an important test object in blood and urine. Some cancer patients have abnormally elevated levels of acetone in their urine. People who lose weight with low carbohydrate diet therapy also have abnormally high concentrations of acetone in their blood and urine. Acetone exists in nature in a free state, mainly in essential oils in the plant kingdom, such as tea oil, Rosin essential oil, citrus essential oil, etc.; Human urine and blood and animal urine, small amounts of acetone are found in the tissues and body fluids of marine animals. The content of acetone in the urine of diabetic patients is abnormally increased. The vapor can be mixed with air to form an explosive mixture with an explosion limit of 2.55% to 12.8% by volume. The carbonyl group of acetone can undergo an addition reaction with a variety of nucleophiles, such as catalytic hydrogenation to generate isopropanol, reduction to generate cyano alcohol; Reaction with ammonia derivatives, hydrocyanic acid, alkynes, organometallic compounds, etc. Acetone can also carry out the reaction of alpha hydrogen, for example, substitution reaction with halogen, and similar aldol condensation reaction with itself or other compounds. |
| molecular structure | The C atom on the carbonyl group in the acetone molecule is bonded with sp2 hybrid orbitals, the methyl C atom is bonded to the sp3 hybrid orbital. |
| limit parameter | spontaneous ignition point: 465 ℃ explosion limit: 2.6% ~ 12.8% maximum explosion pressure: 87.3 N/cm2 most easy ignition concentration: 4.5 to produce the maximum explosion pressure concentration: 6.3% Minimum ignition energy: 1.15 MJ (when the concentration of 4.97%) combustion calorific value: 1792 kJ/mol (liquid, 25 °c) vapor pressure: 53.3 3千帕(39.5 °c). |
| purification and water removal methods | ordinary acetone often contains a small amount of water and methanol, acetaldehyde and other reducing impurities. The purification method includes: (1) adding 2.5g of potassium permanganate to 250ml of acetone for reflux. If the purple color of potassium permanganate disappears quickly, a small amount of potassium permanganate is added to continue reflux until the purple color does not fade. The acetone was then distilled off, dried over anhydrous potassium carbonate or anhydrous calcium sulfate, filtered and distilled to collect fractions at 55-56.5 °c. When purifying acetone by this method, it should be noted that the reducing substances in acetone can not be too much, otherwise it will consume too much potassium permanganate and acetone, so that the processing time increases. (2) fill 10% ml of acetone into a separatory funnel, add 4ml of 3.6 silver nitrate solution, then add ml of 1mol/L sodium hydroxide solution, shake for 10min, separate the acetone layer, further, anhydrous potassium sulfate or anhydrous calcium sulfate was added and dried. The 55-56.5 °c fraction was collected by final distillation. This method is faster than the method (1), but Silver nitrate is more expensive, should only do a small amount of purification. |
| function | in chemical industry, man-made fiber, medicine, paint, plastic, organic glass, cosmetics and other industries as an important organic raw material, is an excellent organic solvent, can dissolve many organic products such as resin, cellulose acetate, acetylene, etc., but also the manufacture of nitrocellulose solvent. In the industry, acetone is mainly used as a solvent for explosives, plastics, rubber, fiber, leather, grease, paint and other industries, acetone can also be used as synthetic ketene, acetic anhydride, iodoform, Polyisoprene Rubber, methacrylic acid, methyl ester, chloroform, epoxy resin and other important raw materials. In the precision copper tube manufacturing industry, acetone is often used to wipe the black ink on the copper tube. Common use examples: solvent: for example, the main (or only) component in the Polish water that removes nail polish is acetone. It is also a washing solvent commonly used in laboratories. Reagent: acetone is a C3 synthon in synthesis and can be used in organic synthesis. In addition, acetone is also a protective group precursor, which protects 1, 2-diol, or 1, 3-diol, by forming ketals. Refrigerant: a mixture of acetone and dry ice can be used as refrigerant (Celsius:-50 °c). |
| preparation method | acetone production methods mainly include isopropanol method, cumene method, fermentation method, acetylene hydration method and propylene direct oxidation method. At present, the industrial production of acetone in the world is mainly based on the cumene method. 2/3 of the world's acetone is a by-product of the preparation of phenol, is one of the products of the oxidation of cumene. At present, the main patent manufacturers of this technology are Kellogg Brown & Root company, three well Chemical Company and UOP company. Solutia has developed a technology for the oxidation of benzene with nitrogen oxides to produce phenol, but last year it canceled its plans to build plants using this process because the gross margin for using this technology is too low. Japanese researchers have also recently developed a one-step process for producing phenol and acetone from benzene using a europium-titanium catalyst. Dry distillation method: In The cumene method has not been invented before, early acetone from the dry distillation of calcium acetate. Fermentation: acetone can be obtained by fermentation with butanol yeast. This law was developed by Haim Weizmann during the First World War, but was soon abandoned because of the extremely low yield. Isopropylbenzene oxidation synthesis: Petroleum industry products isopropylbenzene in sulfuric acid catalyzed by air oxidation rearrangement into acetone, by-products of phenol. This method produces very little waste and is called the "one stone and two birds" method. |
| toxicity and harm | acetone is mainly on the inhibition of the central nervous system, exposure to high concentrations may cause liver, kidney and pancreas damage to individuals. Because of its low toxicity, rapid metabolism and detoxification, acute poisoning under production conditions is relatively rare. Acute poisoning can occur Vomit, shortness of breath, spasm or even Coma. After oral administration, the lips, throat burning sensation, after a few hours of incubation period can occur dry mouth, Vomit, lethargy, acid moderate and ketosis, and even temporary disturbance of consciousness. Acetone long-term damage to the human body, the performance of the eye irritation symptoms such as tears, photophobia and corneal epithelial infiltration, can also be manifested as dizziness, burning sensation, throat stimulation, Cough. Metabolism: acetone by the lung, gastrointestinal tract, skin and other ways to absorb, due to water-soluble, easy to absorb income blood, rapid distribution in the body. Its excretion depends on the dose, the large dose in the original form mainly through the lung and kidney, a very small amount of skin discharge; Small doses of most of the oxidation to carbon dioxide discharge. The biological half-life of acetone in blood was 5.3 h in rats, 11 h in dogs and 3 h in humans. The metabolism of acetone in the human body is mostly a tricarboxylic acid cycle intermediate that is decomposed into acetoacetic acid and converted into glycogen. 1, inhalation: concentration below 500ppm has no effect, between 500 ~ 1000ppm will stimulate the nose and throat, 1000ppm can cause Head Pain and dizziness. 2000~10000 ppm can produce dizziness, feeling drunk, drowsiness, Nausea and Vomit, high concentration leads to loss of consciousness, Coma and death. 2, eye contact: the concentration of 500ppm will produce stimulation, 1000ppm will have mild, temporary stimulation. Liquid can produce toxic irritation. 3, skin irritation: liquid will have mild irritation, the danger caused by intact skin absorption is very small. 4, oral: the throat and stomach have a stimulating effect, take a large number of will produce and inhalation of the same symptoms. 5, skin contact will lead to dryness, swelling and chapping, 3 hours a day inhalation concentration of 1000ppm vapor, in 7~15 years to stimulate the nasal cavity of workers, so that Vertigo, Fatigue. High concentrations of vapor can affect kidney and liver function. |
| first aid and protection | fire fighting: highly flammable, with serious fire hazard, belonging to class a fire hazard. The vapor forms an explosive mixture with air at room temperature. Use dry powder, foam extinguishing agent, halogen extinguishing agent or carbon dioxide to extinguishing. The vessel exposed to the fire was cooled with water and the acetone vapor was dissipated. Personal protection: 1, inhalation: If the vapor concentration is unknown or exceeds the exposure limit, should wear a suitable respirator. 2, the skin: if necessary, should use gloves, work clothes and work shoes, the appropriate material is butyl rubber. Safety shower and eye washing appliances should be available at the site of direct work. 3, eyes: wear chemical splash glasses, if necessary, wear a mask. First aid: 1, inhalation: separation from the acetone source or move the patient to fresh air, such as breathing should be artificial respiration. 2. Eye contact: Open the eyelids and rinse the affected eyes with warm and slow running water for about 10 minutes. 3. Skin Contact: Rinse the affected area with warm, slow running water for at least 10 minutes. 4, oral: Water Gargle, not emetic, to the patient about 250ml water. 5, all patients should ask a doctor for treatment. |
| content analysis | in a flask containing 20ml of water, approximately 1g of sample was accurately weighed and water was added to ML. 10ml of the liquid was put into a flask with a stopper, 25ml of sodium hydroxide solution (TS-224) was added, and the mixture was allowed to stand for 5min. Add 0.1mol/L iodine solution 25ml, add plug, in the cold dark place 10min, add 1mol/L sulfuric acid 30ml. The remaining iodine was titrated with 0.1mol/L sodium thiosulfate using the starch assay solution (TS-235) as an indicator. Make the same blank test according to the sample size and make the necessary correction. 0.1mol/L iodine is equivalent to 0.9675mg C3H6O per ml. |
| toxicity | ADI GMP (FAO/WHO,2001). LD503000mg/kg (mouse, oral). The acute lethal dose in humans is 50ml/person. |
| usage limit | FDA § 173.210(2000): the limit in the spice oleoresin is 30mg/kg. FEMA(mg,/kg): Soft drink 5; Cold drink 5; Candy 8; Baked goods 8; Pudding 5; Smear food 5. |
| Application | is an important raw material for organic synthesis, used in the production of epoxy resin, polycarbonate, organic glass, etc., is also a good solvent, and used as extractant, diluent, etc. used as analytical reagent and solvent is the basic organic raw materials and low boiling point solvent. It is an important raw material for the manufacture of acetic anhydride, chloroform, organic glass, epoxy resin, polyisoprene rubber and the like, and is also used as a solvent and an extracting agent. extraction solvent, mainly used for spices and cocoa beans. GB 2760-96 provides for food processing aids and permitted use of food spices. In Japan, the ingredients used for the extraction of coca beans are restricted for the manufacture of coca beverages, and it is stipulated that all foods produced with acetone-containing raw materials must be removed before the final food is completed; A pigment diluent may also be used. for the preparation of organic glass monomer, bisphenol A, diacetone alcohol, hexanediol, methyl isobutyl ketone, methyl isobutyl methanol, chlorone, isophorone, chloroform, iodoform and other important organic chemical raw materials. It is used as an excellent solvent in the coating, the spinning process of acetic acid fiber, the storage of acetylene in cylinder, and the dewaxing of oil refining industry. In the pharmaceutical industry, it is one of the raw materials for vitamin C and the anesthetic sofona, and is also used as an extractant in the production of various vitamins and hormones. In the pesticide industry, acetone is one of the raw materials for the synthesis of propylene pyrethroids. organic solvents are commonly used to selectively dissolve certain organic compounds. When acetone is present in the aqueous solution, the dissociation of the substances in the solution can be inhibited. It can be used for the Colorimetric Determination of cobalt and iron with cyanide. Barium and strontium were examined by color reaction. Microscopic analysis is used as a fixative, hardening and dehydration of tissues. Extraction of various components from plants and animals. |
| production methods | acetone production methods are more. The ancient method is to use lime and wood dry distillation of wood vinegar, made of calcium acetate, and then by thermal decomposition of acetone. The methods of synthesizing acetone have been studied in industry:(1) calcium acetate is obtained from acetic acid and then decomposed to acetone;(2) Acetylene reacts with water vapor on zinc oxide catalyst to produce acetone;(3) ethanol vapor in the presence of zinc chromate catalyst, high temperature reaction to generate acetone;(4) liquefied natural gas or naphtha oxidation to acetone (oxidation products also include Formaldehyde, Acetic Acid, butanol, etc.);(5) isopropyl alcohol oxidation or dehydrogenation to acetone;(6) isopropyl alcohol hydrogen peroxide to acetone;(7) isopropyl alcohol and acrolein synthesis of acetone;(8) isopropyl benzene to acetone, co-production of phenol with propylene and benzene as raw materials, cumene is prepared by alkylation, and then cumene hydroperoxide is obtained by air oxidation, and then decomposed with sulfuric acid or resin to obtain acetone and phenol at the same time;(9) the process route of direct oxidation of propylene to produce acetone is similar to that of direct oxidation of ethylene to produce acetaldehyde;(10) P-cresol is produced by P-methyl isopropylbenzene hydroperoxide method, and acetone is produced by-product;(11) hydroquinone is produced by diisopropylbenzene method, by-product acetone. However, there are not many methods actually used in the industry. At present, the production of acetone by grain fermentation still accounts for a large proportion in China. In the synthesis of cumene method is the main. A mixture of acetone, butanol and ethanol produced by fermentation of starch-containing agricultural and by-products. The ratio of the three is acetone: butanol = 32:56:12 to 25:70:3 (weight ratio). For each 1t of acetone produced, about 11t of starch or 60-66t of molasses was consumed. The cumene method is the most economical method in the acetone production route, and phenol is obtained at the same time. The ratio is Phenol: acetone = 1:0.6 by weight. Propylene (100,000) is consumed per ton of phenol in the 90% t unit based on phenol. The mixture of acetone and butanol was obtained by fermentation and then separated and refined. |
| category | flammable liquid |
| toxicity grade | poisoning |
| Acute toxicity | oral-rat LD50: 5800 mg/kg; Oral-mouse LD50: LD50:3000 mg/kg |
| stimulation data | eyes-rabbits 20 mg severe; Skin-rabbits 395 mg mild |
| explosive hazard characteristics | explosive when mixed with air |
| flammability hazard characteristics | in case of open flame, high temperature, flammable oxidant, combustion stimulus smoke |
| storage and transportation characteristics | The warehouse is ventilated and dried at low temperature; It is stored separately from the oxidant |
| extinguishing agent | dry powder, dry sand, carbon dioxide, foam, 1211 extinguishing agent |
| Occupational Standard | TWA 2400 mg/m3; Tel 4000 mg/m3 |
| spontaneous combustion temperature | 465°C |
| toxic substance data | information provided by: pubchem.ncbi.nlm.nih.gov (external link) |
| immediate life-and health-threatening concentrations | 2,500 ppm |