dyprosiumsesquioxide
Dysprosium Oxide
CAS: 1308-87-8
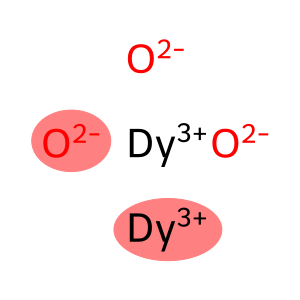
Molecular Formula: Dy2O3
dyprosiumsesquioxide - Names and Identifiers
| Name | Dysprosium Oxide |
| Synonyms | dyspmsia Dysprosia Dysprosium Oxide oxygen(-2) anion dysprosium(3+)oxide didysprosiumtrioxide dyprosiumsesquioxide dysprosium(+3) cation dysprosiumoxide(dy2o3) |
| CAS | 1308-87-8 |
| EINECS | 215-164-0 |
| InChI | InChI=1/2Dy.3O/rDy2O3/c3-1-5-2-4 |
| InChIKey | NLQFUUYNQFMIJW-UHFFFAOYSA-N |
dyprosiumsesquioxide - Physico-chemical Properties
| Molecular Formula | Dy2O3 |
| Molar Mass | 373 |
| Density | 7.81g/mLat 25°C(lit.) |
| Melting Point | 2330-2350 °C |
| Water Solubility | insoluble |
| Appearance | White powder |
| Specific Gravity | 7.81 |
| Color | White |
| Merck | 14,3482 |
| PH | 7.0 |
| Storage Condition | no restrictions. |
| Sensitive | Hygroscopic |
| MDL | MFCD00010986 |
| Physical and Chemical Properties | Properties white crystalline powder. melting point (2340±10)℃ relative density 7.81 solubility insoluble in water, soluble in acid and ethanol. |
| Use | Used as raw materials for the preparation of dysprosium metal, glass, NdFeB permanent magnet additives |
dyprosiumsesquioxide - Risk and Safety
| Safety Description | 24/25 - Avoid contact with skin and eyes. |
| WGK Germany | 2 |
| RTECS | JW1060000 |
| TSCA | Yes |
| HS Code | 2846 90 20 |
| Toxicity | LD50 orally in Rabbit: > 5000 mg/kg |
dyprosiumsesquioxide - Nature
Open Data Verified Data
white crystalline powder. The relative density was 7. 81. Melting point (2340 10) °c. Insoluble in water, soluble in acid and ethanol. Open air easily absorbs carbon dioxide and becomes part of dysprosium carbonate.
dyprosiumsesquioxide - Preparation Method
Open Data Verified Data
ignition method: dysprosium nitrate solution is reacted with sodium hydroxide to generate dysprosium hydroxide, which is then separated and burned to obtain dysprosium oxide.
dyprosiumsesquioxide - Use
Open Data Verified Data
as an additive element of yttrium iron and yttrium aluminum garnet, it can be used as a control material for nuclear reactor. It is used to manufacture a new type of light source Krypton lamp with high brightness and good light color. It can also be used as an anode coating due to its cathode emission capability. For the metal dysprosium raw materials, glass, NdFeB permanent magnet additive, magneto-optical memory materials.
dyprosiumsesquioxide - Safety
Open Data Verified Data
double-layer polyethylene plastic bag heat-sealed packaging, jacket carton protection. 5kg or lOkg net weight per bag (bottle). The bag is then placed in a plastic box (wooden box or iron drum). Should be stored in a ventilated, dry warehouse. During the storage and transportation process should pay attention to moisture and avoid package breakage. See cerium oxide products for toxicity and protection.
dyprosiumsesquioxide - Reference Information
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| introduction | dysprosium oxide is a white powder, slightly hygroscopic, and can absorb moisture and carbon dioxide in the air. Dysprosium oxide is an important rare earth material with a wide range of uses. In addition to the atomic energy industry as a control rod for nuclear reactors, it can also be used for metal halogen lamps, magneto-optical memory materials, glass, neodymium iron boron permanent magnet additives, etc. |
| use | dysprosium oxide is an important raw material for preparing metal dysprosium. dysprosium is a strategic metal with extremely important use and is an important component of infrared generator and laser material. It is an additive element for yttrium iron and yttrium aluminum garnet, which can be used as a control material for nuclear reactors. It is used to manufacture dysprosium lamps with new light sources with high brightness and good light color. Due to its cathode emission capability, it can also be used as an anode coating. Used as raw material for metal dysprosium, glass, NdFeB permanent magnet additive, magneto-optical memory material. It is used as a raw material for the preparation of metal dysprosium, glass, and additives for neodymium iron boron permanent magnets. It is also used in metal halogen lamps, magneto-optical memory materials, yttrium iron or yttrium aluminum garnet, and atomic energy industry. scientific research reagents, biochemical research used as raw materials for the preparation of metal dysprosium, glass, neodymium iron boron permanent magnet additives, can also be used in metal halogen lamps, magneto-optical memory materials, yttrium iron or yttrium aluminum garnet And atomic energy industry. |
| purification method | a method for extracting and purifying dysprosium oxide, comprising the following steps: 1. configuration of liquid to be purified: configuration of mixed solution of dysprosium oxide to be purified; 2. configuration of intermediate agent: configuration of retarder and eluent, wherein the retarder is Cu(NO3)2 · 3H2O, water and nitric acid, the eluent is composed of solid EDTA acid, water and ammonia water; 3. Preparation of the separation column: the separation column is used as a carrier for ion exchange; 4. Leaching column transformation: the separation column is transformed with the retarder in step 2; 5. Adsorption operation: The mixed solution of dysprosium oxide configured in step 1 performs adsorption operation with the separation column; 6. Leaching operation: the separation column in step 5 is rinsed by the rinse agent; 7, the liquid collected by the rinse agent in step 6 is collected in batches by volume, and the purity is analyzed by sampling, and the precipitation is concentrated and collected; 8, waste liquid treatment. |
| preparation | a preparation method of high purity dysprosium oxide, the specific steps are as follows: the first step is: 250g of dysprosium oxide raw material of 99.5% specification is put into 1500mL of deionized water with stirring device, 300mL of refined preparation with 5% mass concentration is slowly added under continuous stirring (electronic grade nitric acid is prepared into solution with deionized water), and the obtained mixture is stirred at room temperature, the speed is controlled at 1200rpm and the stirring time is 6h; The second step: transfer the mixture obtained in the first step to a container, and carry out ultrasonic irradiation oscillation, the ultrasonic frequency is 53KHz, the irradiation oscillation time is 8h, and the temperature is controlled at 30 ℃ ~ 50 ℃; Step 3: transfer the mixture obtained in the second step to a kettle with a stirring device, stir for 3 hours at room temperature, stir at 1800rpm, and filter under reduced pressure to obtain solid; step 4: The solid obtained in the third step is put into deionized water to wash until the liquid PH is neutral, the filter cake is pressure-dried, and the solid obtained in the third step is vacuum dried at 150 ℃ for more than 4 hours to obtain 232g of product. Trace metal analysis and detection by inductively coupled plasma emission spectrometer (ICP) shows that the purity is 99.991%, the total metal impurity content is less than 100ppm, and nitrogen protection package. |
| production method | the burning method reacts the dysprosium nitrate solution to generate dysprosium hydroxide through sodium hydroxide, and then separates and burns to obtain dysprosium oxide. Its 2Dy(OH)3 → Dy2O3 3H2O |
Supplier List
CAS: 1308-87-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 1308-87-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 1308-87-8
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 1308-87-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 1308-87-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 1308-87-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 1308-87-8
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 1308-87-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025



