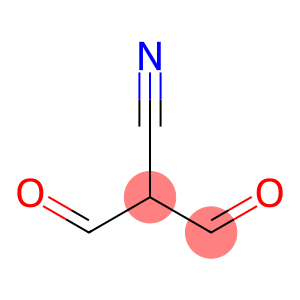
cyanomalondialdehyde
cyanomalondialdehyde
CAS: 69791-38-4
Molecular Formula: C4H3NO2
cyanomalondialdehyde - Names and Identifiers
| Name | cyanomalondialdehyde |
| Synonyms | CyanoMalondialdehyde cyanomalondialdehyde Propanenitrile, 2-formyl-3-oxo- |
| CAS | 69791-38-4 |
cyanomalondialdehyde - Physico-chemical Properties
| Molecular Formula | C4H3NO2 |
| Molar Mass | 97.07 |
| Density | 1.170±0.06 g/cm3(Predicted) |
| Boling Point | 172.8±35.0 °C(Predicted) |
| pKa | -1.86±0.10(Predicted) |
cyanomalondialdehyde - Introduction
cyanomalondialdehyde(cyanomalondialdehyde) is an organic compound with the chemical formula C4H3NO2. The following is a description of its nature, use, formulation and safety information:
Nature:
cyanomalondialdehyde is a yellow to orange-red crystalline solid with a pungent odor. It is soluble in organic solvents, such as alcohols and ethers, but practically insoluble in water. Such compounds readily self-polymerize to form high polymers.
Use:
cyanomalondialdehyde have important uses in organic synthesis. It can be used as a bifunctional reagent in organic synthesis and can react with other compounds, for example, with amines to generate hydrazone compounds, or with aldehydes and ketones to generate hydroxyimines. Due to its diversity and flexibility in the reaction, it has a wide range of applications in the fields of pharmaceuticals, pesticides and dyes.
Preparation Method:
The preparation of cyanomalondialdehyde is usually achieved by the oxygen hybridization reaction of propanedione. The oxidant is usually chlorophyll or peroxychlorophyll, which reacts with propanedione to form cyanomalondialdehyde. The preparation process requires care to prevent the oxidant from contacting the air during the reaction and avoid dangerous auto-oxidation reactions.
Safety Information:
cyanomalondialdehyde is an organic compound that is irritating and corrosive. Appropriate personal protective equipment, including glasses, gloves and lab coats, must be worn during handling and handling. Inhalation of harmful vapors and contact with skin should be avoided. In case of contact with skin or eyes, rinse immediately with plenty of water and seek medical help. In addition, cyanomalondialdehyde should be stored in well-sealed containers, away from fire and oxidizing agents, to prevent dangerous reactions.
Nature:
cyanomalondialdehyde is a yellow to orange-red crystalline solid with a pungent odor. It is soluble in organic solvents, such as alcohols and ethers, but practically insoluble in water. Such compounds readily self-polymerize to form high polymers.
Use:
cyanomalondialdehyde have important uses in organic synthesis. It can be used as a bifunctional reagent in organic synthesis and can react with other compounds, for example, with amines to generate hydrazone compounds, or with aldehydes and ketones to generate hydroxyimines. Due to its diversity and flexibility in the reaction, it has a wide range of applications in the fields of pharmaceuticals, pesticides and dyes.
Preparation Method:
The preparation of cyanomalondialdehyde is usually achieved by the oxygen hybridization reaction of propanedione. The oxidant is usually chlorophyll or peroxychlorophyll, which reacts with propanedione to form cyanomalondialdehyde. The preparation process requires care to prevent the oxidant from contacting the air during the reaction and avoid dangerous auto-oxidation reactions.
Safety Information:
cyanomalondialdehyde is an organic compound that is irritating and corrosive. Appropriate personal protective equipment, including glasses, gloves and lab coats, must be worn during handling and handling. Inhalation of harmful vapors and contact with skin should be avoided. In case of contact with skin or eyes, rinse immediately with plenty of water and seek medical help. In addition, cyanomalondialdehyde should be stored in well-sealed containers, away from fire and oxidizing agents, to prevent dangerous reactions.
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: CyanoMalondialdehyde Request for quotation
CAS: 69791-38-4
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 69791-38-4
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: CyanoMalondialdehyde Request for quotation
CAS: 69791-38-4
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 69791-38-4
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
View History
