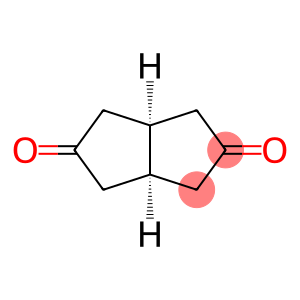
Tetrahydro-2,5(1H,3H)-pentalenedione
(3as,6as)-tetrahydropentalene-2,5(1H,3H)-dione
CAS: 51716-63-3
Molecular Formula: C8H10O2
Tetrahydro-2,5(1H,3H)-pentalenedione - Names and Identifiers
Tetrahydro-2,5(1H,3H)-pentalenedione - Physico-chemical Properties
| Molecular Formula | C8H10O2 |
| Molar Mass | 138.16 |
| Density | 1.191±0.06 g/cm3(Predicted) |
| Melting Point | 83-86°C(lit.) |
| Boling Point | 268.3±33.0 °C(Predicted) |
| Solubility | DCM, Ethyl Acetate, Methanol |
| Appearance | Powder |
| Color | White |
| Storage Condition | Sealed in dry,Room Temperature |
Tetrahydro-2,5(1H,3H)-pentalenedione - Risk and Safety
| WGK Germany | 3 |
Tetrahydro-2,5(1H,3H)-pentalenedione - Reference Information
| Overview | cis-bicyclo [3.3.0] octane -3, 7-dione alias N,N,N ', n'-tetramethyl-1, 3-propanediamine, 1, 3-bis (dimethylamino) propane, cis-bicyclo [3.3.0] octane -3, 7-diketone can be used as pharmaceutical chemical synthesis intermediates, such as the synthesis of Loganin intermediates, Loganin is a kind of iridoid glycosides, has a protective effect, fight liver damage and other diabetic complications associated with abnormal metabolic states and inflammation caused by oxidative stress. |
| emergency measures | If inhalation of cis-bicyclo [3.3.0] octane -3, 7-dione, please move the patient to fresh air; If the skin is in contact, remove the contaminated clothing and thoroughly rinse the skin with soap and water. If you feel uncomfortable, see a doctor; If the eyes are in contact, separate the eyelids, rinse with running water or normal saline and seek medical attention immediately. If you eat, rinse your mouth immediately. |
| preparation | Step 1:(CIS)-tetramethylbicyclo [3.3.0] octane -3, synthesis of 7-dioxo-2, 4,6, 8-tetracarboxylate sodium hydroxide (6.40g,0.16mol) was dissolved in of methanol in an ice-water bath, under stirring, dimethyl 1, 3-propane-dicarboxylate (22.6mL,0.16mol) was added dropwise. The reaction solution was heated to reflux until the salt was completely dissolved, rapidly added dropwise with glyoxal 14a(12.85g,0.09mol) at 65 °c, then cooled to room temperature, stirred for 12 hours and filtered. The filter cake was washed with 50ml of methanol and dissolved in 180ml of a mixed solvent of dichloromethane and water (V / V = 5:4). The resulting solution was cooled to 0 °c, adjusted to pH 6 by dropwise addition of 1M hydrochloric acid and extracted with dichloromethane (50ml x 3). The combined organic phases were washed with saturated sodium chloride solution (50ml × 3), then dried over anhydrous magnesium sulfate, filtered, and the filtrate was concentrated under reduced pressure to give the title compound (CIS).-Tetramethylbicyclo [3.3.0] octane -3, 7-dioxo-2, 4,6, 8-tetracarboxylate 14b(20g, yield: 61.0%) as a white solid. MS m / z(ESI):371.3 [M 1] Step 2: Synthesis of cis-bicyclo [3.3.0] octane -3, 7-dione (CIS)-The tetracyclic bicyclo [3.3.0] octane -3, 7-dioxo-2, 4,6, 8-tetracarboxylate 14b(6.75g,0.02mol) was dissolved in of acetic acid, 30 1 ml of 1M hydrochloric acid were then added. The reaction solution was heated to reflux for 3.5 hours and then cooled to room temperature. The reaction solution was extracted with dichloromethane (50ml x 3). The combined organic phases are concentrated under reduced pressure, 100ml of dichloromethane are added, saturated sodium bicarbonate solution is added dropwise, the pH is adjusted to 7 and separated. The organic phase was dried over anhydrous magnesium sulfate, filtered and the filtrate was concentrated under reduced pressure to give the title compound cis-bicyclo [3.3.0] octane -3, 7-dione 14c(2G, yield: 80.0%), white solid. |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: cis-Bicyclo[3.3.0]octane-3,7-dione Visit Supplier Webpage Request for quotationCAS: 51716-63-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: CIS-BICYCLO[3.3.0]OCTANE-3,7-DIONE Request for quotation
CAS: 51716-63-3
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 51716-63-3
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: Cis-Bicyclo[3.3.0]Octane-3,7-Dione Visit Supplier Webpage Request for quotation
CAS: 51716-63-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 51716-63-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: cis-Bicyclo[3.3.0]octane-3,7-dione Visit Supplier Webpage Request for quotationCAS: 51716-63-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: CIS-BICYCLO[3.3.0]OCTANE-3,7-DIONE Request for quotation
CAS: 51716-63-3
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 51716-63-3
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: Cis-Bicyclo[3.3.0]Octane-3,7-Dione Visit Supplier Webpage Request for quotation
CAS: 51716-63-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 51716-63-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


