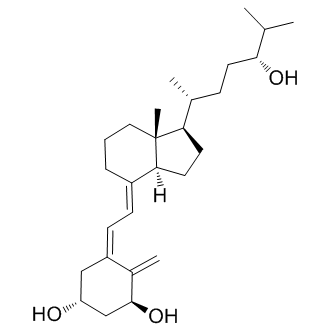
Tacalcitol
Tacalcitol
CAS: 57333-96-7
Molecular Formula: C27H44O3
Tacalcitol - Names and Identifiers
Tacalcitol - Physico-chemical Properties
| Molecular Formula | C27H44O3 |
| Molar Mass | 416.64 |
| Density | 1.06 |
| Boling Point | 565.0±50.0 °C(Predicted) |
| pKa | 14.43±0.40(Predicted) |
| Storage Condition | Sealed in dry,Store in freezer, under -20°C |
| Physical and Chemical Properties | Chemical properties white solid. UV maximum absorption (ethanol):265nm. |
| Use | Use anti-psoriasis, keratosis drugs. Used for psoriasis, ichthyosis, palmoplantar pus scar and keratosis. |
Tacalcitol - Upstream Downstream Industry
| Raw Materials | Tetrahydrofuran Lithium Aluminum Hydride 3-Chloroperoxybenzoic acid Silica gel Potassium tert-butoxide Argon CALCIUM HYDRIDE |
Tacalcitol - Preparation solution concentration reference
| 1mg | 5mg | 10mg | |
|---|---|---|---|
| 1 mM | 2.4 ml | 12 ml | 24.001 ml |
| 5 mM | 0.48 ml | 2.4 ml | 4.8 ml |
| 10 mM | 0.24 ml | 1.2 ml | 2.4 ml |
| 5 mM | 0.048 ml | 0.24 ml | 0.48 ml |
Tacalcitol - Uses and synthesis methods
biological activity
Tacalcitol (1,24(R)-Dihydroxyvitamin D3; 1.alpha.,24R-Dihydroxyvitamin D3) can promote normal bone growth by regulating calcium ions.
Production method
889.5mg compound (I) is dissolved in 30ml dimethyl sulfoxide, potassium tert-butoxide (prepared from 0.5g potassium) is added, and reacted at 20 ℃ for 30min under argon. Pour into ice water and extract with ether. The extract is washed, dried and concentrated with water. The residue is dissolved in ether, at -10 ℃ or below, drop it into the solution of calcium borohydride and calcium chloride in methanol, and then stir at -10 ℃ for 1h. Excess calcium borohydride is destroyed with aqueous acetic acid solution, then concentrated and extracted with dichloromethane. The extract is washed, dried and concentrated. The residue is dissolved in dichloromethane, and compound (II) is added in small batches. After adding, stir at room temperature for 1h. The solvent was distilled out, and the residue was eluted with 1% methanol chloroform solution by silica gel chromatography to obtain 400.35mg compound (Ⅲ).
400.35mg compound (Ⅲ) is dissolved in chloroform, m-chloroperbenzoic acid is added, and stirred at 25 ℃ for 40h. The reaction solution is washed, dried and concentrated with potassium carbonate aqueous solution and water. The residue was eluted with 1% methanol chloroform solution by silica gel chromatography to obtain 176.6mg compound (Ⅳ).
83.0mg compound (Ⅳ) is dissolved in dry tetrahydrofuran, lithium aluminum hydride is added in small batches, and refluxed gently for 1h. Sodium sulfate is added to destroy excess lithium aluminum hydride. The organic layer is separated, dried and concentrated. The residue was purified by SephadexLH-20 chromatography and eluted with chloroform-hexane (65:35 volume ratio) to obtain 21.3mg compound (V).
21.3mg compound (V) is dissolved in ether and irradiated with a 400W high-pressure mercury lamp. During the irradiation, argon is introduced to stir. Concentrated, the residue was eluted with chloroform-hexane (65:35 volume ratio) by SephadexLH-20 chromatography to obtain 3.64mg product.
Tacalcitol - Reference Information
| biological activity | Tacalcitol (1,24(R)-Dihydroxyvitamin D3; 1.alpha., 24R-dihydroxyvitamin D3) can promote normal bone growth by regulating calcium ions. |
| chemical properties | white solid. UV maximum absorption (ethanol):265nm. |
| use | anti-psoriasis and keratosis drugs. Used for psoriasis, ichthyosis, palmoplantar pus scar and keratosis. |
| production method | 889.5mg compound (I) is dissolved in 30ml dimethyl sulfoxide, potassium tert-butoxide (prepared from 0.5g potassium) is added, and reacted at 20 ℃ for 30min under argon gas. Pour into ice water and extract with ether. The extract is washed, dried and concentrated with water. The residue is dissolved in ether, at -10 ℃ or below, drop it into the solution of calcium borohydride and calcium chloride in methanol, and then stir at -10 ℃ for 1h. Excess calcium borohydride is destroyed with aqueous acetic acid solution, then concentrated and extracted with dichloromethane. The extract is washed, dried and concentrated. The residue is dissolved in dichloromethane, and compound (II) is added in small batches. After adding, stir at room temperature for 1h. The solvent was distilled out, and the residue was eluted with 1% methanol chloroform solution by silica gel chromatography to obtain 400.35mg compound (Ⅲ). 40.35mg compound (III) is dissolved in chloroform, with the addition of me-chloroperbenzoic acid, stirring at 25 ℃ for 40h. The reaction solution is washed, dried and concentrated with potassium carbonate aqueous solution and water. The residue was eluted with 1% methanol chloroform solution by silica gel chromatography to obtain 176.6mg compound (Ⅳ). 83.0mg compound (Ⅳ) is dissolved in dry tetrahydrofuran, lithium aluminum hydride is added in small batches, and refluxed gently for 1h. Sodium sulfate is added to destroy excess lithium aluminum hydride. The organic layer is separated, dried and concentrated. The residue was purified by SephadexLH-20 chromatography and eluted with chloroform-hexane (65:35 volume ratio) to obtain 21.3mg compound (V). 21.3mg compound (V) is dissolved in ether, irradiated with a 400W high-pressure mercury lamp, and stirred by argon during the irradiation. Concentrated, the residue was eluted with chloroform-hexane (65:35 volume ratio) by SephadexLH-20 chromatography to obtain 3.64mg product. |
Supplier List
CAS: 57333-96-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 57333-96-7
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
CAS: 57333-96-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 57333-96-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 57333-96-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 57333-96-7
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
CAS: 57333-96-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 57333-96-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565



