TUBELMOSIDEA
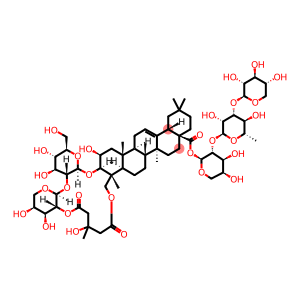
Tubeimoside A
CAS: 102040-03-9
Molecular Formula: C63H98O29
TUBELMOSIDEA - Names and Identifiers
| Name | Tubeimoside A |
| Synonyms | Lobatoside H TUBELMOSIDEA TUBEMOSIDE A Tubeimoside A TUBEIMOSIDE I TUBEIMOSIDE A TUBEIMOSIDE 1 hydroxy-3-methyl-1-oxobutyl)-α-L-arabinopyra nosyl]-β-D-glucopyranosyl]oxy]-2,23-dihydroxy-,28-(O-β-D-xylopyranosyl-(1→3)-O-6-deoxy-α-L-mannopyranosyl-(1→2)-α-L-arabinopyranosyl)ester, intramol. ester, [2β,3β(S),4α]- (1S,4S,7R,8S,9R,11S,13S,14S,18S,21R,22R,23S,24R,25R,27R,28S,29S,30R,32R,34R,35S,37S,38S,41R,42R,46S,53S,54R,56S,57R)-7,8,18,22,23,24,28,29,35,57-decahydroxy-30,54-bis(hydroxymethyl)-13,18,37,41,42,46,48,48,53,54-decamethyl-56-{[(2S,3R,4S,5R)-3,4,5-trihydroxytetrahydro-2H-pyran-2-yl]oxy}-3,5,10,12,15,26,31,33,55-nonaoxadecacyclo[39.9.3.2~11,14~.1~21,25~.1~34,38~.0~1,46~.0~4,9~.0~27,32~.0~37,42~.0~45,53~]heptapentacont-44-ene-2,16,20-trione (non-preferred name) |
| CAS | 102040-03-9 |
| InChI | InChI=1/C65H102O29/c1-27-45-47(90-51-42(79)36(73)30(70)23-84-51)44(81)53(86-27)91-48-37(74)31(71)24-85-54(48)94-56(82)65-17-15-57(2,3)25-62(65,8)34-12-14-64(10)61(7)20-29(69)50(59(5,26-67)33(61)11-13-63(64,9)60(34,6)16-18-65)93-55-49(40(77)38(75)32(22-66)87-55)92-52-43(80)39(76)41(78)46(89-52)28(68)19-58(4,83)21-35(72)88-45/h12,27,29-33,36-55,66-67,69-71,73-81,83H,11,13-26H2,1-10H3/t27-,29-,30+,31+,32+,33+,36-,37-,38+,39-,40-,41+,42+,43+,44+,45-,46-,47-,48+,49+,50-,51-,52-,53-,54-,55-,58-,59-,60+,61-,62-,63-,64+,65+/m0/s1 |
TUBELMOSIDEA - Physico-chemical Properties
| Molecular Formula | C63H98O29 |
| Molar Mass | 1319.43 |
| Density | 1.51±0.1 g/cm3(Predicted) |
| Melting Point | 250~252℃ |
| Boling Point | 250-252°C |
| Specific Rotation(α) | +14.6 (c, 1.09 in MeOH). +15.5 (c, 0.6 in Py) |
| Solubility | Soluble in methanol and ethanol |
| Appearance | Colorless needle crystal (methanol) |
| Color | White to Off-White |
| pKa | 12.54±0.70(Predicted) |
| Storage Condition | under inert gas (nitrogen or Argon) at 2-8°C |
| Sensitive | Easily absorbing moisture |
| Refractive Index | 1.638 |
| MDL | MFCD03427680 |
| Physical and Chemical Properties | White crystalline powder, soluble in methanol ethanol, derived from Fritillaria. |
TUBELMOSIDEA - Risk and Safety
| HS Code | 29389090 |
TUBELMOSIDEA - Reference
| Reference Show more | 1. Wang Xinyue, Zhu Xueting, Liu Tiantian, Li Juan, Li Yunkun, Wu Jiayu, Li Dong, Liu Qijun, Tang Renyong, Du Xiaogang. Molecular Effect of Tubeimoside A on Reversing Drug Resistance of A549/DDP Cell Line [J]. Genomics and Applied Biology, 2020,39(08):3600-3606. 2. [IF = 5.64] Wu Dousheng et al."Oleanolic Acid Induces the Type III Secretion System of Ralstonia solanacearum." Front Microbiol. 2015 Dec;0:1466 |
TUBELMOSIDEA - Reference Information
| traits | light yellow or white-like powder |
| use | is used to treat flat warts with damp-heat toxic syndrome, and can also resist virus and tumor, clear away heat and detoxify, remove dampness and dispel knots. Spread poison and eliminate carbuncle. Tubeimoside A has the effect of anti-tumor metastasis. used for content determination/identification/pharmacological experiment, etc. Pharmacological effects: used for breast carbuncle, scrofula, mastitis, cervical lymph node tuberculosis, chronic lymphadenitis, hypertrophic rhinitis. |
| principle of action | strong analgesia: the analgesic effect is equivalent to that of the common analgesic aminopyrine 7 times, the analgesic effect is equivalent to that of dolantin, and the analgesic effect is maintained for a longer time, which is a substitute for tramadol. Long-lasting effect: The anesthetic isomer of Aconitum has a strong anesthetic effect from deep to shallow. It has been clinically proved that the local anesthetic effect is equivalent to the Kecain; the nerve conduction block effect is 5.25 times that of Kecain and procaine 13 times; infiltration anesthesia is far stronger than procaine and Kecain. High safety: It has high safety, no addiction, mutagenic and carcinogenic effects, and no accumulation, stimulation, allergy and toxic effects on the immune system. The clinical application proves that there is no case of poisoning within the prescribed usage and dosage range. Toxicological animal tests of this product do not cause |
Last Update:2024-04-09 15:16:46
Supplier List
Multiple SpecificationsSpot supply
Product Name: Tubeimoside I Visit Supplier Webpage Request for quotationCAS: 102040-03-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Tubeimoside A Request for quotation
CAS: 102040-03-9
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 102040-03-9
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: Tubeimoside A Request for quotation
CAS: 102040-03-9
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 102040-03-9
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Multiple SpecificationsSpot supply
Product Name: Tubeimoside I Visit Supplier Webpage Request for quotationCAS: 102040-03-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Multiple SpecificationsSpot supply
Product Name: Tubeimoside I Visit Supplier Webpage Request for quotationCAS: 102040-03-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Tubeimoside A Request for quotation
CAS: 102040-03-9
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 102040-03-9
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: Tubeimoside A Request for quotation
CAS: 102040-03-9
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 102040-03-9
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Multiple SpecificationsSpot supply
Product Name: Tubeimoside I Visit Supplier Webpage Request for quotationCAS: 102040-03-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



