Staurosporine
Staurosporine
CAS: 62996-74-1
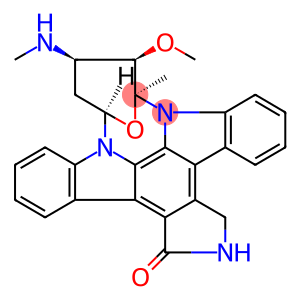
Molecular Formula: C28H26N4O3
Staurosporine - Names and Identifiers
Staurosporine - Physico-chemical Properties
| Molecular Formula | C28H26N4O3 |
| Molar Mass | 466.54 |
| Density | 1.56±0.1 g/cm3(Predicted) |
| Melting Point | 270°C |
| Boling Point | 677.5±55.0 °C(Predicted) |
| Specific Rotation(α) | D25 +35.0° (c = 1 in methanol); D22 +56.1° (c = 0.14 in methanol) |
| Flash Point | 363.6°C |
| Water Solubility | Soluble in DMSO or ethanol.Soluble in dimethyl sulfoxide , dimethyl formamide, ethyl acetate, hot acetone and ethanol. Slightly soluble in chloroform and methanol. Insoluble in water. |
| Solubility | DMSO: soluble |
| Vapor Presure | 3.25E-18mmHg at 25°C |
| Appearance | White to pale yellow solid |
| Color | Off white to pale yellow |
| BRN | 1060573 |
| pKa | 14.25±0.70(Predicted) |
| Storage Condition | 2-8°C |
| Stability | Stable for 1 year from date of purchase as supplied. Solutions in DMSO may be stored at -20° for up to 4 months. |
| MDL | MFCD00077402 |
| In vitro study | Staurosporine also strongly inhibited HeLa S3 cells with an IC50 of 4 nM. Staurosporine also inhibits a variety of other protein kinases, including PKA, PKG, phosphokinase, S6 kinase, myosin light chain kinase (MLCK), CAM PKII, cdc2, v-Src, Lyn, c-Fgr, and Syk , IC50 were 15 nM, 18 nM, 3 nM, 5 nM, 21 nM, 20 nM, 9 nM, 6 nM, 20 nM, 2 nM, And 16 nM. Staurosporine (1μm) induces apoptosis in PC12 cells in more than 90%, which can be inhibited by overexpressed Bcl-2, or zVAD-fmk, cycloheximide (10μm) and actinomycin D (5μm) treatment can also be inhibited. Accordingly, Staurosporine treatment induces intracellular free calcium levels [Ca through caspase-8 activation and Bid Division, and the expression of functional caspase-3 can enhance Staurosporine-induced MCF7 cell death. 1 μm Staurosporine treatment only partially inhibited IL-3-stimulated Bcl2 phosphorylation, while completely blocked PKC-regulated Bcl2 phosphorylation. Staurosporine induced AG-1518 apoptosis in human foreskin fibroblasts, regulated cytochrome c release and caspase activation according to lysosomal cathepsin D. In addition to activating the traditional mitochondrial apoptotic pathway, Staurosporine triggers a novel intrinsic apoptotic pathway that relies on caspase-9 activation in the presence of Apaf-1. |
| In vivo study | Pretreatment of locally anemic gerbils and rat models with Staurosporine(0.1-10 ng) before local anaemia prevented neuronal damage in a dose-dependent manner, indicating that PKC is involved in calamus pyramidal cell death. |
Staurosporine - Risk and Safety
| Risk Codes | R40 - Limited evidence of a carcinogenic effect R45 - May cause cancer R36/37/38 - Irritating to eyes, respiratory system and skin. R46 - May cause heritable genetic damage |
| Safety Description | S36/37 - Wear suitable protective clothing and gloves. S53 - Avoid exposure - obtain special instructions before use. S36 - Wear suitable protective clothing. S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) |
| UN IDs | UN 2811 6.1 / PGII |
| WGK Germany | 2 |
| RTECS | KD5084000 |
| FLUKA BRAND F CODES | 8-10 |
| HS Code | 29419090 |
| Hazard Class | 3 |
Staurosporine - Reference
| Reference Show more | 1. [IF=6.023] Zhuang Hong et al."Rosmarinic acid attenuates acrylamide induced apoptosis of BRL-3A cells by inhibiting oxidative stress and endoplasmic reticulum stress."Food Chem Toxicol. 2021 May;151:112156 |
Staurosporine - Reference Information
| Overview | Staurosporine has biological activity ranging from antifungal to antihypertensive. The interest in these biological activities has led to extensive research work in chemistry and biology and the discovery of the potential for anti-cancer therapeutics. The main biological activity of Staurosporine is the inhibition of protein kinases by preventing the binding of ATP to kinases. This is achieved by a stronger affinity of staurosporine for the ATP binding site on the kinase. |
| Application | Staurosporine is a typical ATP-competitive kinase inhibitor because it binds many kinases with high affinity, but the selectivity is very small. The structural analysis of the kinase pocket showed that the main chain atoms conserved in the relative position to the staurosporine contributed to the disorder of the staurosporine. Staurosporine is a key intermediate of midostaurin, and its production process directly affects the production cost and quality of midostaurin. |
| structural properties | staurosporine (sta), structural formula (1) it is a natural product with good pharmaceutical and biological activity. This compound was first isolated from soil actinomycetes by omura in 1997 as an indole carbazole alkaloid. x-ray single crystal diffraction analysis showed that the central indolocarbazole ring, ring below the n linked to a small molecule sugar, linked to the above is the lactam ring. sta is a strong pkc inhibitor (ic50 = 2.7nm), which mainly acts on various kinases, topoisomerases and cell cycle regulators in cell signal transduction pathway. |
| extraction method | this method is to adjust the pH value of the culture medium to 10 with ammonia water after the fermentation is completed, N-butyl acetate was added at room temperature for extraction; Aqueous hcl was added to transfer the product to aqueous solution; PH was adjusted to 10 with ammonia, extracted with ethyl acetate, and concentrated to obtain Brown coagulant; Washed with ether, removal of fat; Silica Gel chromatography, eluent chloroform-methanol, using thin layer control, concentrated, dried to give a pure product. The purity was more than 99%. |
| biological activity | Staurosporine (CGP 41251, Antibiotic AM-2282, STS, AM-2282) it is a potent PKC inhibitor, which acts on pkcα, pkcγ and pkcη in a cell-free assay with IC50 of 2 nM,5 nM and 4 nM, respectively, the effect on PKC δ(20 nM) and PKC ε(73 nM) was weak, and the activity on PKC ζ (1086 nM) was very low. It also showed inhibitory activity against other kinases PKA, PKG, S6K, CaMKII, etc. Phase 3. |
| Target | Value |
| PKCα (Cell-free assay) | 2 nM |
| c-Fgr (Cell-free assay) | 2 nM |
| phosphorylase kinase (Cell-free assay) | 3 nM |
| PKCη (Cell-free assay) | 4 nM |
| PKCγ (Cell-free assay) | 5 nM |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: Staurosporine Visit Supplier Webpage Request for quotationCAS: 62996-74-1
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: STAUROSPORINE Request for quotation
CAS: 62996-74-1
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 62996-74-1
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Multiple SpecificationsSpot supply
Product Name: Staurosporine Visit Supplier Webpage Request for quotationCAS: 62996-74-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Staurosporine from Streptomyces sp Visit Supplier Webpage Request for quotationCAS: 62996-74-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Staurosporine Visit Supplier Webpage Request for quotationCAS: 62996-74-1
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: STAUROSPORINE Request for quotation
CAS: 62996-74-1
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 62996-74-1
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Multiple SpecificationsSpot supply
Product Name: Staurosporine Visit Supplier Webpage Request for quotationCAS: 62996-74-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Staurosporine from Streptomyces sp Visit Supplier Webpage Request for quotationCAS: 62996-74-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



