SUMIVERDE
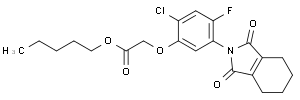
pentyl [2-chloro-5-(1,3-dioxo-1,3,4,5,6,7-hexahydro-2H-isoindol-2-yl)-4-fluorophenoxy]acetate
CAS: 87546-18-7
Molecular Formula: C21H23ClFNO5
SUMIVERDE - Names and Identifiers
| Name | pentyl [2-chloro-5-(1,3-dioxo-1,3,4,5,6,7-hexahydro-2H-isoindol-2-yl)-4-fluorophenoxy]acetate |
| Synonyms | S 23031 RESOURCE SUMIVERDE FLUMICLORAC-PENTYL flumiclorac-pentyl (bsi,pa iso,ansi) PENTYL2-CHLORO-4-FLUORO-5-(3,4,5,6-TETRAHYDROPHTHALIMIDO). PENTYL2-CHLORO-4-FLUORO-5-(3,4,5,6-TETRAHYDROPHTHALIMIDO)PHENOXYACETATE pentyl-[2-chloro-5-(cyclohex-1-ene-1,2-dicarboxamido)-4-fluorophenoxy] acetate pentyl [2-chloro-5-(1,3-dioxo-1,3,4,5,6,7-hexahydro-2H-isoindol-2-yl)-4-fluorophenoxy]acetate |
| CAS | 87546-18-7 |
| InChI | InChI=1/C21H23ClFNO5/c1-2-3-6-9-28-19(25)12-29-18-11-17(16(23)10-15(18)22)24-20(26)13-7-4-5-8-14(13)21(24)27/h10-11H,2-9,12H2,1H3 |
SUMIVERDE - Physico-chemical Properties
| Molecular Formula | C21H23ClFNO5 |
| Molar Mass | 423.86 |
| Density | d20 1.3316 |
| Melting Point | 90-91℃ |
| Boling Point | 558.0±50.0 °C(Predicted) |
| Flash Point | 291.3°C |
| Vapor Presure | 1.74E-12mmHg at 25°C |
| pKa | -2.32±0.20(Predicted) |
| Storage Condition | 0-6°C |
| Refractive Index | 1.575 |
| Physical and Chemical Properties | The pure product is a white powdery solid. Melting point 88.9~90.1 ℃, relative density 1.33(20 ℃), vapor pressure 1.0 × 10-5Pa (25 ℃), partition coefficient 4.99 (20 ℃), solubility at 25 ℃: methanol 47.8g/L, n-octanol 16.0g/L, acetone 590g/L, n-hexane 3.28g/L, water 0.189mg/L. The half-life in water is 6min (pH = 9), 19h(pH = 7) and 42d (pH = 5). |
SUMIVERDE - Risk and Safety
| HS Code | 29251900 |
| Toxicity | LD50 in rats (mg/kg): >5000 orally; >2000 dermally (Kamoshita) |
SUMIVERDE - Reference Information
| toxicity | acute oral LD50>3600mg/kg in rats, acute percutaneous LD502000mg/kg in rabbits, and acute inhalation of LC505.51 mg/l in rats. It has moderate irritation to rabbit eyes and skin, no mutagenicity, no teratogenic effect. Acute oral LD50 of quail> 2250mg/kg, LC50 of quail and mallard> 5620mg/L. LC5013 ~ 21mg/L for sunfish and 1.1mg/L for rainbow trout (96h). |
| Chemical properties | The pure product is a white powdery solid. Melting point 88.9~90.1 ℃, relative density 1.33(20 ℃), vapor pressure 1.0 × 10-5Pa (25 ℃), partition coefficient 4.99 (20 ℃), solubility at 25 ℃: methanol 47.8g/L, n-octanol 16.0g/L, acetone 590g/L, n-hexane 3.28g/L, water 0.189mg/L. The half-life in water is 6min (pH = 9), 19h(pH = 7) and 42d (pH = 5). |
| use | protophylline oxidase inhibitor. After the agent is absorbed by the sensitive miscellaneous leaf surface, it quickly acts on the plant tissue, causing the accumulation of protoporphyrin and enhancing the lipid peroxidation of the cell membrane, which leads to irreversible damage to the cell structure and cell function of sensitive weeds. This agent is a post-seedling herbicide, which is used to control broad-leaved weeds known in soybean fields for one year, such as Xanthium, ragweed, quinoa, amaranth weeds, spotted brocade, yellow flower, Datura, Abutilon, etc. It can also be used for corn crops. The recommended dosage is 45~67.5g active ingredient/hm2, and there are also 40~100g active ingredient/hm2. |
| production method | preparation of 2-chloro -4-fluoro -5-nitrophenol 112g of p-fluorophenol and 300mL of solvent are introduced into about 75-81g of chlorine at 0-10 ℃. after the reaction is completed, the water is washed to a pH value of about 6-7, and the water layer is separated for later use. Add 114g of ethyl chloroformate to the reaction solution of the previous step at 0~10 ℃, add 84g of 50% NaOH solution dropwise, maintain the temperature at 0~10 ℃, continue the reaction for 2h, wash with water, separate the water layer, and stay for use. The reaction solution of the previous step is added 250mL of concentrated sulfuric acid at 0~10 ℃, and mixed acid (90mL of concentrated sulfuric acid +90mL of concentrated nitric acid) is added dropwise. After dropping, the reaction is continued for 0.5h. After the reaction is completed, the reaction is stratified, the inorganic layer is extracted, the organic layer is combined, the organic layer is washed with dilute alkali until neutral, the water is separated, and the organic layer falls off to obtain 245g of light yellow solid product with a content of 92% (external standard). The total yield of the above three: 85.6%. 58g of the previous product was added into 80g of 20% NaOH aqueous solution for reflux reaction for 4h, filtration and acidification of filtrate to obtain 25.5g of product. The yield is 70% and the content is 95%. Synthesis of fluoroamine ester 52g of 2-chloro-4-fluoro-5-nitrophenol, anhydrous K2CO3 43.3g and 250g of N,N-dimethylformamide were mixed, heated to 50 ℃, 47.5g of n-amyl chloroacetate was added to react at 60 ℃ for 5h to cool down, desolubilize under reduced pressure, add water, extract with ethyl ester, and desolubilize. 57.5g of iron powder, 14.5g of acetic acid and 276g of water are heated to 80 ℃, 70g of the solution prepared by the previous product and 100g of acetic acid are added dropwise, after dropping, reflux reaction for 6h, filtration, 100mL of water is added to the filtrate, extraction with ethyl acetate, and desolution. 12.0g of the previous product, 3,4,5,6-tetrahydrophthalic anhydride (7.6g), piperidine 0.2g, propionic acid 0.5g and toluene 50mL were refluxed for 7h, and water was azeotropic. After the reaction, water and toluene were added, stratified and desolated, and the remaining material was recrystallized to obtain 5.5g of product. |
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: flumiclorac-pentyl Visit Supplier Webpage Request for quotationCAS: 87546-18-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Flumiclorac-pentyl Visit Supplier Webpage Request for quotationCAS: 87546-18-7
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: FLUMICLORAC-PENTYL Request for quotation
CAS: 87546-18-7
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 87546-18-7
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Multiple Specifications
Product Name: Flumiclorac-Pentyl Visit Supplier Webpage Request for quotationCAS: 87546-18-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: flumiclorac-pentyl Visit Supplier Webpage Request for quotationCAS: 87546-18-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Flumiclorac-pentyl Visit Supplier Webpage Request for quotationCAS: 87546-18-7
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: FLUMICLORAC-PENTYL Request for quotation
CAS: 87546-18-7
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 87546-18-7
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Multiple Specifications
Product Name: Flumiclorac-Pentyl Visit Supplier Webpage Request for quotationCAS: 87546-18-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



