Resminostat
Resminostat
CAS: 864814-88-0
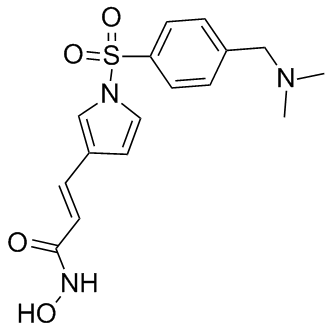
Molecular Formula: C16H19N3O4S
Resminostat - Names and Identifiers
Resminostat - Physico-chemical Properties
| Molecular Formula | C16H19N3O4S |
| Molar Mass | 349.4 |
| Density | 1.28±0.1 g/cm3(Predicted) |
| pKa | 8.68±0.23(Predicted) |
| Storage Condition | -20℃ |
| Use | Resminostat dose-dependent selective inhibition of HDAC1/3/6, IC50 is 42.5 nM/50.1 nM/71.8 nM, the effect on HDAC8 is weak, IC50 is 877 nM. |
| In vitro study | Resminostat[HCl], a potent inhibitor, is a recombinant of HDAC1,3 and 6 isozymes that competitively bind to the matrix. It acts on MM cells and can induce high acetylation of histone H4. Resminostat acts at low micromolar concentrations on MM cell lines (OPM-2,NCI-H929,U266) to prevent cell growth and strongly induce apoptosis, consistent with the effect on primary MM cells. At 1 μm, Resminostat acts on the MM cell line, inhibiting cell proliferation and inducing G0/G1 phase arrest of the cell cycle at OPM-2,NCI-H929, u266. The cell lines were accompanied by decreased levels of cyclin D1,CDC25A,CDK4 and Rb and up-regulation of the p21 gene. Resminostat directs interference with the Akt signaling pathway by reducing phosphorylation of 4E-BP1 and p70S6K. Treatment with Resminostat resulted in increased levels of Bim and Bax protein expression and a decrease of Bcl-xL levels. Resminostat activates Caspases 3, 8, and 9. In addition, Resminostat synergizes with Melphalan and Proteasome inhibitors bortezomib and S-2209. |
| In vivo study | Resminostat oral 600 mg once daily was well tolerated for 1-5 days in a 14 day cycle. Resminostat showed good PK profile with high bioavailability and low mutation rate. Resminostat oral apparent t 1/2 from 2.7 to 4.4 hours. Modulation of plasma biomarkers further indicates the activity of the drug. |
Resminostat - Preparation solution concentration reference
| 1mg | 5mg | 10mg | |
|---|---|---|---|
| 1 mM | 2.862 ml | 14.31 ml | 28.62 ml |
| 5 mM | 0.572 ml | 2.862 ml | 5.724 ml |
| 10 mM | 0.286 ml | 1.431 ml | 2.862 ml |
| 5 mM | 0.057 ml | 0.286 ml | 0.572 ml |
Last Update:2024-01-02 23:10:35
Supplier List
Spot supply
Product Name: Resminostat Visit Supplier Webpage Request for quotationCAS: 864814-88-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Resminostat Request for quotation
CAS: 864814-88-0
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 864814-88-0
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: RESMINOSTAT; 4SC-201; RAS2410 Request for quotation
CAS: 864814-88-0
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 864814-88-0
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: Resminostat Visit Supplier Webpage Request for quotation
CAS: 864814-88-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 864814-88-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Resminostat Visit Supplier Webpage Request for quotationCAS: 864814-88-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Resminostat Request for quotation
CAS: 864814-88-0
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 864814-88-0
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: RESMINOSTAT; 4SC-201; RAS2410 Request for quotation
CAS: 864814-88-0
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 864814-88-0
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: Resminostat Visit Supplier Webpage Request for quotation
CAS: 864814-88-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 864814-88-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



