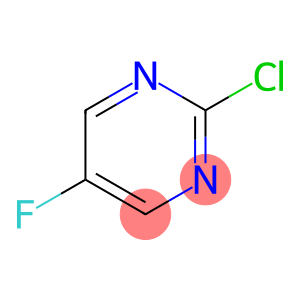
Pyrimidine,2-chloro-5-fluoro-
2-CHLORO-5-FLUOROPYRIMIDINE
CAS: 62802-42-0
Molecular Formula: C4H2ClFN2
Pyrimidine,2-chloro-5-fluoro- - Names and Identifiers
| Name | 2-CHLORO-5-FLUOROPYRIMIDINE |
| Synonyms | 2-Chloro-5-fluoropyr 2-Chlor-5-fluorpyrimidin 2-chloro-5fluoro-pyrimidine 5-Fluoro-2-chloropyrimidine 2-CHLORO-5-FLUOROPYRIMIDINE 2-Chloro-5-fluoropyriMidi... Pyrimidine,2-chloro-5-fluoro- 2-Chloro-5-fluoro-1,3-diazine Pyrimidine, 2-chloro-5-fluoro- Pyrimidine, 2-chloro-5-fluoro- (9CI) |
| CAS | 62802-42-0 |
| InChI | InChI=1/C6H3ClFNO2/c7-5-4(8)3(6(10)11)1-2-9-5/h1-2H,(H,10,11) |
| InChIKey | AGYUQBNABXVWMS-UHFFFAOYSA-N |
Pyrimidine,2-chloro-5-fluoro- - Physico-chemical Properties
| Molecular Formula | C4H2ClFN2 |
| Molar Mass | 132.52 |
| Density | 1.439 g/mL at 20 °C (lit.) |
| Boling Point | 149.0-162.0°C |
| Flash Point | 150°F |
| Vapor Presure | 9.41E-07mmHg at 25°C |
| Appearance | Liquid |
| Color | Clear colorless to yellow |
| pKa | -2.54±0.22(Predicted) |
| Storage Condition | under inert gas (nitrogen or Argon) at 2-8°C |
| Refractive Index | n20/D 1.503(lit.) |
| Physical and Chemical Properties | Appearance colorless liquid boiling point 160-162 ℃ |
| Use | Pharmaceutical Intermediates |
Pyrimidine,2-chloro-5-fluoro- - Risk and Safety
| Risk Codes | R22 - Harmful if swallowed R34 - Causes burns R41 - Risk of serious damage to eyes R37/38 - Irritating to respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) |
| UN IDs | UN 2735 8/PG 2 |
| WGK Germany | 3 |
| HS Code | 29335990 |
| Hazard Note | Harmful |
| Hazard Class | IRRITANT |
| Packing Group | Ⅲ |
Pyrimidine,2-chloro-5-fluoro- - Reference Information
| Application | 2-chloro-5-fluoropyrimidine can be used as a pharmaceutical and biochemical intermediate, such as for the preparation of 2-hydroxy-5-fluoropyrimidine. 5-fluoropyrimidine derivative is a kind of broad-spectrum anti-tumor, high efficiency anti-metabolic drugs in clinic, it has inhibitory effect on a variety of tumors. |
| preparation | add 2, 4-dichloro-5-fluoropyrimidine and reducing metal powder to the solvent, the mixture was stirred to warm up to the reaction temperature, and the acid was slowly added dropwise. After dropping, the reaction temperature was maintained until the reaction was completed. The solvent was removed by filtration, phase separation or direct evaporation, and distillation under reduced pressure to obtain 2-chloro-5-fluoropyrimidine. |
Last Update:2024-04-09 02:00:10
Supplier List
Spot supply
Product Name: Fampridine Impurity 213 Visit Supplier Webpage Request for quotationCAS: 62802-42-0
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: 2-Chloro-5-fluoropyrimidine Visit Supplier Webpage Request for quotationCAS: 62802-42-0
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: 2-Chloro-5-fluoropyrimidine Visit Supplier Webpage Request for quotationCAS: 62802-42-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-Chloro-5-fluoropyrimidine Request for quotation
CAS: 62802-42-0
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 62802-42-0
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: Fampridine Impurity 213 Visit Supplier Webpage Request for quotationCAS: 62802-42-0
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: 2-Chloro-5-fluoropyrimidine Visit Supplier Webpage Request for quotationCAS: 62802-42-0
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: 2-Chloro-5-fluoropyrimidine Visit Supplier Webpage Request for quotationCAS: 62802-42-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-Chloro-5-fluoropyrimidine Request for quotation
CAS: 62802-42-0
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 62802-42-0
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
View History



