PotassiumSodiumSilicate
PotassiumSodiumSilicate
CAS: 37328-88-4
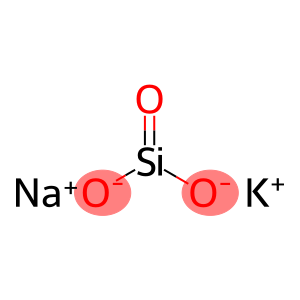
Molecular Formula: KNaO3Si
PotassiumSodiumSilicate - Names and Identifiers
PotassiumSodiumSilicate - Physico-chemical Properties
| Molecular Formula | KNaO3Si |
| Molar Mass | 138.17177 |
| Physical and Chemical Properties | Yellow-green or yellow viscous liquid, no impurity products are colorless and transparent. Odorless, tasteless. |
PotassiumSodiumSilicate - References
| Application | preparation of potassium sulfate and calcium silicate: the mother liquor of calcium silicate precipitation and potassium extraction is obtained by adding 15wt% milk of lime with mass concentration equivalent to 0.4 times of the weight of the mother liquor into the mixed mother liquor of potassium sodium silicate, stirring for 0.5h, and filtering; the mother liquor for extracting potassium is concentrated by evaporation, so that the mass content of K2O is 4.1; 98wt% sulfuric acid, which is 0.3 times the weight of mother liquor, is added to the concentrated mother liquor, and stirred for 0.5h to form crude potassium sulfate precipitate; the mixture was subjected to solid-liquid separation, washed with a potassium sulfate saturated mother liquor corresponding to 0.5 times the weight of the crude potassium sulfate precipitate to remove impurities, and then dried at 100 ° C. For 2H to obtain a potassium sulfate product. |
| purpose | mainly used as a binder, detergent, is used as a binder for phosphor screen and precision casting. It is also used as a coating self-adhesive in the manufacture of welding electrodes. It is a raw material for the production of inorganic coatings, vat dyes and silicon compounds. |
| production method | Dry mixing potassium carbonate, sodium carbonate and silica sand in a certain proportion into the reflector, controlling the temperature of 1400~1450 ℃ melting, the molten material is quenched into fragments with cold water at the discharge port, and then heated and dissolved, and then settled in the clear tank for 24h. The clear liquid is evaporated and concentrated to prepare a potassium sodium silicate finished product. Its Na2CO3 K2CO3 2nSiO2 → Na2O? Nso2 K2O? nSiO2 2CO2 wet method to the white carbon black, caustic potassium and water, into the reactor reaction, the formation of potassium silicate is not clear after adding 40 ° Bé sodium water glass, into the steam heating reaction, potassium sodium silicate finished product was prepared. |
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: PotassiumSodiumSilicate Request for quotation
CAS: 37328-88-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 37328-88-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: PotassiumSodiumSilicate Request for quotation
CAS: 37328-88-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 37328-88-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
View History
