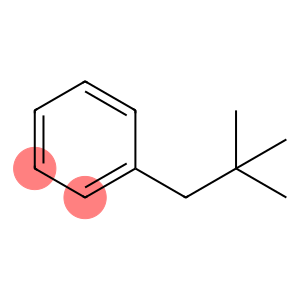
Neopentybenzene
NEOPENTYLBENZENE
CAS: 1007-26-7
Molecular Formula: C11H16
Neopentybenzene - Names and Identifiers
| Name | NEOPENTYLBENZENE |
| Synonyms | Neopentybenzene NEOPENTYLBENZENE 1-NEOPENTYLBENZENE Benzene, neopentyl- (2,2-Dimethylpropyl)Benzen (2,2-DIMETHYLPROPYL)BENZENE 2,2-DIMETHYL-1-PHENYLPROPANE (2,2-pimethylpropyl) benzene 1-(2,2-Dimethylpropyl)benzene (2,2-DIMETHYL-1-PROPYL)BENZENE (2,2-Dimethyl-1-propyl)benzene,Neopentyl benzene |
| CAS | 1007-26-7 |
| InChI | InChI=1/C11H16/c1-11(2,3)9-10-7-5-4-6-8-10/h4-8H,9H2,1-3H3 |
Neopentybenzene - Physico-chemical Properties
| Molecular Formula | C11H16 |
| Molar Mass | 148.24 |
| Density | 0.858 g/mL at 25 °C (lit.) |
| Melting Point | -44.72°C (estimate) |
| Boling Point | 185-186 °C (lit.) |
| Flash Point | 123°F |
| Water Solubility | Slightly soluble in water. |
| Vapor Presure | 0.953mmHg at 25°C |
| Appearance | clear liquid |
| Color | Colorless to Almost colorless |
| BRN | 2037544 |
| Storage Condition | Flammables area |
| Refractive Index | n20/D 1.488(lit.) |
| MDL | MFCD00038279 |
| Physical and Chemical Properties | Storage Conditions: flagmables area WGK Germany:3 |
Neopentybenzene - Risk and Safety
| Risk Codes | 10 - Flammable |
| Safety Description | 16 - Keep away from sources of ignition. |
| UN IDs | UN 3295 3/PG 3 |
| WGK Germany | 3 |
| HS Code | 29029000 |
| Hazard Class | 3 |
| Packing Group | III |
Neopentybenzene - Introduction
(2,2-Dimethylpropyl) benzene, also known as o-di-tert-butylbenzene (OTB), has the following properties:
1. Physical properties: (2,2-dimethylpropyl) benzene is a colorless liquid with aromatic odor.
2. chemical properties: it is a stable compound, not easy to be oxidized, hydrolyzed or other chemical reactions. It is stable at room temperature and not volatile.
3. Solubility: It can be dissolved in general organic solvents, such as alcohols, ethers and ketones.
The application of (2,2-dimethylpropyl) benzene mainly includes the following aspects:
1. Solvent: Due to the good solubility of (2,2-dimethylpropyl) benzene in most organic solvents, it is often used as a solvent, especially in the production of coatings, adhesives and resins.
2. Chemical intermediate: It can also be used as an important chemical intermediate for the synthesis of other organic compounds, such as polymers, dyes and drugs.
The method for preparing (2,2-dimethylpropyl) benzene mainly includes the following steps:
1. Propyl chloride reacts with benzene under acidic conditions to generate propyl benzene.
2. Under alkaline conditions, propyl benzene reacts with butyl lithium to generate (2,2-dimethylpropyl) benzene.
For safety information, (2,2-dimethylpropyl) benzene is not currently classified as a carcinogen or a teratogenic substance. However, as an organic compound, it still has a certain degree of danger. Contact with skin and eyes should be avoided and use in a well-ventilated place. During use, attention should be paid to fire prevention measures to avoid contact with strong oxidants and acidic substances. In addition, the handling and storage, should comply with the relevant safety procedures and measures.
1. Physical properties: (2,2-dimethylpropyl) benzene is a colorless liquid with aromatic odor.
2. chemical properties: it is a stable compound, not easy to be oxidized, hydrolyzed or other chemical reactions. It is stable at room temperature and not volatile.
3. Solubility: It can be dissolved in general organic solvents, such as alcohols, ethers and ketones.
The application of (2,2-dimethylpropyl) benzene mainly includes the following aspects:
1. Solvent: Due to the good solubility of (2,2-dimethylpropyl) benzene in most organic solvents, it is often used as a solvent, especially in the production of coatings, adhesives and resins.
2. Chemical intermediate: It can also be used as an important chemical intermediate for the synthesis of other organic compounds, such as polymers, dyes and drugs.
The method for preparing (2,2-dimethylpropyl) benzene mainly includes the following steps:
1. Propyl chloride reacts with benzene under acidic conditions to generate propyl benzene.
2. Under alkaline conditions, propyl benzene reacts with butyl lithium to generate (2,2-dimethylpropyl) benzene.
For safety information, (2,2-dimethylpropyl) benzene is not currently classified as a carcinogen or a teratogenic substance. However, as an organic compound, it still has a certain degree of danger. Contact with skin and eyes should be avoided and use in a well-ventilated place. During use, attention should be paid to fire prevention measures to avoid contact with strong oxidants and acidic substances. In addition, the handling and storage, should comply with the relevant safety procedures and measures.
Last Update:2024-04-10 22:40:09
Supplier List
Spot supply
Product Name: (2,2-DIMETHYL-1-PROPYL)BENZENE Visit Supplier Webpage Request for quotationCAS: 1007-26-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Multiple Specifications
Product Name: NEOPENTYLBENZENE Request for quotationCAS: 1007-26-7
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: (2,2-Dimethylpropyl)Benzene Visit Supplier Webpage Request for quotation
CAS: 1007-26-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 1007-26-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: (2,2-DIMETHYL-1-PROPYL)BENZENE Visit Supplier Webpage Request for quotationCAS: 1007-26-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Multiple Specifications
Product Name: NEOPENTYLBENZENE Request for quotationCAS: 1007-26-7
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: (2,2-Dimethylpropyl)Benzene Visit Supplier Webpage Request for quotation
CAS: 1007-26-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 1007-26-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


