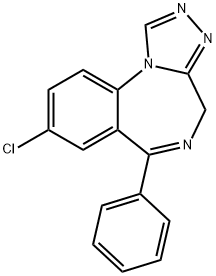
Nemurel
Estazolam
CAS: 29975-16-4
Molecular Formula: C16H11ClN4
Nemurel - Names and Identifiers
| Name | Estazolam |
| Synonyms | ProSom Julodin Nemurel Nuctalon Estazolam estazolam--dea schedule iv item estazolam 100 ug per ml in methanol 3-a)(1,4)benzodiazepine,8-chloro-6-phenyl-4h-s-triazolo( 4H-s-Triazolo(4,3-a)(1,4)benzodiazepine, 8-chloro-6-phenyl- 8-chloro-6-phenyl-4H-[1,2,4]triazolo[4,3-a][1,4]benzodiazepine 2,4)triazolo(4,3-a)(1,4)-benzodiazepine,8-chloro-6-phenyl-4h-( 4H-[1,2,4]Triazolo[4,3-a][1,4]benzodiazepine, 8-chloro-6-phenyl- |
| CAS | 29975-16-4 |
| EINECS | 249-982-4 |
| InChI | InChI=1/C16H11ClN4/c17-12-6-7-14-13(8-12)16(11-4-2-1-3-5-11)18-9-15-20-19-10-21(14)15/h1-8,10H,9H2 |
Nemurel - Physico-chemical Properties
| Molecular Formula | C16H11ClN4 |
| Molar Mass | 294.74 |
| Density | 1.2294 (rough estimate) |
| Melting Point | 228-229° |
| Boling Point | 448.04°C (rough estimate) |
| Flash Point | 11°C |
| Vapor Presure | 9.55E-10mmHg at 25°C |
| pKa | pKa 2.84(H2O) (Uncertain) |
| Storage Condition | 2-8°C |
| Refractive Index | 1.6110 (estimate) |
| Physical and Chemical Properties | White or yellowish crystals or powders. Melting point 228-229. Soluble in chloroform, slightly soluble in methanol, insoluble in acetone, ethanol, ethyl acetate and benzene. Almost insoluble in ether and water, odorless, bitter taste. |
| Use | Central nervous system, can sleep, sedation, anticonvulsant, anti Anxiety |
Nemurel - Risk and Safety
| Risk Codes | R36/37/38 - Irritating to eyes, respiratory system and skin. R39/23/24/25 - R23/24/25 - Toxic by inhalation, in contact with skin and if swallowed. R11 - Highly Flammable |
| Safety Description | S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) S36/37 - Wear suitable protective clothing and gloves. S16 - Keep away from sources of ignition. S7 - Keep container tightly closed. |
| UN IDs | 3249 |
| WGK Germany | 3 |
| RTECS | XZ5473500 |
| HS Code | 2933910000 |
| Hazard Class | 6.1(b) |
| Packing Group | III |
| Toxicity | LD50 orally in male mice, rats and rabbits: 740, 3200, 300 mg/kg (Yokotani) |
Nemurel - Standard
Authoritative Data Verified Data
This product is 6-phenyl-8-chloro-4h-[l,2,4]-triazole [4,3-a][l,4] Benzodiazepine cover. Calculated as dried product, the content of C16H11C1N4 shall not be less than 98.5%.
Nemurel - Trait
Authoritative Data Verified Data
- This product is white or white crystalline powder; Odorless.
- This product is soluble in chloroform, soluble in methanol, slightly soluble in ethyl acetate or ethanol, almost insoluble in water; Soluble in acetic anhydride.
melting point
The melting point of this product (General 0612) is 229~232°C.
absorption coefficient
take this product, precision weighing, and hydrochloric acid solution (9- 1000) dissolved and quantitatively diluted to make a solution containing about lOug per lml, the absorbance was measured at a wavelength of 0401 NM according to ultraviolet-visible spectrophotometry (General rule 349), and the absorption coefficient was 367.
Nemurel - Differential diagnosis
Authoritative Data Verified Data
- take about 10 mg of this product, add 15ml of hydrochloric acid solution (1-2), slowly boil for 15 minutes, let it cool, and the solution shows the identification reaction of aromatic first amine (General rule 0301).
- take about 1 mg of this product, add 1-2 drops of dilute sulfuric acid, and put it under UV light (365mn) to display sky-blue fluorescence.
- The infrared absorption spectrum of this product should be consistent with that of the control (Spectrum set 63) (if not, it is determined after recrystallization with methanol).
Nemurel - Exam
Authoritative Data Verified Data
chloride
take this product l. Add 50ml of water, shake for 10 minutes, filter; Take 25ml of filtrate, check according to law (General rule 0801), and standard sodium chloride solution 7. 0ml of the control solution should not be more concentrated (0.014%).
Related substances
take this product, add mobile phase to dissolve and dilute to make a solution containing 0.2mg per 1 ml as a test solution; Take appropriate amount with precision, as a control solution, a solution containing about 2ug per 1 ml was prepared by dilution with mobile phase. Determined by high performance liquid chromatography (General 0512). Silica gel bonded with eighteen alkyl silane was used as the filler; Methanol-water (65:35) was used as the mobile phase; The detection wavelength was 223mn. The number of theoretical plates shall not be less than 2000 based on the calculation of estazolam peak. 20ul of control solution and 20ul of test solution were respectively injected into human liquid chromatograph, and the chromatogram was recorded to 3 times of the retention time of the main component chromatographic peak. If there are impurity peaks in the chromatogram of the test solution, the sum of each impurity peak area shall not be greater than 0.5 times (0.5%) of the main peak area of the control solution.
loss on drying
take this product, dry to constant weight at 105°C, weight loss shall not exceed 0.5% (General rule 0831).
ignition residue
not more than 0.1% (General rule 0841).
Nemurel - Content determination
Authoritative Data Verified Data
take this product about 0.lg, precision weighing, add acetic anhydride 50ml dissolved, add crystal violet indicator 2 drops, with perchloric acid titration solution (0.1 mol/L) titration to the solution yellow, and the results of the titration with blank test correction. Each 1 ml of perchloric acid titration solution (0.1 mol/L) corresponds to 14.74mg of C16H11C1N4.
Nemurel - Category
Authoritative Data Verified Data
Anti-Anxiety drug.
Nemurel - Storage
Authoritative Data Verified Data
sealed storage.
Nemurel - Estazolam Tablets
Authoritative Data Verified Data
This product contains estazolam (C16H11C1N4) should be 90.0% ~ 110.0% of the label amount.
trait
This product is white tablet.
identification
take an appropriate amount of fine powder of this product (about 10 mg equivalent to estazolam), add 10ml of ethanol, shake to dissolve estazolam, filter, evaporate the filtrate, identify the residue under the item of estazolam (1) (2) the test showed the same reaction.
examination
- Related Substances: take an appropriate amount of fine powder of this product (equivalent to 2mg of estazolam), make a solution containing about 0.2mg per 1 ml with mobile phase, filter, and take the continued filtrate as the test solution; an appropriate amount was taken in a precise amount and diluted with the mobile phase to prepare a solution containing about 2ug per 1 ml as a control solution. The determination was carried out according to the method for related substances of estazolam. If there are impurity peaks in the chromatogram of the test solution, the sum of each impurity peak area shall not be greater than the main peak area of the control solution (1.0%).
- 1 tablet of this product shall be placed in a measuring flask with the size of lOOmKlmg) or 200ml(2mg), add an appropriate amount of hydrochloric acid solution (9-1000), and fully shake to dissolve estazolam, dilute to the scale with hydrochloric acid solution (9-1000), shake well, filter, take the continued filtrate as a test solution, and determine the content according to the method under the content determination item, the provisions shall be met (General rule 0941).
- dissolution of this product, according to the dissolution and release determination method (General rule 0931 third method), hydrochloric acid solution (9-1000)100ml(lmg specification) or 200ml(2mg specification) for the dissolution of the medium, speed of 100 rpm, according to the law, after 30 minutes, take the solution of 10ml, filtered, take the filtrate, according to UV-visible spectrophotometry (General 0401), the absorbance was measured at a wavelength of 268nm, and the elution amount per sheet was calculated as the absorption coefficient of C16H11C1N4 was 352. The limit is 80% of the labeled amount and shall be in accordance with the provisions.
- others shall be in accordance with the relevant provisions under the item of tablets (General rule 0101).
Content determination
Take 30 tablets of this product, precision weighing, fine grinding, precision weighing appropriate amount (about equivalent to estazolam lOmg), put in 100ml measuring flask, add hydrochloric acid solution (9-1000)60ml, fully shake to dissolve estazolam, dilute to the scale with hydrochloric acid solution (9-1000), shake well, filter, accurately take 5ml continuous filtrate, put 50ml flask, use hydrochloric acid solution (9-1000) dilute to the scale, shake, according to UV-visible spectrophotometry (General 0401), at the wavelength of 268mn absorbance, Cl6H11ClN4 absorption coefficient is calculated as 352, obtained.
category
Same as estazolam.
specification
(l ) lmg (2)2mg
storage
light shielding, sealed storage.
Nemurel - Estazolam injection
Authoritative Data Verified Data
This product is a sterile aqueous solution made of estazolam with suitable cosolvent. The inclusion of esmlen (C16H11ClN4) shall be between 90.0% and 110.0% of the label amount.
trait
This product is a clear colorless liquid.
identification
- take an appropriate amount of this product (about equivalent to estazolam lmg), add 2 drops of dilute sulfuric acid, and put it under UV light (365nm) to display sky-blue fluorescence.
- in the chromatogram recorded under the content determination item, the retention time of the main peak of the test solution should be consistent with the retention time of the main peak of the reference solution.
examination
- the pH value should be 5.6 to 7.0 (General 0631).
- Related substances take this product and dilute it with mobile phase to prepare a solution containing about 0.2mg estazolam per 1 ml as a test solution, A solution containing approximately estazolam per 1 ml was prepared as a control solution by dilution with mobile phase. If there are impurity peaks in the chromatogram of the test solution, the sum of each impurity peak area shall not be greater than the main peak area of the control solution (1.0%) except the sodium benzoate peak.
- others should comply with the relevant provisions under injection (General 0102).
Content determination
- measured by high performance liquid chromatography (General 0512).
- chromatographic conditions and system suitability test using eighteen alkyl silane bonded silica gel as filler; Methanol-water (65:35) as mobile phase; The detection wavelength was 223mn. The number of theoretical plates shall not be less than 2000 based on the calculation of estazolam peak.
- determination of precision quantity take appropriate amount of this product, dilute with mobile phase to a solution containing 10ug of estazolam per 1 ml, as a sample solution, and inject 20u1 into liquid chromatograph to record the chromatogram; another reference substance of estazolam was precision weighed, dissolved and quantitatively diluted with mobile phase to prepare a solution containing about 10ug per lml, which was determined by the same method. According to the external standard method to calculate the peak area, that is.
category
Same as estazolam.
specification
(l) lml : lmg (2)lml : 2mg
storage
light shielding, closed storage.