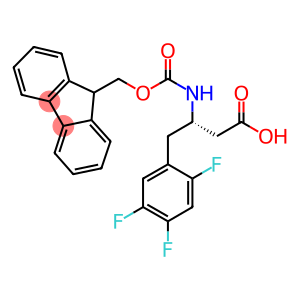
N-Fmoc-(S)-2,4,5-trifluoro-b-homophenylalanine
Fmoc-(S)-3-Amino-4-(2,4,5-trifluoro-phenyl)-butyric acid
CAS: 959580-94-0
Molecular Formula: C25H20F3NO4
N-Fmoc-(S)-2,4,5-trifluoro-b-homophenylalanine - Names and Identifiers
N-Fmoc-(S)-2,4,5-trifluoro-b-homophenylalanine - Physico-chemical Properties
| Molecular Formula | C25H20F3NO4 |
| Molar Mass | 455.43 |
N-Fmoc-(S)-2,4,5-trifluoro-b-homophenylalanine - Introduction
Fmoc-(S)-3-Amino-4-(2,4,5-trifluoro-phenyl)-butyric acid is an organic compound. The following is a description of its nature, use, formulation and safety information:
Nature:
-Molecular formula: C19H17F3NO4
-Molecular weight: 389.34g/mol
-Appearance: White crystalline solid
-Melting point: 176-180°C
-Solubility: Soluble in organic solvents (such as dichloromethane, dimethylformamide), insoluble in water
Use:
Fmoc-(S)-3-Amino-4-(2,4,5-trifluoro-phenyl)-butyric acid is an oximine-protected amino acid, which is commonly used in peptide synthesis in chemical synthesis. Its main uses include solid phase synthesis, peptide synthesis, drug development and biomedical fields.
Preparation Method:
The preparation of Fmoc-(S)-3-Amino-4-(2,4,5-trifluoro-phenyl)-butyric acid usually requires a multi-step reaction, and the specific steps may vary depending on experimental conditions. A common method of preparation is to react (S)-3-amino -4-(2,4, 5-trifluorophenyl) butanoic acid with Fmoc-Cl under nitrogen to produce the desired product. The preparation process requires attention to the control of reaction conditions, and proper preparation and experimental operations.
Safety Information:
Fmoc-(S)-3-Amino-4-(2,4,5-trifluoro-phenyl)-butyric acid is a relatively safe compound under normal operating conditions. However, because it is still an organic compound, it is still necessary to comply with laboratory safety practices. This includes wearing appropriate protective gloves, goggles, and laboratory coats to avoid inhalation or contact with the compound. It should be used in well-ventilated laboratory conditions and following proper waste disposal procedures. In the event of any accident, appropriate emergency measures should be taken immediately and medical assistance should be sought. The Safety Technical Instructions (SDS) should also be read carefully before performing any experiments.
Nature:
-Molecular formula: C19H17F3NO4
-Molecular weight: 389.34g/mol
-Appearance: White crystalline solid
-Melting point: 176-180°C
-Solubility: Soluble in organic solvents (such as dichloromethane, dimethylformamide), insoluble in water
Use:
Fmoc-(S)-3-Amino-4-(2,4,5-trifluoro-phenyl)-butyric acid is an oximine-protected amino acid, which is commonly used in peptide synthesis in chemical synthesis. Its main uses include solid phase synthesis, peptide synthesis, drug development and biomedical fields.
Preparation Method:
The preparation of Fmoc-(S)-3-Amino-4-(2,4,5-trifluoro-phenyl)-butyric acid usually requires a multi-step reaction, and the specific steps may vary depending on experimental conditions. A common method of preparation is to react (S)-3-amino -4-(2,4, 5-trifluorophenyl) butanoic acid with Fmoc-Cl under nitrogen to produce the desired product. The preparation process requires attention to the control of reaction conditions, and proper preparation and experimental operations.
Safety Information:
Fmoc-(S)-3-Amino-4-(2,4,5-trifluoro-phenyl)-butyric acid is a relatively safe compound under normal operating conditions. However, because it is still an organic compound, it is still necessary to comply with laboratory safety practices. This includes wearing appropriate protective gloves, goggles, and laboratory coats to avoid inhalation or contact with the compound. It should be used in well-ventilated laboratory conditions and following proper waste disposal procedures. In the event of any accident, appropriate emergency measures should be taken immediately and medical assistance should be sought. The Safety Technical Instructions (SDS) should also be read carefully before performing any experiments.
Last Update:2024-04-09 21:04:16
Supplier List
Product Name: FMOC-(S)-3-AMINO-4-(2,4,5-TRIFLUORO-PHENYL)-BUTYRIC ACID Request for quotation
CAS: 959580-94-0
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 959580-94-0
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: FMOC-(S)-3-AMINO-4-(2,4,5-TRIFLUORO-PHENYL)-BUTYRIC ACID Visit Supplier Webpage Request for quotationCAS: 959580-94-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: FMOC-(S)-3-AMINO-4-(2,4,5-TRIFLUORO-PHENYL)-BUTYRIC ACID Request for quotation
CAS: 959580-94-0
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 959580-94-0
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: FMOC-(S)-3-AMINO-4-(2,4,5-TRIFLUORO-PHENYL)-BUTYRIC ACID Visit Supplier Webpage Request for quotationCAS: 959580-94-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
View History
N-Fmoc-(S)-2,4,5-trifluoro-b-homophenylalanine
Heptanedinitrile, 2,6-bis(3,4-dimethoxyphenyl)-2,6-bis(1-methylethyl)-
Heptanedinitrile, 2,6-bis(3,4-dimethoxyphenyl)-2,6-bis(1-methylethyl)-

