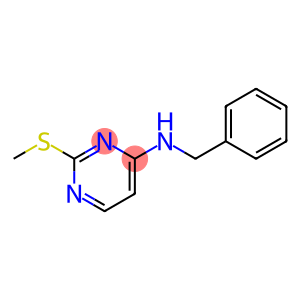
N-Benzyl-2-(methylthio)pyrimidin-4-amine
N-Benzyl-2-(methylthio)pyrimidin-4-amine
CAS: 91719-61-8
Molecular Formula: C12H13N3S
N-Benzyl-2-(methylthio)pyrimidin-4-amine - Names and Identifiers
| Name | N-Benzyl-2-(methylthio)pyrimidin-4-amine |
| Synonyms | N-Benzyl-2-(methylthio)pyrimidin-4-amine Benzyl-(2-Methylsulfanyl-Pyrimidin-4-Yl)-Amine BENZYL-(2-METHYLSULFANYL-PYRIMIDIN-4-YL)-AMINE 4-Pyrimidinamine, 2-(methylthio)-N-(phenylmethyl)- |
| CAS | 91719-61-8 |
| EINECS | 205-525-8 |
| InChI | InChI=1/C12H13N3S/c1-16-12-13-8-7-11(15-12)14-9-10-5-3-2-4-6-10/h2-8H,9H2,1H3,(H,13,14,15) |
N-Benzyl-2-(methylthio)pyrimidin-4-amine - Physico-chemical Properties
| Molecular Formula | C12H13N3S |
| Molar Mass | 231.32 |
| Storage Condition | 2-8°C |
| Refractive Index | 1.632 |
N-Benzyl-2-(methylthio)pyrimidin-4-amine - Risk and Safety
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. |
| HS Code | 29335990 |
N-Benzyl-2-(methylthio)pyrimidin-4-amine - Introduction
N-Benzyl-2-(methylthio)pyrimidin-4-amine is an organic compound with the chemical formula C13H12N4S.
Its nature mainly includes the following aspects:
1. appearance: pure product is white or white crystal powder.
2. melting point: about 180-190 degrees Celsius.
3. Solubility: high solubility in common organic solvents, such as methanol, chloroform and dimethyl sulfoxide.
This compound is commonly used as an intermediate in the pharmaceutical field and has the following uses:
1. Anti-tumor drugs: Its derivatives usually have anti-tumor activity and can be used to develop anti-cancer drugs.
2. Antibacterial drugs: The compound and its derivatives also show certain potential in antibacterial activity and can be applied to the development of antibacterial drugs.
The preparation method of the compound can be achieved by the following steps:
1. firstly, 2-methylthio -4-benzyl thiolopyrimidine reacts with benzylamine to generate N-Benzyl-2-(methylthio)pyrimidin-4-amine under the action of alkali.
2. After completion of the synthesis, the target product can be obtained by purification methods such as crystallization or column chromatography.
Some safety information needs to be noted when using and storing this compound:
1. Because the compound may produce toxic gases or vapors under certain conditions, it must be operated in a well-ventilated laboratory, wearing protective gloves, glasses and laboratory clothes.
2. Avoid contact with strong oxidants to avoid dangerous reactions.
3. Store in a dry, cool, ventilated place, avoid direct sunlight.
Please note that the information provided above is for reference only. Laboratory safety regulations and relevant regulations should be followed when handling and using this compound.
Its nature mainly includes the following aspects:
1. appearance: pure product is white or white crystal powder.
2. melting point: about 180-190 degrees Celsius.
3. Solubility: high solubility in common organic solvents, such as methanol, chloroform and dimethyl sulfoxide.
This compound is commonly used as an intermediate in the pharmaceutical field and has the following uses:
1. Anti-tumor drugs: Its derivatives usually have anti-tumor activity and can be used to develop anti-cancer drugs.
2. Antibacterial drugs: The compound and its derivatives also show certain potential in antibacterial activity and can be applied to the development of antibacterial drugs.
The preparation method of the compound can be achieved by the following steps:
1. firstly, 2-methylthio -4-benzyl thiolopyrimidine reacts with benzylamine to generate N-Benzyl-2-(methylthio)pyrimidin-4-amine under the action of alkali.
2. After completion of the synthesis, the target product can be obtained by purification methods such as crystallization or column chromatography.
Some safety information needs to be noted when using and storing this compound:
1. Because the compound may produce toxic gases or vapors under certain conditions, it must be operated in a well-ventilated laboratory, wearing protective gloves, glasses and laboratory clothes.
2. Avoid contact with strong oxidants to avoid dangerous reactions.
3. Store in a dry, cool, ventilated place, avoid direct sunlight.
Please note that the information provided above is for reference only. Laboratory safety regulations and relevant regulations should be followed when handling and using this compound.
Last Update:2024-04-10 22:40:09
Supplier List
Product Name: BENZYL-(2-METHYLSULFANYL-PYRIMIDIN-4-YL)-AMINE Request for quotation
CAS: 91719-61-8
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 91719-61-8
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: BENZYL-(2-METHYLSULFANYL-PYRIMIDIN-4-YL)-AMINE Visit Supplier Webpage Request for quotation
CAS: 91719-61-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 91719-61-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: BENZYL-(2-METHYLSULFANYL-PYRIMIDIN-4-YL)-AMINE Request for quotation
CAS: 91719-61-8
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 91719-61-8
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: BENZYL-(2-METHYLSULFANYL-PYRIMIDIN-4-YL)-AMINE Visit Supplier Webpage Request for quotation
CAS: 91719-61-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 91719-61-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History

