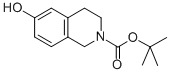
N-BOC-6-HYDROXY-3,4-DIHYDRO-ISOQUINOLINE
TERT-BUTYL 6-HYDROXY-3,4-DIHYDROISOQUINOLINE-2(1H)-CARBOXYLATE
CAS: 158984-83-9
Molecular Formula: C14H19NO3
N-BOC-6-HYDROXY-3,4-DIHYDRO-ISOQUINOLINE - Names and Identifiers
N-BOC-6-HYDROXY-3,4-DIHYDRO-ISOQUINOLINE - Physico-chemical Properties
| Molecular Formula | C14H19NO3 |
| Molar Mass | 249.31 |
| Density | 1.170±0.06 g/cm3(Predicted) |
| Melting Point | 110-111.5 °C |
| Boling Point | 391.4±42.0 °C(Predicted) |
| pKa | 9.79±0.20(Predicted) |
| Storage Condition | 2-8°C |
| Use | This product is for scientific research only and shall not be used for other purposes. |
N-BOC-6-HYDROXY-3,4-DIHYDRO-ISOQUINOLINE - Risk and Safety
| Hazard Class | IRRITANT |
N-BOC-6-HYDROXY-3,4-DIHYDRO-ISOQUINOLINE - Reference Information
| Use | N-BOC-6-hydroxy -3, 4-dihydroisoquinoline is a pharmaceutical intermediate, and it has been reported in the literature that it can be used to prepare SMO antagonists. |
| Preparation | N-BOC-6-hydroxy -3, 4-dihydroisoquinoline can be 1,2,3, 4-tetrahydroisoquinoline-6-ol and ditert-butyl dicarbonate are prepared by one-step reaction. 1,2,3, 4-tetrahydroisoquinolin-6-ol (2.0g,13.4mmol) and 4-dimethylaminopyridine (24mg) were added to tetrahydrofuran (60mL), ditert-butyl dicarbonate (3.07mL,13.4mmol) were added dropwise in an ice bath, and then stirred at room temperature for half an hour. After the reaction, spin the solvent under reduced pressure, add dichloromethane for dilution, then wash with saturated sodium bicarbonate solution, the organic layer is dried by anhydrous sodium sulfate, then concentrated under reduced pressure, and separated and purified by column chromatography (petroleum ether: ethyl acetate = 5:1) to obtain white oily N-BOC-6-hydroxy -3, 4-dihydroisoquinoline (2.34g,70%). |
Last Update:2024-04-09 01:59:59
Supplier List
Spot supply
Product Name: tert-Butyl 6-hydroxy-3,4-dihydroisoquinoline-2(1H)-carboxylate Visit Supplier Webpage Request for quotationCAS: 158984-83-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: TERT-BUTYL 6-HYDROXY-3,4-DIHYDROISOQUINOLINE-2(1H)-CARBOXYLATE Request for quotation
CAS: 158984-83-9
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 158984-83-9
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: TERT-BUTYL 6-HYDROXY-3,4-DIHYDROISOQUINOLINE-2(1H)-CARBOXYLATE Request for quotation
CAS: 158984-83-9
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 158984-83-9
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: TERT-BUTYL 6-HYDROXY-3,4-DIHYDROISOQUINOLINE-2(1H)-CARBOXYLATE Visit Supplier Webpage Request for quotation
CAS: 158984-83-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 158984-83-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: tert-Butyl 6-hydroxy-3,4-dihydroisoquinoline-2(1H)-carboxylate Visit Supplier Webpage Request for quotationCAS: 158984-83-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: TERT-BUTYL 6-HYDROXY-3,4-DIHYDROISOQUINOLINE-2(1H)-CARBOXYLATE Request for quotation
CAS: 158984-83-9
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 158984-83-9
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: TERT-BUTYL 6-HYDROXY-3,4-DIHYDROISOQUINOLINE-2(1H)-CARBOXYLATE Request for quotation
CAS: 158984-83-9
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 158984-83-9
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: TERT-BUTYL 6-HYDROXY-3,4-DIHYDROISOQUINOLINE-2(1H)-CARBOXYLATE Visit Supplier Webpage Request for quotation
CAS: 158984-83-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 158984-83-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



