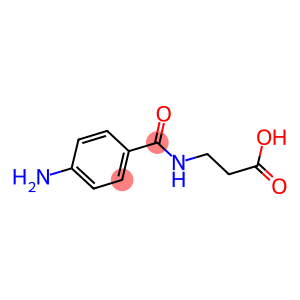
N-4-(Aminobenzoyl)-beta-Alanine
N-4-(Aminobenzoyl)-beta-Alanine
CAS: 7377-08-4
Molecular Formula: C10H12N2O3
N-4-(Aminobenzoyl)-beta-Alanine - Names and Identifiers
| Name | N-4-(Aminobenzoyl)-beta-Alanine |
| Synonyms | TIMTEC-BB SBB000472 Timtec-Bb Sbb000472 n-(4-aminobenzoyl)-alanine n-(4-aminobenzoyl)--alanine N-(4-Aminobenzoyl)--alanine n-(4-aminobenzoyl)-á-alanine N-(4-Aminobenzoyl)-b-alanine N-(4-Aminobenzoyl)-B-Alanine N-(4-Aminobenzoyl)-Ss-Alanine N-(P-Aminobenzoyl)-Beta-Alanine N-(4-AMINOBENZOYL)-BETA-ALANINE N-(4-Aminobenzoyl)-Beta-Alanine N-4-(Aminobenzoyl)-beta-Alanine 3-(4-Aminobenzoylamino)-Propionic Acid 3-(4-AMINOBENZOYLAMINO)-PROPIONIC ACID 3-{[(4-aminophenyl)carbonyl]amino}propanoate |
| CAS | 7377-08-4 |
| EINECS | 616-017-7 |
| InChI | InChI=1/C10H12N2O3/c11-8-3-1-7(2-4-8)10(15)12-6-5-9(13)14/h1-4H,5-6,11H2,(H,12,15)(H,13,14)/p-1 |
| InChIKey | VHAXWROFYVPXMZ-UHFFFAOYSA-N |
N-4-(Aminobenzoyl)-beta-Alanine - Physico-chemical Properties
| Molecular Formula | C10H12N2O3 |
| Molar Mass | 208.21 |
| Density | 1.2420 (rough estimate) |
| Melting Point | 152-154°C(lit.) |
| Boling Point | 347.41°C (rough estimate) |
| Flash Point | 267.9°C |
| Solubility | DMSO, Methanol (Slightly), Water (Slightly, Heated) |
| Vapor Presure | 0Pa at 20℃ |
| Appearance | Solid |
| Color | White to Off-White |
| pKa | 4.36±0.10(Predicted) |
| Storage Condition | under inert gas (nitrogen or Argon) at 2–8 °C |
| Refractive Index | 1.4830 (estimate) |
| MDL | MFCD00009805 |
N-4-(Aminobenzoyl)-beta-Alanine - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S37/39 - Wear suitable gloves and eye/face protection |
| WGK Germany | 3 |
N-4-(Aminobenzoyl)-beta-Alanine - Reference Information
| LogP | -1.19 at 25℃ and pH7.5 |
| surface tension | 71.6mN/m at 1g/L and 20 ℃ |
| Use | p-Aminobenzoyl-beta-alanine is an organic intermediate, it can be prepared from β-alanine as a starting material by a two-step reaction. It has been reported in the literature that it can be used for the preparation of sodium balsalazide. Balsalazide sodium is a prodrug, after oral administration of the original drug reaches the colon, under the action of colonic bacteria release 5-aminosalicylic acid (active ingredient) and 4-aminobenzoyl-beta-alanine. 5-aminosalicylic acid may play its role in reducing inflammation by blocking the production of arachidonic acid metabolites in the colon, mainly used for ulcerative colon salt and mild to moderate acute and chronic colitis. Treatment of mild to moderate active ulcerative colitis. |
| preparation | Step 1: 4-nitrobenzoyl-β-alanine sodium salt in a 500ml three-necked flask, then, β-alanine (5.7g,64.86mmol),20g of sodium hydroxide and 200g of an aqueous sodium hydroxide solution prepared from purified water were successively added and dissolved with stirring. The reaction temperature was lowered to 5 °c and 4-nitrobenzoyl chloride (a small number of times, 10g,54.05mmol) was added. The reaction was carried out for 3H while maintaining the reaction temperature at 5 °c. The aqueous solution of the target product was obtained, and the next step was directly applied without post-treatment. Step 2: P-Aminobenzoyl-beta-alanine was added to a Zr12-TPDC-Pd ml three-necked flask of the upper reaction aqueous solution, H2 was introduced, and the mixture was stirred at room temperature and normal pressure until no hydrogen was absorbed. Hydrochloric acid was then slowly added to adjust PH = 1-2 and then stirred for 10 minutes to give a solution of p-Aminobenzoyl-beta-alanine. |
Last Update:2024-04-09 20:52:54
Supplier List
Spot supply
Product Name: N-(4-Aminobenzoyl)-β-alanine Visit Supplier Webpage Request for quotationCAS: 7377-08-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: N-(4-Aminobenzoyl)-beta-alanine Request for quotation
CAS: 7377-08-4
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 7377-08-4
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Multiple Specifications
Product Name: N-(4-Aminobenzoyl)-β-alanine Visit Supplier Webpage Request for quotationCAS: 7377-08-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: N-(4-Aminobenzoyl)-β-alanine Visit Supplier Webpage Request for quotationCAS: 7377-08-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: N-(4-Aminobenzoyl)-beta-alanine Request for quotation
CAS: 7377-08-4
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 7377-08-4
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Multiple Specifications
Product Name: N-(4-Aminobenzoyl)-β-alanine Visit Supplier Webpage Request for quotationCAS: 7377-08-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


