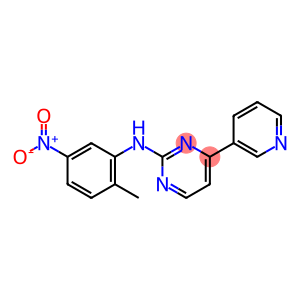
N-(2-methyl-5-nitrophenyl)-4-(3-pyridinyl)-2-pydimidineamine
N-(2-Methyl-5-nitrophenyl)-4-(pyridin-3-yl)pyrimidin-2-amine
CAS: 152460-09-8
Molecular Formula: C16H13N5O2
N-(2-methyl-5-nitrophenyl)-4-(3-pyridinyl)-2-pydimidineamine - Names and Identifiers
N-(2-methyl-5-nitrophenyl)-4-(3-pyridinyl)-2-pydimidineamine - Physico-chemical Properties
| Molecular Formula | C16H13N5O2 |
| Molar Mass | 307.31 |
| Density | 1.344±0.06 g/cm3(Predicted) |
| Melting Point | 188-193°C |
| Boling Point | 546.6±60.0 °C(Predicted) |
| Appearance | Solid |
| Color | Yellow |
| pKa | 2.68±0.12(Predicted) |
| Storage Condition | Keep in dark place,Inert atmosphere,Room temperature |
| Physical and Chemical Properties | N-(2-methyl-5-nitrophenyl)-4-(3-pyridyl)-2-pyrimidinamine has a melting point of 188-193°C and a boiling point of 546.6±60.0°C(Predicted), the density was 1.344±0.06g/cm3(Predicted, and the acidity coefficient (pKa) was 2.68±0.12(Predicted). |
N-(2-methyl-5-nitrophenyl)-4-(3-pyridinyl)-2-pydimidineamine - Reference Information
| Uses | N-(2-methyl-5-nitrophenyl)-4-(3-pyridyl)-2-pyrimidamines are a very important class of heterocyclic compounds, widely used in biology, medicine and agriculture and other fields. Studies have shown that the compound has good biological activities, such as sterilization, insecticidal, weeding, anti-virus, anti-cancer, etc. It is also used as a precursor for the synthesis of some important anti-cancer drugs, such as imatinib, which is a selective casinic acid kinase inhibitor mainly used in the treatment of chronic spinal leukemia and gastrointestinal stromal tumors. |
| Synthesis method | Step 1: The preparation of 1-(2-methyl -5-nitrophenyl) guanidinyl -2-methyl -5-nitroaniline-(50g,0.328 mol) and n-butanol (200ml) is placed in a reaction bottle. 35% concentrated hydrochloric acid (19ml) was slowly added to the reaction mass for 15 minutes. Stir the reactants for 15 minutes, and slowly add 50% aqueous cyanamide (82.3ml,1.978 moles) to the reactants for 15 minutes. The reaction mixture was heated to 90-95°C and stirred at the same temperature for four hours, and concentrated hydrochloric acid (19ml) was slowly added drop by drop within 15 minutes. The reaction mixture was stirred further for four hours while maintaining the temperature at 90-95°C. Concentrated hydrochloric acid (29ml) was added drop by drop, and the reaction mixture was kept at 90°C for 4 hours. The reaction mass was maintained at the same temperature for a total of 20 hours. The auxiliary reaction completed the reaction mass was cooled to 10°C and alkalized with 10% aqueous sodium hydroxide solution (400ml). The solid product was filtered, washed with 200ml of water and dried to give 1-(2-methyl-5-nitrophenyl) guanidine. Yield (63g,98.7%). HPLC purity: 99% Step 2: N-(5-nitro-2-methylphenyl)-4-(3-pyridyl)-2-pyrimidine -1-(2-methyl-5-nitrophenyl) guanidine (63g, the preparation of 0.324 mol), 3-dimethylamino-1-(3-pyridine)-2-propen-1-one (62.5g,0.357) and n-butanol (560ml) was placed in a reaction bottle. The reaction mixture was heated to 120°C for 9 hours. The reaction mass was adjusted to room temperature, water (450ml) was added, and stirred at room temperature for 3-4 hours. The precipitated solids were separated by filtration and dried to give N-(2-methyl-5-nitrophenyl)-4-(3-pyridyl)-2-pyrimidinamine. Yield (73.7g, 81.9%). HPLC purity: 99.9%. Figure N-(2-methyl-5-nitrophenyl)-4-(3-pyridyl)-2-pyrimidinamine synthesis route. |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: N-(2-Methyl-5-nitrophenyl)-4-(pyridin-3-yl)pyrimidin-2-amine Visit Supplier Webpage Request for quotationCAS: 152460-09-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Imatinib Impurity 14 Visit Supplier Webpage Request for quotationCAS: 152460-09-8
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: N-(2-Methyl-5-nitrophenyl)-4-(pyridin-3-yl)pyrimidin-2-amine Request for quotation
CAS: 152460-09-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 152460-09-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: N-(2-Methyl-5-nitrophenyl)-4-(pyridin-3-yl)pyrimidin-2-amine Visit Supplier Webpage Request for quotationCAS: 152460-09-8
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Product Name: N-(2-Methyl-5-nitrophenyl)-4-(pyridin-3-yl)pyrimidin-2-amine Request for quotation
CAS: 152460-09-8
Tel: +86 19849939632
Email: 2470479589@qq.com
Mobile: +86 19849939632
QQ: 2470479589
Wechat: +86 19849939632
CAS: 152460-09-8
Tel: +86 19849939632
Email: 2470479589@qq.com
Mobile: +86 19849939632
QQ: 2470479589
Wechat: +86 19849939632
Product Name: N-(2-Methyl-5-nitrophenyl)-4-(pyridin-3-yl)pyrimidin-2-amine Request for quotation
CAS: 152460-09-8
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 152460-09-8
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Spot supply
Product Name: N-(2-Methyl-5-Nitrophenyl)-4-(Pyridin-3-yl)Pyrimidin-2-Amine Visit Supplier Webpage Request for quotationCAS: 152460-09-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: N-(2-Methyl-5-nitrophenyl)-4-(pyridin-3-yl)pyrimidin-2-amine Visit Supplier Webpage Request for quotationCAS: 152460-09-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Imatinib Impurity 14 Visit Supplier Webpage Request for quotationCAS: 152460-09-8
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: N-(2-Methyl-5-nitrophenyl)-4-(pyridin-3-yl)pyrimidin-2-amine Request for quotation
CAS: 152460-09-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 152460-09-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: N-(2-Methyl-5-nitrophenyl)-4-(pyridin-3-yl)pyrimidin-2-amine Visit Supplier Webpage Request for quotationCAS: 152460-09-8
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Product Name: N-(2-Methyl-5-nitrophenyl)-4-(pyridin-3-yl)pyrimidin-2-amine Request for quotation
CAS: 152460-09-8
Tel: +86 19849939632
Email: 2470479589@qq.com
Mobile: +86 19849939632
QQ: 2470479589
Wechat: +86 19849939632
CAS: 152460-09-8
Tel: +86 19849939632
Email: 2470479589@qq.com
Mobile: +86 19849939632
QQ: 2470479589
Wechat: +86 19849939632
Product Name: N-(2-Methyl-5-nitrophenyl)-4-(pyridin-3-yl)pyrimidin-2-amine Request for quotation
CAS: 152460-09-8
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 152460-09-8
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Spot supply
Product Name: N-(2-Methyl-5-Nitrophenyl)-4-(Pyridin-3-yl)Pyrimidin-2-Amine Visit Supplier Webpage Request for quotationCAS: 152460-09-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History






