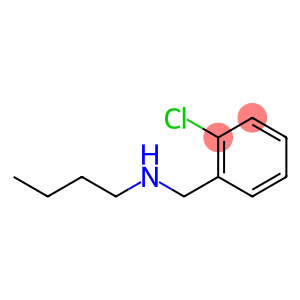
N-(2-CHLOROBENZYL)BUTAN-1-AMINE
N-(2-chlorobenzyl)butan-1-amine
CAS: 16183-39-4
Molecular Formula: C11H16ClN
N-(2-CHLOROBENZYL)BUTAN-1-AMINE - Names and Identifiers
| Name | N-(2-chlorobenzyl)butan-1-amine |
| Synonyms | Aids107179 Aids-107179 UKRORGSYN-BB BBV-125477 16183-40-7 (Hydrochloride) N-(2-CHLOROBENZYL)BUTAN-1-AMINE N-(2-chlorobenzyl)butan-1-amine N-(2-CHLOROPHENYLMETHYL)BUTYLAMINE Benzenemethanamine, N-butyl-2-chloro- benzenemethanamine, N-butyl-2-chloro- |
| CAS | 16183-39-4 |
| InChI | InChI=1/C11H16ClN/c1-2-3-8-13-9-10-6-4-5-7-11(10)12/h4-7,13H,2-3,8-9H2,1H3 |
N-(2-CHLOROBENZYL)BUTAN-1-AMINE - Physico-chemical Properties
| Molecular Formula | C11H16ClN |
| Molar Mass | 197.7 |
| Density | 1.026±0.06 g/cm3(Predicted) |
| Boling Point | 264.2±15.0 °C(Predicted) |
| Flash Point | 113.6°C |
| Vapor Presure | 0.00984mmHg at 25°C |
| pKa | 9.11±0.19(Predicted) |
| Storage Condition | Room Temprature |
| Refractive Index | 1.516 |
N-(2-CHLOROBENZYL)BUTAN-1-AMINE - Introduction
N-(2-chlorobenzyl)butan-1-amine is an organic compound with the chemical formula C10H15ClN, commonly abbreviated as BBAlamine. The following is a description of the nature, use, preparation and safety information of N-(2-chlorobenzyl)butan-1-amine:
Nature:
N-(2-chlorobenzyl)butan-1-amine is a colorless or pale yellow liquid with a fragrant odor. It has a relatively low density and is soluble in many organic solvents, such as ethanol, ether and acetone. It is stable to air and light, and relatively stable at room temperature.
Use:
N-(2-chlorobenzyl)butan-1-amine is a common organic synthesis reagent with strong ortho-selectivity. It can be used in the synthesis of various amino-containing compounds, such as amino acids and amino acid esters. In addition, it can also be used to prepare other organic compounds, such as pesticides, drugs and dyes.
Method:
N-(2-chlorobenzyl)butan-1-amine can react acrylonitrile and chlorobenzyl magnesium iodide to generate the corresponding tertiary propylamine, and then introduce butylamine into tertiary propylamine through N-alkylation reaction, finally, the chlorine atom is replaced by a benzyl group.
Safety Information:
N-(2-chlorobenzyl)butan-1-amine is an organic compound and must be used in accordance with the appropriate safety procedures. It is irritating to the eyes and skin, and personal protective equipment such as gloves, goggles and protective clothing should be used. Avoid inhaling its vapor during operation and use it in a well-ventilated place. In addition, it is also a flammable liquid and should be kept away from open flames and high temperature sources.
Nature:
N-(2-chlorobenzyl)butan-1-amine is a colorless or pale yellow liquid with a fragrant odor. It has a relatively low density and is soluble in many organic solvents, such as ethanol, ether and acetone. It is stable to air and light, and relatively stable at room temperature.
Use:
N-(2-chlorobenzyl)butan-1-amine is a common organic synthesis reagent with strong ortho-selectivity. It can be used in the synthesis of various amino-containing compounds, such as amino acids and amino acid esters. In addition, it can also be used to prepare other organic compounds, such as pesticides, drugs and dyes.
Method:
N-(2-chlorobenzyl)butan-1-amine can react acrylonitrile and chlorobenzyl magnesium iodide to generate the corresponding tertiary propylamine, and then introduce butylamine into tertiary propylamine through N-alkylation reaction, finally, the chlorine atom is replaced by a benzyl group.
Safety Information:
N-(2-chlorobenzyl)butan-1-amine is an organic compound and must be used in accordance with the appropriate safety procedures. It is irritating to the eyes and skin, and personal protective equipment such as gloves, goggles and protective clothing should be used. Avoid inhaling its vapor during operation and use it in a well-ventilated place. In addition, it is also a flammable liquid and should be kept away from open flames and high temperature sources.
Last Update:2024-04-09 20:52:54
Supplier List
Product Name: CHEMBRDG-BB 4024926 Visit Supplier Webpage Request for quotation
CAS: 16183-39-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 16183-39-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: CHEMBRDG-BB 4024926 Visit Supplier Webpage Request for quotation
CAS: 16183-39-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 16183-39-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History
