ML380
ML380
CAS: 1627138-52-6
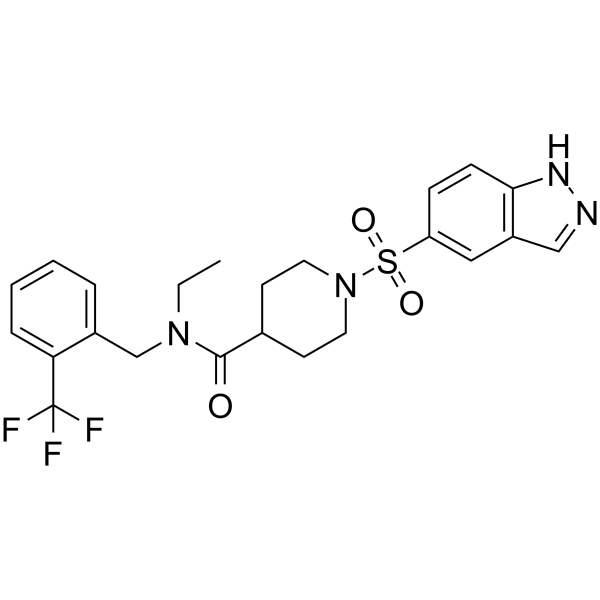
Molecular Formula: C23H25F3N4O3S
ML380 - Names and Identifiers
| Name | ML380 |
| Synonyms | ML380 ML380 ML-380 ML 380 N-ethyl-1-(1H-indazol-5-ylsulfonyl)-N-[[2-(trifluoromethyl)phenyl]methyl]piperidine-4-carboxamide |
| CAS | 1627138-52-6 |
ML380 - Physico-chemical Properties
| Molecular Formula | C23H25F3N4O3S |
| Molar Mass | 494.5298096 |
| Solubility | Soluble in DMSO |
| Storage Condition | -20℃ |
| Use | ML380 is a potent, subtype-selective, and brain-penetrant positive allosteric modulator (PAM) of M5 mAChR, with EC50s of 190 and 610 nM for human and rat M5, respectively. ML380 exhibits moderate selectivity versus the M1 and M3 mAChR subtypes. ML380 could increase the affinity of ACh for the M5 mAChR. |
| Target | IC50: 190 nM (hM5), 610 nM (rM5) |
| In vitro study | ML380 (0.01 nM-100 μM) robustly stimulates inositol phosphate (IP) accumulation and Ca 2+ mobilization in CHO-hM 5 cells, with pEC 50 s of 5.33 and 5.71, respectively. ML380 (0.01-30 μM) increases the ACh-stimulated IP accumulation and Ca 2+ mobilization in CHO-hM 5 cells. |
| In vivo study | ML380 (1 mg/kg; i.v.) displays high clearance (66 mL/min/kg), a moderate volume of distribution (1.6 L/kg), and a short half-life (t 1/2 , 22 min) in rats. |
ML380 - Reference
| Reference Show more | 1: Berizzi AE, Gentry PR, Rueda P, Den Hoedt S, Sexton PM, Langmead CJ, Christopoulos A. Molecular Mechanisms of Action of M5 Muscarinic Acetylcholine Receptor Allosteric Modulators. Mol Pharmacol. 2016 Oct;90(4):427-36. doi: 10.1124/mol.116.104182. PubMed PMID: 27461343. 2: Gentry PR, Kokubo M, Bridges TM, Daniels JS, Niswender CM, Smith E, Chase P, Hodder PS, Rosen H, Conn PJ, Engers J, Brewer KA, Lindsley CW, Wood MR. Development of the First CNS penetrant M5 Positive Allosteric Modulator (PAM) Based on a Novel, non-Isatin Core. 2013 Dec 15 [updated 2015 Feb 11]. Probe Reports from the NIH Molecular Libraries Program [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2010-. Available from http://www.ncbi.nlm.nih.gov/books/NBK280054/ PubMed PMID: 25834908. 3: Gentry PR, Kokubo M, Bridges TM, Noetzel MJ, Cho HP, Lamsal A, Smith E, Chase P, Hodder PS, Niswender CM, Daniels JS, Conn PJ, Lindsley CW, Wood MR. Development of a highly potent, novel M5 positive allosteric modulator (PAM) demonstrating CNS exposure: 1-((1H-indazol-5-yl)sulfoneyl)-N-ethyl-N-(2-(trifluoromethyl)benzyl)piperidine-4- carboxamide (ML380). J Med Chem. 2014 Sep 25;57(18):7804-10. doi: 10.1021/jm500995y. PubMed PMID: 25147929; PubMed Central PMCID: PMC4175000. |
ML380 - Preparation solution concentration reference
| 1mg | 5mg | 10mg | |
|---|---|---|---|
| 1 mM | 2.022 ml | 10.111 ml | 20.221 ml |
| 5 mM | 0.404 ml | 2.022 ml | 4.044 ml |
| 10 mM | 0.202 ml | 1.011 ml | 2.022 ml |
| 5 mM | 0.04 ml | 0.202 ml | 0.404 ml |
Last Update:2024-01-02 23:10:35
ML380 - Reference Information
| biological activity | ML380 is an effective, subtype-selective and blood-brain barrier permeable M5 mAChR positive allosteric regulator (PAM). The EC50 values for human and rat M5 are 190 and 610 nM respectively. ML380 is moderately selective for M1 and M3 mAChR subtypes. ML380 can increase Ach's affinity for M5 mAChR. |
| target | IC50: 190 nM (hM5), 610 nM (rM5) |
| in vitro study | ML380 (0.01 nM-100 μM) robustly stimulates inositol phosphate (IP) accumulation and Ca 2 mobilization in CHO-hM 5 cells, with pEC 50 s of 5.33 and 5.71, respectively. ML380 (0.01-30 μM) increases the ACh-stimulated IP accumulation and Ca 2 mobilization in CHO-hM 5 cells. |
| in vivo study | ML380 (1 mg/kg; I. v.) displays high clearance (66 mL/min/kg), a moderate volume of distribution (1.6 L/kg), and a short half-life (t 1/2 , 22 min) in rats. |
Last Update:2024-04-09 21:01:54
Supplier List
Spot supply
Product Name: ML380 Visit Supplier Webpage Request for quotationCAS: 1627138-52-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: N-ethyl-1-(1H-indazol-5-ylsulfonyl)-N-[[2-(trifluoromethyl)phenyl]methyl]piperidine-4-carboxamide Request for quotation
CAS: 1627138-52-6
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 1627138-52-6
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Spot supply
Product Name: ML380 Visit Supplier Webpage Request for quotationCAS: 1627138-52-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: ML380 Visit Supplier Webpage Request for quotationCAS: 1627138-52-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: N-ethyl-1-(1H-indazol-5-ylsulfonyl)-N-[[2-(trifluoromethyl)phenyl]methyl]piperidine-4-carboxamide Request for quotation
CAS: 1627138-52-6
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 1627138-52-6
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Spot supply
Product Name: ML380 Visit Supplier Webpage Request for quotationCAS: 1627138-52-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


