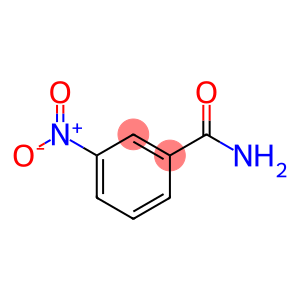
M-NITROBENZAMIDE
3-Nitrobenzamide
CAS: 645-09-0
Molecular Formula: C7H6N2O3
M-NITROBENZAMIDE - Names and Identifiers
| Name | 3-Nitrobenzamide |
| Synonyms | NSC 37327 BRN 0777185 3-Nitrobenzamide m-Nitrobenzamide N-nitrobenzamide 3-nitro-benzamid m-nitro-benzamid 3-NITROBENZAMIDE M-NITROBENZAMIDE Benzamide, 3-nitro- Benzamide, m-nitro- Benzamide, m-nitro- (8CI) 3-nitrobenzenecarboximidic acid 4-09-00-01061 (Beilstein Handbook Reference) |
| CAS | 645-09-0 |
| EINECS | 211-431-0 |
| InChI | InChI=1/C7H6N2O3/c8-7(10)5-2-1-3-6(4-5)9(11)12/h1-4H,(H2,8,10) |
M-NITROBENZAMIDE - Physico-chemical Properties
| Molecular Formula | C7H6N2O3 |
| Molar Mass | 166.13 |
| Density | 1.4994 (rough estimate) |
| Melting Point | 140-143 °C (lit.) |
| Boling Point | 312.55°C |
| Flash Point | 145.852°C |
| Vapor Presure | 0mmHg at 25°C |
| Appearance | Crystalline Powder or Crystals |
| Color | Yellow |
| BRN | 777185 |
| pKa | 14.86±0.50(Predicted) |
| Storage Condition | Sealed in dry,Room Temperature |
| Stability | Stable. Combustible. Incompatible with strong oxidizing agents. |
| Refractive Index | 1.5880 (estimate) |
M-NITROBENZAMIDE - Risk and Safety
| Risk Codes | R22 - Harmful if swallowed R36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S24/25 - Avoid contact with skin and eyes. S36/37 - Wear suitable protective clothing and gloves. S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. |
| WGK Germany | 3 |
| RTECS | CV5601250 |
| TSCA | Yes |
| HS Code | 29242990 |
M-NITROBENZAMIDE - Reference Information
| EPA chemical substance information | information provided by: ofmpeb.epa.gov (external link) |
| Overview | 3-nitrobenzamide, also known as M-nitrobenzamide, is an organic intermediate, it can be prepared from 3-nitrobenzaldehyde or m-nitrobenzonitrile. |
| preparation | The general method is as follows: the synthesis of 3-nitrobenzamide refers to the following synthesis of 4-nitrophenol. To a solution of phenol (1mmol) in dichloromethane was added 69% nitric acid (1mmol) and 1mmol transition metal complex ([Co(NH3)5Cl] Cl2), the reaction mixture was stirred at room temperature for 2 hours. The progress of the reaction was monitored by TLC. After completion of the reaction as indicated by TLC, the reaction mixture was treated with sodium carbonate solution. the reaction mixture was extracted with dichloromethane. The organic layer was separated, dried over sodium sulfate and evaporated to give the crude product. The crude product was purified on silica gel to give 4-nitrophenol as the product. 3-nitrobenzamide, yield 82%. |
Last Update:2024-04-09 02:00:10
Supplier List
Spot supply
Product Name: 3-Nitrobenzamide Visit Supplier Webpage Request for quotationCAS: 645-09-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-NITROBENZAMIDE Request for quotation
CAS: 645-09-0
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 645-09-0
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: 3-Nitrobenzamide Request for quotation
CAS: 645-09-0
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 645-09-0
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: 3-Nitrobenzamide Visit Supplier Webpage Request for quotationCAS: 645-09-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-NITROBENZAMIDE Request for quotation
CAS: 645-09-0
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 645-09-0
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: 3-Nitrobenzamide Request for quotation
CAS: 645-09-0
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 645-09-0
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
View History


