L-2-Nitrophe
L-2-Nitrophe
CAS: 19883-75-1
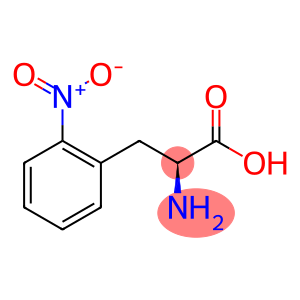
Molecular Formula: C9H10N2O4
L-2-Nitrophe - Names and Identifiers
| Name | L-2-Nitrophe |
| Synonyms | L-2-NITROPHE L-PHE(2-NO2) L-2-Nitrophe H-PHE(2-NO2)-OH H-O-NITRO-PHE-OH 2-nitrophenylalanine L-2-NITROPHENYLALANINE L-2-Nitrophenylalanine 2-nitro-L-phenylalanine 2-NITRO-L-PHENYLALANINE L-2-AMINO-3-(2-NITRO PHENYL)-PROPIONIC ACID (2S)-2-amino-3-(2-nitrophenyl)propanoic acid |
| CAS | 19883-75-1 |
| InChI | InChI=1/C9H10N2O4/c10-7(9(12)13)5-6-3-1-2-4-8(6)11(14)15/h1-4,7H,5,10H2,(H,12,13)/t7-/m0/s1 |
L-2-Nitrophe - Physico-chemical Properties
| Molecular Formula | C9H10N2O4 |
| Molar Mass | 210.19 |
| Density | 1.408 |
| Melting Point | 223°C (dec.) |
| Boling Point | 385.4±32.0 °C(Predicted) |
| Flash Point | 186.9°C |
| Solubility | Aqueous Acid (Slightly), DMSO (Slightly, Heated, Sonicated), Methanol (Slightly) |
| Vapor Presure | 1.25E-06mmHg at 25°C |
| Appearance | Solid |
| Color | White to Off-White |
| pKa | 2.03±0.10(Predicted) |
| Storage Condition | under inert gas (nitrogen or Argon) at 2–8 °C |
| Refractive Index | 1.614 |
L-2-Nitrophe - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. |
L-2-Nitrophe - Introduction
L-2-Nitrophe is an organic compound with the chemical formula C9H10N2O4. It is a derivative of an amino acid with a carboxyl group, an amine group and a 2-nitrophenyl side chain. It exists in two stereoisomers, the L-form and the D-form, L-2-Nitrophe of which is the native form.
L-2-Nitrophe are widely used in the field of medicine, especially in the pharmaceutical industry as intermediates for the synthesis of certain drugs. It is also used as a research tool for biologically active molecules. In addition, it also has antioxidant, anti-inflammatory and anti-tumor activities, so it is also studied for medical treatment.
The method for preparing L-2-Nitrophe can be achieved by a substitution reaction between phenylalanine and nitric acid. First, phenylalanine is reacted with nitric acid to obtain a bitter intermediate, which is then subjected to alkali treatment and reduction reaction to finally obtain the target product.
Regarding safety information, L-2-Nitrophe is a toxic substance and needs to be used with caution. During operation, care should be taken to avoid inhalation, skin contact and ingestion, and to operate under good ventilation conditions. Appropriate personal protective equipment such as gloves, goggles and masks should be worn. Segregate from other chemicals during use and storage and follow safe operating procedures.
To sum up, L-2-Nitrophe is an organic compound widely used in the pharmaceutical field and has the properties of a pharmaceutical intermediate. Its preparation method involves the substitution reaction of nitric acid. Follow safe operating procedures, use with care, and take appropriate personal protective measures.
L-2-Nitrophe are widely used in the field of medicine, especially in the pharmaceutical industry as intermediates for the synthesis of certain drugs. It is also used as a research tool for biologically active molecules. In addition, it also has antioxidant, anti-inflammatory and anti-tumor activities, so it is also studied for medical treatment.
The method for preparing L-2-Nitrophe can be achieved by a substitution reaction between phenylalanine and nitric acid. First, phenylalanine is reacted with nitric acid to obtain a bitter intermediate, which is then subjected to alkali treatment and reduction reaction to finally obtain the target product.
Regarding safety information, L-2-Nitrophe is a toxic substance and needs to be used with caution. During operation, care should be taken to avoid inhalation, skin contact and ingestion, and to operate under good ventilation conditions. Appropriate personal protective equipment such as gloves, goggles and masks should be worn. Segregate from other chemicals during use and storage and follow safe operating procedures.
To sum up, L-2-Nitrophe is an organic compound widely used in the pharmaceutical field and has the properties of a pharmaceutical intermediate. Its preparation method involves the substitution reaction of nitric acid. Follow safe operating procedures, use with care, and take appropriate personal protective measures.
Last Update:2024-04-09 21:01:54
Supplier List
Spot supply
Product Name: (S)-2-Amino-3-(2-nitrophenyl)propanoic acid Visit Supplier Webpage Request for quotationCAS: 19883-75-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: L-2-NITROPHENYLALANINE Request for quotation
CAS: 19883-75-1
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 19883-75-1
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: 2-Nitrophenylalanine Visit Supplier Webpage Request for quotation
CAS: 19883-75-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 19883-75-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: (S)-2-Amino-3-(2-nitrophenyl)propanoic acid Visit Supplier Webpage Request for quotationCAS: 19883-75-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: L-2-NITROPHENYLALANINE Request for quotation
CAS: 19883-75-1
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 19883-75-1
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: 2-Nitrophenylalanine Visit Supplier Webpage Request for quotation
CAS: 19883-75-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 19883-75-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


