Jacesiolin
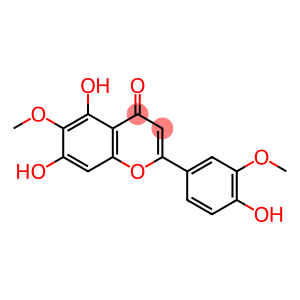
5,7-dihydroxy-2-(4-hydroxy-3-methoxy-phenyl)-6-methoxy-chromen-4-one
CAS: 18085-97-7
Molecular Formula: C17H14O7
Jacesiolin - Names and Identifiers
| Name | 5,7-dihydroxy-2-(4-hydroxy-3-methoxy-phenyl)-6-methoxy-chromen-4-one |
| Synonyms | jaceosidin JACEOSIDIN Jacesiolin 6-Hydroxyluteolin 3',6-dimethyl ether 3',6-Dimethoxy-4',5,7-trihydroxyflavone 4',5,7-Trihydroxy-3',6-dimethoxyflavone 5,7-dihydroxy-2-(4-hydroxy-3-methoxy-phenyl)-6-methoxy-chromen-4-one 5,7-dihydroxy-2-(4-hydroxy-3-methoxyphenyl)-6-methoxy-4H-chromen-4-one 5,7-Dihydroxy-2-(4-hydroxy-3-methoxyphenyl)-6-methoxy-4H-1-benzopyran-4-one 4H-1-Benzopyran-4-one, 5,7-dihydroxy-2-(4-hydroxy-3-methoxyphenyl)-6-methoxy- |
| CAS | 18085-97-7 |
| InChI | InChI=1/C17H14O7/c1-22-13-5-8(3-4-9(13)18)12-6-10(19)15-14(24-12)7-11(20)17(23-2)16(15)21/h3-7,18,20-21H,1-2H3 |
Jacesiolin - Physico-chemical Properties
| Molecular Formula | C17H14O7 |
| Molar Mass | 330.29 |
| Density | 1.483±0.06 g/cm3(Predicted) |
| Melting Point | 259-260 °C |
| Boling Point | 619.0±55.0 °C(Predicted) |
| Flash Point | 232°C |
| Solubility | Soluble in DMSO, ethanol, methanol |
| Vapor Presure | 6.48E-16mmHg at 25°C |
| Appearance | Yellow needle crystal |
| Color | Pale Yellow to Light Yellow |
| pKa | 6.47±0.40(Predicted) |
| Storage Condition | 2-8°C(protect from light) |
| Stability | Hygroscopic |
| Refractive Index | 1.67 |
| MDL | MFCD01081948 |
| Physical and Chemical Properties | Yellow crystalline powder, soluble in methanol, ethanol, DMSO and other organic solvents, derived from Artemisia argyi, Hypericum campestris, Artemisia argyi aerial part, stevia, Rehmannia leaf, overground part of jiangteng. |
Jacesiolin - Reference
| Reference Show more | 1. Cao Li, Lu Jinqing, Ye Xin, et al. Simultaneous determination of kaempferol brown cyanidin isoleanthin in different brands of moxa sticks and moxa columns by HPLC [J]. Chinese Pharmacist 2018 v.21(08):73-75 80. 2. Gong Min, Lu Jinqing, Xiao Yushuo. Determination of Total Flavonoids and Their Three Main Alycones in moxa leaves from different habitats [J]. Chinese Pharmacist 2019 22(05):174-176 183. 3. Pu Rui, Wan Dingrong, Zhao Baixiao, et al. Research on the Influence of Environmental Conditions on the Quality of Artemisia argyi Leaves [J]. World Science and Technology-Modernization of Traditional Chinese Medicine, 2019, v.21(12):206-212. 4. Wang Xinge, Zhao Lu, Ye Junwei, et al.. Study on the preservation effect of moxa powder extract and its compound preservative on cherry tomatoes and grapes. 5. Pu Rui, Wan Dingrong, Chen Pusheng, Xia Hengjian, Wu Juan, Guo Shuangxi. Effects of Environmental Factors on Contents of Seven Active Components in Artemisia argyi Leaves [J]. Chinese Journal of Traditional Chinese Medicine, 2020,35(10):4948-4952. 6. Hu Qian, Li Jing, Liu Da, Cao Yan. Antioxidant Activity of Total Flavonoids Extracts from Artemisia argyi Leaves in Vitro and in Vivo [J]. Food Industry Technology, 2021,42(06):304-309. 7. [IF = 4.411] Min Li et al."Study of the Variation of Phenolic Acid and Flavonoid Content from Fresh Artemisiae argyi Folium to Moxa Wool." Molecules 2019 Jan;24(24):4603 8. [IF = 4.36] Qian Hu et al."Antioxidant capacity of flavonoids from Folium Artemisiae Argyi and the molecular mechanism in Caenorhabditis elegans." J Ethnopharmacol. 2021 Oct;279:114398 9. [IF = 3.645] Lu Huang et al."Qualitative and quantitative analysis of chemical components in Eupatorium lindleyanum DC. by ultra-performance liquid chromatography-mass spectrometry integrated with anti-inflammatory activity research." J Sep Sci. 2021 Sep;44(17):3174-31 |
Jacesiolin - Reference Information
| Plant source: | Folium Artemisia |
| application | brown cyanidin and isothalanthin isolated from mongyi leaves belong to iridoid glycosides, which mainly have heat-clearing, analgesic and other related activities. |
| biological activity | Jaceosidin are flavonoids obtained from Artemisia vestita, which can activate Bax, down-regulate the expression of Mcl-1 and c-FLIP, and induce cancer cell apoptosis. Jaceosidin has anti-cancer and anti-inflammatory effects, can reduce the level of inflammatory factors, activate NF-κB and inhibit the expression of COX-2. |
| Cell Line: | Hec1A, KLE, HES and HESC cells |
| Concentration: | 3.125, 6.25, 12.5, 25, 50, and 100 μM |
| Incubation Time: | 48 hour |
| Result: | Showed cytostatic activity to HES and HESC cells with IC 50 s of 52.68 and 55.10 μM, less cytotocxic on Hec1 A and KLE (IC 50 , 70.54, 147.14 μM). Decreased the volumes of exudates (inflammatory markers), cell number and protein levels. Inhibited TNF-α by 46.7% and 50.8%, IL-1β by 46.0% and 44.7%, and PGE2 by 21.7% and 16.9%, respectively, at 20 mg/kg. Blocked COX-2 expression and NF-κB activation. Reduced hind paw edema volume by 27.1% at 1 h, and 24.0% at 2 h, respectively. |
| Animal Model: | 5-week-old male BALB/c mice (23-26 g) Male Sprague-Dawley rats (180-200 g) |
| Dosage: | 10 and 20 mg/kg 20 mg/kg |
| Administration: | P.O. once a day for 3 days P.O., for 2 hour |
| use | brown pythrin has the effect of lowering blood lipid. used for content determination/identification/pharmacological experiment, etc. Pharmacological effects: anti-tumor, antioxidant, scavenging oxygen free radicals. |
Last Update:2024-04-09 21:54:55
Supplier List
Featured ProductsSpot supply
Product Name: Jaceosidin Visit Supplier Webpage Request for quotationCAS: 18085-97-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Multiple SpecificationsSpot supply
Product Name: 5,7-Dihydroxy-2-(4-hydroxy-3-methoxyphenyl)-6-methoxy-4H-chromen-4-one Visit Supplier Webpage Request for quotationCAS: 18085-97-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 5,7-dihydroxy-2-(4-hydroxy-3-methoxy-phenyl)-6-methoxy-chromen-4-one Request for quotation
CAS: 18085-97-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 18085-97-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: Jaceosidin Request for quotation
CAS: 18085-97-7
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 18085-97-7
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Featured ProductsSpot supply
Product Name: Jaceosidin Visit Supplier Webpage Request for quotationCAS: 18085-97-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Multiple SpecificationsSpot supply
Product Name: 5,7-Dihydroxy-2-(4-hydroxy-3-methoxyphenyl)-6-methoxy-4H-chromen-4-one Visit Supplier Webpage Request for quotationCAS: 18085-97-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 5,7-dihydroxy-2-(4-hydroxy-3-methoxy-phenyl)-6-methoxy-chromen-4-one Request for quotation
CAS: 18085-97-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 18085-97-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: Jaceosidin Request for quotation
CAS: 18085-97-7
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 18085-97-7
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
View History



