Hexadecanol
hexadecanol
CAS: 29354-98-1
Molecular Formula: C16H34O
Hexadecanol - Names and Identifiers
| Name | hexadecanol |
| Synonyms | hexadecanol hexadecan-4-ol |
| CAS | 29354-98-1 |
| EINECS | 249-583-5 |
Hexadecanol - Physico-chemical Properties
| Molecular Formula | C16H34O |
| Molar Mass | 242.45 |
| Density | 0.8323 (estimate) |
| Boling Point | 315.27°C (estimate) |
| Refractive Index | 1.4278 (estimate) |
Hexadecanol - Introduction
hexadecanol (also known as hexyl alcohol or n-hexanol) is a colorless, odorless organic compound with the chemical formula C16H34O. Next, the nature, use, preparation and safety information of hexadecanol will be introduced.
Nature:
-Appearance: Colorless liquid.
-Solubility: Soluble in organic solvents, such as ethers, alcohols and esters, almost insoluble in water.
-Boiling point: About 153 ° C.
-Density: about 0.8g/cm³.
-ignition point: about 61 ° C.
-non-toxic, low volatility.
Use:
- hexadecanol is widely used in cosmetics and personal care products, such as shampoo, conditioner, perfume, etc.
-It is also used as an industrial solvent, especially for the preparation of lubricants and surfactants.
- hexadecanol can also be used as an additive to adjust the rheological properties of paints and coatings or increase their viscosity.
Preparation Method:
- hexadecanol can be synthesized by a variety of methods, the most commonly used method is hydrogenation. Typically, hexadecanol is obtained by reacting hexadecane (C16H34) with hydrogen in the presence of a suitable catalyst.
Safety Information:
- hexadecanol is a relatively safe compound, non-toxic to human body. However, it has a certain degree of irritation and may cause eye and skin irritation, so you need to pay attention to protective measures when handling.
-If you accidentally inhale or come into contact with hexadecanol, you should immediately move to a ventilated place and flush the affected area with water.
-it belongs to low volatile substances, can slowly volatilize from water. When in use, it is necessary to avoid contact with fire sources such as open flames and high temperatures.
-During storage and transportation, the container should be sealed and avoid contact with oxidants, strong acids, strong bases and other substances.
Nature:
-Appearance: Colorless liquid.
-Solubility: Soluble in organic solvents, such as ethers, alcohols and esters, almost insoluble in water.
-Boiling point: About 153 ° C.
-Density: about 0.8g/cm³.
-ignition point: about 61 ° C.
-non-toxic, low volatility.
Use:
- hexadecanol is widely used in cosmetics and personal care products, such as shampoo, conditioner, perfume, etc.
-It is also used as an industrial solvent, especially for the preparation of lubricants and surfactants.
- hexadecanol can also be used as an additive to adjust the rheological properties of paints and coatings or increase their viscosity.
Preparation Method:
- hexadecanol can be synthesized by a variety of methods, the most commonly used method is hydrogenation. Typically, hexadecanol is obtained by reacting hexadecane (C16H34) with hydrogen in the presence of a suitable catalyst.
Safety Information:
- hexadecanol is a relatively safe compound, non-toxic to human body. However, it has a certain degree of irritation and may cause eye and skin irritation, so you need to pay attention to protective measures when handling.
-If you accidentally inhale or come into contact with hexadecanol, you should immediately move to a ventilated place and flush the affected area with water.
-it belongs to low volatile substances, can slowly volatilize from water. When in use, it is necessary to avoid contact with fire sources such as open flames and high temperatures.
-During storage and transportation, the container should be sealed and avoid contact with oxidants, strong acids, strong bases and other substances.
Last Update:2024-04-09 19:04:59
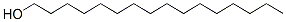
Supplier List
Product Name: hexadecanol Request for quotation
CAS: 29354-98-1
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 29354-98-1
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: hexadecanol Request for quotation
CAS: 29354-98-1
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 29354-98-1
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
View History
