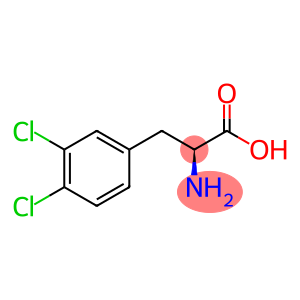
H-Phe(3,4-DiCl)-OH
L-3,4-Dichlorophe
CAS: 52794-99-7
Molecular Formula: C9H9Cl2NO2
H-Phe(3,4-DiCl)-OH - Names and Identifiers
| Name | L-3,4-Dichlorophe |
| Synonyms | L-3,4-Dichlorophe H-PHE(M,P-CL2)-OH L-3,4-DICHLOROPHE H-Phe(3,4-DiCl)-OH H-PHE(3,4-DICL)-OH H-PHE(3,4-CL 2)-OH RARECHEM BK PT 0115 L-3,4-DICHLOROPHENYLALANINE L-3,4-Dichlorophenylalanine 3,4-DICHLORO-L-PHENYLALANINE 3,4-dichloro-L-phenylalanine (S)-2-AMINO-3-(3,4-DICHLORO-PHENYL)-PROPIONIC ACID |
| CAS | 52794-99-7 |
| InChI | InChI=1/C9H9Cl2NO2/c10-6-2-1-5(3-7(6)11)4-8(12)9(13)14/h1-3,8H,4,12H2,(H,13,14) |
H-Phe(3,4-DiCl)-OH - Physico-chemical Properties
| Molecular Formula | C9H9Cl2NO2 |
| Molar Mass | 234.08 |
| Density | 1.450±0.06 g/cm3(Predicted) |
| Boling Point | 370.6±42.0 °C(Predicted) |
| Flash Point | 177.9°C |
| Vapor Presure | 3.78E-06mmHg at 25°C |
| Appearance | Solid |
| pKa | 2.14±0.20(Predicted) |
| Storage Condition | Store at RT. |
| Refractive Index | 1.601 |
H-Phe(3,4-DiCl)-OH - Risk and Safety
| Hazard Symbols | Xi - Irritant |
H-Phe(3,4-DiCl)-OH - Introduction
L-3,4-Dichlorophe(L-3) is an organic compound whose chemical formula is C8H7Cl2NO2.
Nature:
-Appearance: Colorless crystalline solid.
-Melting point: about 162-164°C.
-Solubility: Soluble in ethanol, ether and acid solution, slightly soluble in water.
-Optical activity: L-configuration, with chirality.
Use:
-Pharmaceutical field: L-3,4-Dichlorophe can be used as an intermediate in drug synthesis, such as the synthesis of certain ketones.
-Research use: The compound has important application value in organic synthesis and medicinal chemistry research.
Preparation Method:
L-3,4-Dichlorophe can be synthesized from styrene. The following is a possible synthesis method:
1. Dissolve styrene in hydrochloric acid and add ferrous chloride as a catalyst.
2. the chlorination reaction is carried out at an appropriate temperature, so that the chlorine gas catalyzed by ferrous chloride reacts with styrene, and the two chlorine atoms are introduced into the benzene ring.
3. The resulting product is reacted with sulfonic acid to generate sulfonic acid ester.
4. After further reduction, acidification, regeneration crystallization and other steps, L-3,4-Dichlorophe was finally obtained.
Safety Information:
L-3,4-Dichlorophe is less toxic and dangerous, but as an organic compound, it still needs to be handled with caution. During use or storage, the following should be noted:
-Avoid contact with strong oxidants to avoid chemical reactions.
-Wear appropriate personal protective equipment such as gloves, goggles, and lab coats prior to operation.
-Avoid inhaling dust or contact with skin and eyes. If contact occurs, rinse the affected area immediately with plenty of water and seek medical attention immediately.
-When storing, keep it in a dry and cool place, and seal the container away from fire.
Nature:
-Appearance: Colorless crystalline solid.
-Melting point: about 162-164°C.
-Solubility: Soluble in ethanol, ether and acid solution, slightly soluble in water.
-Optical activity: L-configuration, with chirality.
Use:
-Pharmaceutical field: L-3,4-Dichlorophe can be used as an intermediate in drug synthesis, such as the synthesis of certain ketones.
-Research use: The compound has important application value in organic synthesis and medicinal chemistry research.
Preparation Method:
L-3,4-Dichlorophe can be synthesized from styrene. The following is a possible synthesis method:
1. Dissolve styrene in hydrochloric acid and add ferrous chloride as a catalyst.
2. the chlorination reaction is carried out at an appropriate temperature, so that the chlorine gas catalyzed by ferrous chloride reacts with styrene, and the two chlorine atoms are introduced into the benzene ring.
3. The resulting product is reacted with sulfonic acid to generate sulfonic acid ester.
4. After further reduction, acidification, regeneration crystallization and other steps, L-3,4-Dichlorophe was finally obtained.
Safety Information:
L-3,4-Dichlorophe is less toxic and dangerous, but as an organic compound, it still needs to be handled with caution. During use or storage, the following should be noted:
-Avoid contact with strong oxidants to avoid chemical reactions.
-Wear appropriate personal protective equipment such as gloves, goggles, and lab coats prior to operation.
-Avoid inhaling dust or contact with skin and eyes. If contact occurs, rinse the affected area immediately with plenty of water and seek medical attention immediately.
-When storing, keep it in a dry and cool place, and seal the container away from fire.
Last Update:2024-04-09 02:00:46
Supplier List
Product Name: L-3,4-Dichlorophe Request for quotation
CAS: 52794-99-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 52794-99-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: H-Phe(3,4-DiCl)-OH Visit Supplier Webpage Request for quotationCAS: 52794-99-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3,4-Dichlorophenylalanine Request for quotation
CAS: 52794-99-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 52794-99-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: L-3,4-Dichloro Phenylalanine Visit Supplier Webpage Request for quotation
CAS: 52794-99-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 52794-99-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: L-3,4-Dichlorophe Request for quotation
CAS: 52794-99-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 52794-99-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: H-Phe(3,4-DiCl)-OH Visit Supplier Webpage Request for quotationCAS: 52794-99-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3,4-Dichlorophenylalanine Request for quotation
CAS: 52794-99-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 52794-99-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: L-3,4-Dichloro Phenylalanine Visit Supplier Webpage Request for quotation
CAS: 52794-99-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 52794-99-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



