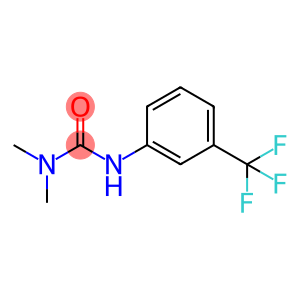
Fluometuran
fluometuron
CAS: 2164-17-2
Molecular Formula: C10H11F3N2O
Fluometuran - Names and Identifiers
| Name | fluometuron |
| Synonyms | Fluometuran fluometuron Fluometuron 0 fluometuron (bsi,iso,ansi,wssa) FLUOMETHURON PESTANAL (N,N-DIMETHYL-N'-( 1,1-dimethyl-3-(3-trifluoromethylphenyl) 1-(3-Trifluoromethylphenyl)-3,3-dimethylurea 1,1-dimethyl-3-(alpha,alpha,alpha-trifluoro-m-tolyl)urea |
| CAS | 2164-17-2 |
| EINECS | 218-500-4 |
| InChI | InChI=1/C10H11F3N2O/c1-15(2)9(16)14-8-5-3-4-7(6-8)10(11,12)13/h3-6H,1-2H3,(H,14,16) |
Fluometuran - Physico-chemical Properties
| Molecular Formula | C10H11F3N2O |
| Molar Mass | 232.2 |
| Density | 1.39 |
| Melting Point | 163°C |
| Boling Point | 280 °C |
| Water Solubility | Slightly soluble. |
| Solubility | Chloroform (Sparingly, Heated), Methanol (Slightly) |
| Vapor Presure | 0Pa at 25℃ |
| Appearance | Powder |
| Color | White |
| Merck | 14,4154 |
| BRN | 2217354 |
| pKa | 14.55±0.70(Predicted) |
| Storage Condition | 0-6°C |
| Physical and Chemical Properties | Density: 1.39 Melting Point: 163°C water-soluble; <0.1g/100 mL at 21.5°C |
| Use | Used to control weeds in cotton, corn, potato, onion, sugarcane, fruit trees and other fields |
Fluometuran - Risk and Safety
| Hazard Symbols | Xn - Harmful |
| Risk Codes | 22 - Harmful if swallowed |
| UN IDs | UN2767 (Phenyl urea pesti-cides, solid, toxic) |
| WGK Germany | 3 |
| RTECS | YT1575000 |
| Toxicity | LD50 in rats (mg/kg): 8900 orally; in rabbits (mg/kg): >10,000 dermally (Ben-Dyke) |
Fluometuran - Nature
Open Data Verified Data
pure white crystals. Melting point 163~164.5 ℃ (original 155 ℃), relative density (20 ℃)1. 39, vapor pressure (20 ℃)6.65 × 10-s Pa. 25 ° C water solubility of 80 x 10-6, soluble in acetone, ethanol and other organic solvents. It is easy to decompose in case of strong acid and alkali.
Fluometuran - Preparation Method
Open Data Verified Data
with a trifluoromethyl aniline, phosgene, two as raw materials. Using toluene as solvent, phosgene and meta-trifluoromethylaniline were reacted at room temperature to generate isocyanate. The subsequent addition of isocyanate with dimethylamine gives the result of the vortazone.
Fluometuran - Use
Open Data Verified Data
It is an early herbicide with internal absorption conduction type, which is substituted for urea herbicide. The control of cotton, corn and other crops in the field of annual weeds effect is outstanding. For cotton, corn, sugar cane, fruit tree control of annual grass weeds and broad-leaved grass, such as humulus scandens, crickets, Kentucky bluegrass, Maldon, bitter vegetables, Cyperus papyrifera, dampenia, amaranth, dog tail grass, purslane, gold, etc.
Fluometuran - Safety
Open Data Verified Data
The oral LD50 of rats was 8900mg/kg. Rats were fed at a dose of 100mg/kg per day. 180 days later, no toxic symptoms were observed. Rats fed for 2 years test no effect of the dose of 30mg/kg, mice 10mg/kg, animal test no teratogenic, carcinogenic, mutagenic effect, reproductive test also found no abnormalities. Rainbow trout LC50 47mg/L(96H), carp LC50 170mg/L. Bee LD50 is 193VG/, low toxicity to birds. Slightly irritating to the eyes and skin.
Fluometuran - Reference Information
| LogP | 2.28 at 23.5℃ |
| NIST chemical information | Information provided by: webbook.nist.gov (external link) |
| (IARC) carcinogen classification | 3 (Vol. 30, Sup 7) 1987 |
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| toxicity | acute oral LD50 of rats is 6400 mg/kg, acute percutaneous LD50>2000 mg/kg. Rats were fed at a daily dose of 100mg/kg, and there was no poisoning symptoms for 180d. The 2-year feeding test was 30mg/kg for rats and 10mg/kg for mice. Animal experiments have no teratogenic, carcinogenic, and mutagenic effects, and reproduction tests have no abnormalities. Rainbow trout LC5047mg/L(96h), carp LC50170mg/L. Bee LD50193μg/bee. Low toxicity to black class. Slight irritation to eyes and skin. |
| use | internal absorption conduction dryland herbicide. Weeds are absorbed through the roots, inhibiting photosynthesis. Used for cotton, corn, sugarcane, fruit trees to control annual gramineous weeds and broadleaf grasses, such as barnyard grass, crickets, bluegrass, crickets, sorrel, sedge, kanmai Niang, amaranth, Setaria, Chenopodium, Portulaca, Thousand Gold, etc. Forzaolong is used as soil treatment agent. For example, cotton seedlings are applied before transplanting, and water is sprayed on the soil surface with 80% wettable powder of 15-23g/100 m2. Direct seeding cotton is sprayed on the soil surface with 80% wettable powder of 15-18.8g/100 m2 4-5 days after sowing. As a substituted urea herbicide, Fushuron has an outstanding effect on controlling annual weeds in cotton, corn and other crops. used to control weeds in fields such as cotton, corn, potatoes, green onions, sugarcane, and fruit trees |
| Production method | The m-trifluorotoluene isocyanate is prepared by the reaction of m-aminotrifluorotoluene and phosgene, and then reacted with dimethylamine to obtain voltaron. 1. Preparation of m-trifluorotoluene isocyanate The phosgene is passed into toluene. After absorbing a part of phosgene, one side continues to pass the phosgene, and the toluene solution of m-aminotrifluorotoluene is added dropwise. After dropping, the temperature will be slowly raised to about 75 ℃, and the light gas will continue until the reaction liquid becomes clear. Continue stirring for 30min, quickly raise the temperature to about 100 ℃, and remove excess phosgene and hydrogen chloride gas generated by reaction through dry air to obtain toluene solution of m-trifluorotoluene isocyanate [392-01-1]. 2. Preparation of Voltaron Add the above-mentioned m-trifluorotoluene isocyanate toluene solution into a synthetic pot, stir, control the temperature at about 50 ℃, and add dimethylamine dropwise. After dropping, the reaction was maintained for 1h, and the pH value of the reaction solution was stable at 7.5-8 at the end point. After standing, it will be filtered and dried at 80-90 ℃ to obtain Fushaolong raw powder with a content of more than 95% and a yield of about 92%. isocyanate method uses m-trifluoromethylaniline, phosgene and dimethylamine as raw materials. Using toluene as solvent, phosgene and m-trifluoromethylaniline are reacted at normal temperature, and then the temperature is increased to (75±5)℃ to form isocyanate. At (50±5)℃, it was added with dimethylamine to generate voluron, and the end pH value was 7.5~8. The product is separated by centrifugation, the mother liquor is distilled to recover toluene, and the HCl produced by the reaction is desorbed with a yield of 92%. Carbamoyl chloride method |
| category | pesticide |
| toxicity classification | poisoning |
| acute toxicity | oral-rat LD50: 1450 mg/kg; Oral-mouse LD50: 900 mg/kg |
| flammability hazard characteristics | Combustion produces toxic nitrogen oxides and fluoride gases |
| storage and transportation characteristics | warehouse ventilation and low temperature drying; separate from food raw materials storage and transportation |
| fire extinguishing agent | dry powder, foam, sand |
| occupational standard | STEL 5 mg/m3 |
| toxic substance data | information provided by: pubchem.ncbi.nlm.nih.gov (external link) |
Supplier List
CAS: 2164-17-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 2164-17-2
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 2164-17-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 2164-17-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 2164-17-2
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 2164-17-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025


