Ecalta
Anidulafungin
CAS: 166663-25-8
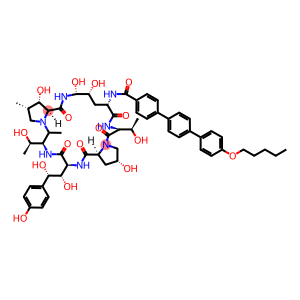
Molecular Formula: C58H73N7O17
Ecalta - Names and Identifiers
| Name | Anidulafungin |
| Synonyms | Eraxis Ecalta LY 303366 Anidulafungin LY 303366, Eraxis Anidulafungin(LY303366) 1-[(4R,5R)-4,5-dihydroxy-N2-[[4''-(pentyloxy)[1,1'',1''-terphenyl]-4-yl]carbonyl]-L-ornithine]-echinocandin B |
| CAS | 166663-25-8 |
| EINECS | 658-060-4 |
| InChI | InChI=1/C58H73N7O17/c1-5-6-7-24-82-40-22-18-35(19-23-40)33-10-8-32(9-11-33)34-12-14-37(15-13-34)51(74)59-41-26-43(70)54(77)63-56(79)47-48(71)29(2)27-65(47)58(81)45(31(4)67)61-55(78)46(50(73)49(72)36-16-20-38(68)21-17-36)62-53(76)42-25-39(69)28-64(42)57(80)44(30(3)66)60-52(41)75/h8-23,29-31,39,41-50,54,66-73,77H,5-7,24-28H2,1-4H3,(H,59,74)(H,60,75)(H,61,78)(H,62,76)(H,63,79)/t29-,30+,31+,39+,41-,42-,43+,44-,45-,46-,47-,48-,49-,50-,54+/m0/s1 |
Ecalta - Physico-chemical Properties
| Molecular Formula | C58H73N7O17 |
| Molar Mass | 1140.24 |
| Density | 1.47±0.1 g/cm3(Predicted) |
| Melting Point | >196°C (subl.) |
| Boling Point | 1477.0±65.0 °C(Predicted) |
| Flash Point | 847°C |
| Solubility | DMSO : 30 mg/mL mother liquor preservation: sub-package and freeze storage to avoid repeated freezing and thawing;-20 ℃,1 month;-80 ℃,6 months (after dilution, the solution temperature is low and storage may precipitate, try to use it now) Cell experiment: Dissolve with DMSO first: dilute with culture medium then, and the dilution process is recommended to be carried out in stages to avoid too fast concentration change leading to compound precipitation. If the compound is precipitated during the dilution process, it can be redissolved by ultrasound. During dilution, ensure that the final concentration of DMSO in the working fluid should be below 0.1% as far as possible, and the maximum should not exceed 0.5%, and set up a DMSO control group with corresponding concentration. Animal experiment: Dissolve with DMSO first: dilute with water or normal saline, etc. The dilution process is recommended to be carried out in sections to avoid excessive concentration changes leading to compound p |
| Vapor Presure | 0mmHg at 25°C |
| Appearance | Solid |
| Color | White to Pale Beige |
| pKa | 9.86±0.26(Predicted) |
| Storage Condition | under inert gas (nitrogen or Argon) at 2-8°C |
| Refractive Index | 1.688 |
| Use | Anidulafungin, also known as LY303366, is a semisynthetic echinocandin used as an antifungal drug. Anidulafungin was approved on 2/21/2006. Anidulafungin has proven efficacy against esophageal candidiasis, but its main use will probably be in invasive Candida infection; it may also have application in treating invasive Aspergillus infection. It is a member of the class of antifungal drugs known as the echinocandins; its mechanism of action is by inhibition of (1→3)-β-D-glucan synthase, an enzyme important to the synthesis of the fungal cell wall. Anidulafungin inhibits glucan synthase, an enzyme important in the formation of (1→3)-β-D-glucan, a major fungal cell wall component. Glucan synthase is not present in mammalian cells, so it is an attractive target for antifungal activity. |
| Target | glucan synthase;Antifungal |
Ecalta - Reference
| Reference Show more | 1: Hinske LC, Weis F, Heyn J, Hinske P, Beiras-Fernandez A. The role of micafungin and anidulafungin in the treatment of systemic fungal infections: applications and patents for two novel echinocandins. Recent Pat Antiinfect Drug Discov. 2012 Apr;7(1):1-7. Review. PubMed PMID: 22044354. 2: George J, Reboli AC. Anidulafungin: when and how? The clinician's view. Mycoses. 2012 Jan;55(1):36-44. doi: 10.1111/j.1439-0507.2011.02052.x. Epub 2011 Jun 13. Review. PubMed PMID: 21668526. 3: Wilke M. Treatment and prophylaxis of invasive candidiasis with anidulafungin, caspofungin and micafungin and its impact on use and costs: review of the literature. Eur J Med Res. 2011 Apr 28;16(4):180-6. Review. PubMed PMID: 21486732; PubMed Central PMCID: PMC3352074. 4: Glöckner A. Treatment and prophylaxis of invasive candidiasis with anidulafungin, caspofungin and micafungin:review of the literature. Eur J Med Res. 2011 Apr 28;16(4):167-79. Review. PubMed PMID: 21486731; PubMed Central PMCID: PMC3352073. 5: Kofla G, Ruhnke M. Pharmacology and metabolism of anidulafungin, caspofungin and micafungin in the treatment of invasive candidosis: review of the literature. Eur J Med Res. 2011 Apr 28;16(4):159-66. Review. PubMed PMID: 21486730; PubMed Central PMCID: PMC3352072. 6: Tapısız A. Anidulafungin: is it a promising option in the treatment of pediatric invasive fungal infections? Expert Rev Anti Infect Ther. 2011 Mar;9(3):339-46. doi: 10.1586/eri.11.7. Review. PubMed PMID: 21417873. 7: Mayr A, Aigner M, L |
Ecalta - Preparation solution concentration reference
| 1mg | 5mg | 10mg | |
|---|---|---|---|
| 1 mM | 0.877 ml | 4.385 ml | 8.77 ml |
| 5 mM | 0.175 ml | 0.877 ml | 1.754 ml |
| 10 mM | 0.088 ml | 0.439 ml | 0.877 ml |
| 5 mM | 0.018 ml | 0.088 ml | 0.175 ml |
Last Update:2024-01-02 23:10:35
Ecalta - Cell Experiment
Anidulafungin is dissolved in DMSO and stored, and then diluted[2]. Stocks of CD101 (formerly SP 3025, biafungin, AF-025) are prepared fresh in 100% DMSO prior to use. The comparator antifungals Anidulafungin (ANF), Caspofungin (CSF), and Amphotericin B (AMB) are also prepared in 100% DMSO. MIC assays are performed via broth microdilution in accordance with CLSI guidelines, with the exception that test compounds are made up at a 50× final assay concentration and 100 μL assay mixture volumes are used (2 μL added to 98 μL of broth containing cells at 0.5×103 to 2.5×103 CFU/mL). All MIC assays are run at least three times, and a representative data set is shown. Quality control (QC) is assessed throughout the study via comparison of MIC values derived for WT C. krusei strain ATCC 6258 for AMB, CSF, and ANF with previously reported CLSI 24-h broth microdilution QC ranges.
Last Update:2023-08-16 21:32:38
Ecalta - Reference Information
| antifungal drug | anifenjing was developed and patented by Lilly, and then transferred to Vicuron company for application and development. the company was acquired by Pfizer in September 2005. Pfizer's purpose of acquiring the product was to continue its shortage of fungal line products after the expiration of the antifungal drug fluconazole patent. in 2006, the product was approved for listing through FDA review. Fungal infection, especially Candida infection, is a common cause of death for hospital-acquired blood infections. The mortality rate after infection is about 40%. Triazole drugs, especially fluconazole, have long been regarded as Candida and other fungal infections. For the gold standard drug, Anifenjing is the first drug that has been proven to be more effective than fluconazole in clinical studies. Anifenjing is an echinocandin antifungal drug. Similar products include micafungin and caspofungin, which are used to treat esophageal candidiasis, candidiasis and other types of candidal infections, including peritonitis and abdominal abscess. Studies have confirmed that it has strong antifungal activity in vitro and in vivo, and there is no cross-resistance. Fenjing antifungal antibiotics have become an important drug for the clinical treatment of systemic fungal infections, and these drugs have the advantages of high antibacterial activity and low adverse reactions. |
| indications | anifenjing is used for candidal septicemia, oropharyngeal and esophageal candidiasis, abdominal abscess caused by candida, candidal peritonitis and aspergillus infection. |
| pharmacological action | anifenjing is an antifungal drug of Acanthocin, and its mechanism of action is the same as that of "caspofungin". It has antibacterial activity against Candida and Aspergillus, but is ineffective against Cryptococcus, Fusarium and Trichospora. |
| pharmacokinetics | in echinocicosin drugs. Anifenjing is unique. Through a series of biological transformations. It is slowly degraded in plasma, not metabolized. More than 90% of anifenjing is slowly chemically degraded in blood and is acted on by non-specific peptidases to form ring-opening products. Anifenjing t1/2 is about 24 h, while its degradation product t1/2 is about 4 d. Anifenjing degradation is not metabolized by the cytochrome P450 enzyme system, and there are few drugs or degradation products in the urine. Anifenjing degradation products are excreted in the feces through bile. For patients with any degree of liver injury and patients with renal insufficiency, the use of anifenjing does not require dose adjustment. |
| usage and dosage | intravenous administration. 1. Candida septicemia, intraperitoneal disseminated candidiasis and peritonitis: 200mg on the 1st and 100mg once a day thereafter. The course of treatment lasted until at least 14 days after the last positive culture. 2. Esophageal candidiasis: 100mg on the 1st and 50mg every time thereafter, once a day. The course of treatment is at least 14 days, and the symptoms will last at least 7 days after elimination. 3. Oropharyngeal Candida infection (HIV infection): for patients who resist fluconazole, 100mg on the 1st and 50mg every time thereafter, once a day. |
| biological activity | Anidulafungin (LY303366), a echinocandin derivative that inhibits the activity of glucan synthase and is used as an antifungal drug. |
| Target | Value |
Last Update:2024-04-09 15:16:48
Supplier List
Spot supply
Product Name: Anidulafungin Visit Supplier Webpage Request for quotationCAS: 166663-25-8
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Anidulafungin Visit Supplier Webpage Request for quotationCAS: 166663-25-8
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: Anidulafungin Visit Supplier Webpage Request for quotationCAS: 166663-25-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Anidulafungin Request for quotation
CAS: 166663-25-8
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 166663-25-8
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Spot supply
Product Name: N-((2R,6S,9S,11R,12R,14aS,15S,16S,20S,23S,25aS)-23-((1S,2S)-1,2-dihydroxy-2-(4-hydroxyphenyl)ethyl)-2,11,12,15-tetrahydroxy-6,20-bis((R)-1-hydroxyethyl)-16-methyl-5,8,14,19,22,25-hexaoxotetracosahydro Visit Supplier Webpage Request for quotationCAS: 166663-25-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Anidulafungin Visit Supplier Webpage Request for quotationCAS: 166663-25-8
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Anidulafungin Visit Supplier Webpage Request for quotationCAS: 166663-25-8
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: Anidulafungin Visit Supplier Webpage Request for quotationCAS: 166663-25-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Anidulafungin Request for quotation
CAS: 166663-25-8
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 166663-25-8
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Spot supply
Product Name: N-((2R,6S,9S,11R,12R,14aS,15S,16S,20S,23S,25aS)-23-((1S,2S)-1,2-dihydroxy-2-(4-hydroxyphenyl)ethyl)-2,11,12,15-tetrahydroxy-6,20-bis((R)-1-hydroxyethyl)-16-methyl-5,8,14,19,22,25-hexaoxotetracosahydro Visit Supplier Webpage Request for quotationCAS: 166663-25-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History




