ENIDE
DIPHENAMID
CAS: 957-51-7
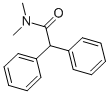
Molecular Formula: C16H17NO
ENIDE - Names and Identifiers
| Name | DIPHENAMID |
| Synonyms | ENIDE ENIDE(R) DYMID(R) DIPHENAMID LABOTEST-BB LT00159758 N,N-DIMETHYL-2,2-DIPHENYLACETAMIDE 2,2-DIPHENYL-N,N-DIMETHYL-ACETAMIDE |
| CAS | 957-51-7 |
| EINECS | 213-482-4 |
ENIDE - Physico-chemical Properties
| Molecular Formula | C16H17NO |
| Molar Mass | 239.31 |
| Density | 1.1700 |
| Melting Point | 132-136°C(lit.) |
| Boling Point | 381.97°C (rough estimate) |
| Water Solubility | 239.9mg/L(25 ºC) |
| Appearance | Huff Powder |
| Color | White, light |
| Merck | 13,3339 |
| BRN | 2052009 |
| pKa | -0.68±0.70(Predicted) |
| Storage Condition | 0-6°C |
| Stability | Stable. Incompatible with strong oxidizing agents, strong bases. |
| Refractive Index | 1.5500 (estimate) |
| Physical and Chemical Properties | Pure white crystals. m.p.134.5 ~ 135.5 deg C, the relative density of 1.17. At room temperature in acetone solubility of 15%, xylene 3%, dimethyl formamide 165g/L. Storage at room temperature stable for 5 years, light, moderate thermal stability, non-corrosive. Industrial products are off-white solids at 95%-98% m. P. 128-135 °c. |
ENIDE - Risk and Safety
| Hazard Symbols | Xn - Harmful |
| Risk Codes | R22 - Harmful if swallowed R52/53 - Harmful to aquatic organisms, may cause long-term adverse effects in the aquatic environment. |
| Safety Description | 61 - Avoid release to the environment. Refer to special instructions / safety data sheets. |
| UN IDs | UN3077(solid) |
| WGK Germany | 2 |
| RTECS | AB8050000 |
| HS Code | 29242990 |
| Toxicity | LD50 orally in rats: 700 mg/kg (Bailey, White) |
ENIDE - Reference Information
| toxicity | acute oral LD50 in rats is 1373mg/kg (970mg/kg), 1500mg/kg in rabbits and 1000mg/kg in dogs. No irritation to eyes, slight irritation to skin. The 2-year feeding test for dogs and rats had no effect on the dose of 2000mg/kg. LC50 is 40mg/L for carp, 32mg/L for blue gill fish and 34mg/L for goldfish. Bee oral LD50>241.72 μg/bee. |
| chemical properties | pure product is white crystal. M. p.134.5 ~ 135.5 ℃, relative density 1.17. The solubility in acetone at normal temperature is 15%, xylene is 3%, dimethylformamide is 165g/L. Stable storage period at room temperature for 5 years, moderate to light and thermal stability, non-corrosive. Industrial products are gray-white solids with a content of 95% ~ 98%,m.p.128~135 ℃. |
| use | is a herbicide for selective pre-bud soil treatment, which mainly inhibits the cell division of weed meristem through root absorption, prevents the formation of young buds and secondary roots, and causes weeds to die. It can be used for soil treatment before sowing of soybeans, peanuts, tobacco, cabbage, etc. Control and eliminate a variety of annual gramineous, sedge and broad-leaved weeds, such as soybean and peanut fields, spray the soil surface with 90% wettable powder 22.5~37.5g/100m2 before sowing, and then mix the soil. |
| production method | preparation of diphenyltrichloroethane 0.3molSG, 0.1mol trichloral and 0.1mol 98% sulfuric acid are cooled to 0-5 ℃, 0.3mol 20% fuming sulfuric acid is added dropwise, kept at 5-10 ℃, dripped at 2-4h, slowly heated to room temperature, and post-treated to obtain diphenyltrichloroethane with 80% yield. Preparation of Diphenylacetic Acid After dissolving 0.1mol of diphenyltrichloroethane with 25g of benzene, it was dripped into a solution containing 50g of ethylene glycol monoethyl ether and 0.5mol of sodium hydroxide at 155~160 ℃. After dropping for about 1h, the heating reaction was continued for 5h, and diphenylacetic acid was obtained by post-treatment with 85% yield. Preparation of diphenylacetyl chloride 4.24g of diphenylacetic acid was dissolved in 35mL chloroform, 2 drops of dimethylformamide and 3mL of thionyl chloride were added, refluxed in a water bath for 0.5h, sulfur dioxide and hydrogen chloride gas produced by the reaction were eliminated, chloroform was evaporated, cooled, and 15mL of benzene was added for the preparation of diphenyloxalamine. Synthesis of diphenyloxalamine 2.84g of dimethylamine is mixed with 25mL benzene, the acyl chlorobenzene solution prepared by the previous reaction is added, refluxed in a water bath for 2.5h, 20mL chloroform is added after cooling, and the solvent is washed, dried and recovered to obtain diphenyloxalamine. The yield of the above two steps is 85.8%. M. p. 130 ℃, recrystallized with benzene and petroleum ether mixture, m.p.133~135 ℃. |
| NIST chemical information | Information provided by: webbook.nist.gov (external link) |
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| toxic substance data | information provided by: pubchem.ncbi.nlm.nih.gov (external link) |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: Diphenamid Visit Supplier Webpage Request for quotationCAS: 957-51-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: DIPHENAMID Request for quotation
CAS: 957-51-7
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 957-51-7
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: Diphenamid Visit Supplier Webpage Request for quotationCAS: 957-51-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: DIPHENAMID Request for quotation
CAS: 957-51-7
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 957-51-7
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
View History

