Durabuflo
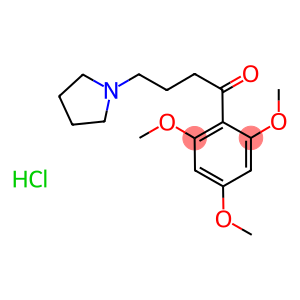
Buflomedil Hydrochloride
CAS: 35543-24-9
Molecular Formula: C17H26ClNO4
Durabuflo - Names and Identifiers
| Name | Buflomedil Hydrochloride |
| Synonyms | Buflan ll1656 Durabuflo BUFLOMEDIL HCL Buflomedil HCl BUFLOMEDIL HCL EP Buflomedil alkali Buflomedil Hcl EP5 BUFLOMEDIL HYDROCHLORIDE Buflomedil Hydrochloride BuflomedilHclC17H25No4.Hcl 1-[4-oxo-4-(2,4,6-trimethoxyphenyl)butyl]pyrrolidinium chloride 2',4',6-Trimethoxy-4-[1-pyrrolidinyl] butyrophenone hydrochloride |
| CAS | 35543-24-9 |
| EINECS | 252-611-9 |
| InChI | InChI=1/C18H26O4.ClH/c1-20-14-11-16(21-2)18(17(12-14)22-3)15(19)10-6-9-13-7-4-5-8-13;/h11-13H,4-10H2,1-3H3;1H |
| InChIKey | ZDPACSAHMZADFZ-UHFFFAOYSA-N |
Durabuflo - Physico-chemical Properties
| Molecular Formula | C17H26ClNO4 |
| Molar Mass | 343.85 |
| Melting Point | 192-193° |
| Solubility | Freely soluble in water, soluble in ethanol (96 per cent), very slightly soluble in acetone. |
| Appearance | neat |
| Color | White to Off-White |
| Storage Condition | Inert atmosphere,Room Temperature |
| Use | Is a multi-drug effect of cerebral vasodilator |
Durabuflo - Risk and Safety
| WGK Germany | 3 |
| RTECS | EL9885000 |
| Toxicity | LD50 in mice (mg/kg): 80 ±4.6 i.v. (Lafon) |
Durabuflo - Preparation solution concentration reference
| 1mg | 5mg | 10mg | |
|---|---|---|---|
| 1 mM | 2.908 ml | 14.541 ml | 29.082 ml |
| 5 mM | 0.582 ml | 2.908 ml | 5.816 ml |
| 10 mM | 0.291 ml | 1.454 ml | 2.908 ml |
| 5 mM | 0.058 ml | 0.291 ml | 0.582 ml |
Last Update:2024-01-02 23:10:35
Durabuflo - Reference Information
| Overview | Buflomedil hydrochloride, the chemical name is 4-(1-pyrrolidinyl) -1-( 2,4, 6-trimethoxyphenyl)-1-butanone hydrochloride, trade name: Fonzylane, Huonanling, Furlan, Celerus, etc., is a vasoactive compound with multiple mechanisms of action. It can inhibit α1 and α2 adrenergic receptors, relax vascular smooth muscle, improve hemodynamics and oxygen supply at the ischemic site; inhibit platelet aggregation, reduce blood viscosity, and improve microcirculation; increase and improve the deformability of red blood cells, The effect of increasing oxygen partial pressure improves hemorheology; it has a weak calcium antagonism. At present, it is widely used in clinical treatment of ischemic cerebrovascular diseases, vascular dementia and peripheral vascular diseases. [Pharmacokinetics] Buflomedil is easily absorbed by gastrointestinal tract, and can reach the average highest blood concentration 1.5~4 hours after oral administration. The area under the plasma concentration time curve (AUC) and the average maximum plasma concentration showed that the absorption pharmacokinetics was linearly related to the dose. Single-dose studies have shown that the oral bioavailability of buflomedil is about 50% ~ 80% compared with the AUC of various oral or intravenous dosage forms. After intravenous injection of 50, 100 or 200mg, the apparent distribution volume of the drug is 82 ~ 109L and the central chamber is 25 ~ 38L. These values reflect the wide distribution in peripheral chambers and tissues. At human therapeutic concentrations, the drug binds to plasma proteins 60% ~ 80%. The total body clearance rate was 16 ~ 38L/h. The kidney 2 ~ 7L/h, metabolism 12 ~ 19L/h. It indicates that the metabolite of the drug plays an important role in excretion. About 20% of the inhalation dose is eliminated by the first pass, and 4d after oral administration, the 90% of the dose is excreted in the urine, and about 20% is the prototype. |
| Preparation method | 1. 4-pyrrolidinylbutyronitrile is obtained by condensation of 4-chlorobutyronitrile with pyrrolidine, and then with 1,3, 5-trimethoxybenzene The Houben-Hoesch reaction produces buflomedil hydrochloride. The reaction is shown in Figure 1: Figure 1 is the synthetic route of buflomedil hydrochloride 2. The route one is improved, and 4-chloropyrrolylbutyronitrile is prepared by dehydration and N-alkylation with 4-chloropedamide as raw material, avoiding the use of highly toxic sodium cyanide, eliminating the danger in the preparation operation. The rest of the process is basically similar to route one. The synthesis route is shown in Figure 2: Figure 2 is the synthesis route of buflomedil hydrochloride 3. The 1,3, 5-trimethoxybenzene is reacted with butyl lithium to prepare lithium 1,3, 5-trimethoxybenzene, and then react with γ-butyrolactone to prepare 2,4, 6-trimethoxybenzene-γ-hydroxypropyl ketone, and then react with thionyl chloride to generate 2,4, 6-trimethoxybenzene-γ-chloropropyl ketone, finally, the finished product of buflomedil hydrochloride was prepared with pyrrolidine. The synthetic route is shown in Figure 3: Figure 3 is the synthetic route of buflomedil hydrochloride 4. The finished buflomedil hydrochloride can be prepared by Friedel-Crafts reaction with 4-chlorobutyyl chloride and 1,3, 5-trimethoxybenzene-γ-chloropropyl ketone, and then reacted with pyrrolidine to prepare the finished buflomedil hydrochloride. The reaction route is shown in Figure 4: Figure 4 is the synthetic route of buflomedil hydrochloride |
| Pharmacodynamics | The main pharmacological effect of this drug on vascular smooth muscle is a non-selective competitive inhibition of alpha adrenergic receptors. Human hemodynamic studies show that the drug increases peripheral arterial blood flow, improves the perfusion of the damaged vascular bed, and has little effect on central hemodynamics. Animal experiments have confirmed that the drug can reduce collateral resistance and increase collateral blood flow, and show the effect of dilating skin and skeletal muscle blood vessels, thereby reducing vascular resistance. The drug can increase the deformability of red blood cells, accompanied by an increase in ATP and cAMP in red blood cells and a decrease in glyceryl 2, 3-diphosphate. The drug can inhibit platelet aggregation induced by various stimulants, reduce blood viscosity and increase red blood cell flow. Isotope tracer showed that single dose (50-200 mg) intravenous injection or arterial injection, as well as long-term treatment of 600mg oral for 14 days), can increase the cerebral blood flow in patients with cerebrovascular disease, hemisphere and whole brain, and poor perfusion of regional blood supply improved the most. |
| clinical application | for skin necrosis caused by severe obstructive vascular disease. The clinical effective rate (obvious or complete improvement of skin lesions) was 50% ~ 90%. The drug has been shown to be promising in the treatment of diseases such as Raynaud's phenomenon, diabetic retinopathy, frostbite and painful malnutrition. Clinical trials show that the drug 450~600mg/d can significantly reduce the recognition ability and physical and mental function decline of patients with cerebral insufficiency and senile dementia. In the control study, the drug is significantly better than placebo, and slightly better than cinnarizine, flunarizine, ergot nicotine, vinemide and Co-dergocrine mesylate. For patients with cochlear-vestibular lesions, the drug 450 mg/d can obviously improve dizziness, tinnitus, vertigo, sudden deafness and other symptoms. There are signs that the drug is best for people with circulatory disorders, suggesting that the drug can improve the microcirculation of the cochlea and brainstem. (2016-03-03) |
| usage and dosage | the recommended oral dose is 450~600mg (tablets or drops) for 1 day, usually divided into 2~3 times. Some countries have 600mg of sustained-release agent, which can be taken once a day. Intravenous injection is generally 50~200 mg for 1 day, divided or continuous slow infusion. You can also inject 50mg intramuscularly 3 times a day for up to 14 days. |
| adverse reactions | buflomedil can be well tolerated by taking 300~600mg of buflomedil on the 1st, and it is rarely necessary to stop the drug (about 1%). The most common side effects are flushing, headache, dizziness, gastrointestinal discomfort and dizziness. |
| precautions | 1. those who are allergic to this product, postpartum, severe arterial hemorrhage, acute cerebral hemorrhage, acute myocardial infarction, angina pectoris, hyperthyroidism, paroxysmal tachycardia, patients with other bleeding tendency or massive blood loss in the near future are prohibited. 2. Patients with hypofunction of liver and kidney function should adjust the dose appropriately; those who are allergic to this drug, pregnant women, lactating women, and children should be used with caution. 3. This product can cause dizziness or drowsiness. Workers who need high concentration should use it with caution. 4. The infusion of this product is buflomedil hydrochloride and sodium chloride injection, I .e. 250ml contains 0.1g buflomedil and 2.25g sodium chloride. |
| use | is a cerebral vasodilator with multiple drug effects |
| category | toxic substances |
| toxicity classification | highly toxic |
| acute toxicity | oral-rat LD50: 410 mg/kg; Oral-mouse LD50: 275 mg/kg |
| flammability hazard characteristics | thermal decomposition discharges toxic nitrogen oxides and hydrogen chloride smoke |
| storage and transportation characteristics | warehouse ventilation and low temperature drying; Separate from food raw materials |
| fire extinguishing agent | water, dry powder, dry sand, carbon dioxide, foam, 1211 fire extinguishing agent |
Last Update:2024-04-09 20:49:11
Supplier List
Spot supply
Product Name: Buflomedil HCl Visit Supplier Webpage Request for quotationCAS: 35543-24-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Buflomedil Hydrochloride Request for quotation
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: 1-[3-(2,4,6-Trimethoxybenzoyl)propyl]pyrrolidinium chloride Request for quotation
CAS: 35543-24-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 35543-24-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: Buflomedil Hydrochloride Visit Supplier Webpage Request for quotation
CAS: 35543-24-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 35543-24-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Buflomedil HCl Visit Supplier Webpage Request for quotationCAS: 35543-24-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Buflomedil Hydrochloride Request for quotation
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: 1-[3-(2,4,6-Trimethoxybenzoyl)propyl]pyrrolidinium chloride Request for quotation
CAS: 35543-24-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 35543-24-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: Buflomedil Hydrochloride Visit Supplier Webpage Request for quotation
CAS: 35543-24-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 35543-24-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



